Abstract
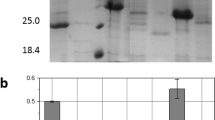
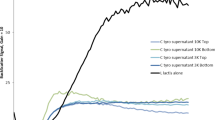
Pediococcus acidilactici ATCC 8042 is a lactic acid bacteria that inhibits pathogenic microorganisms such as Staphylococcus aureus through the production of two proteins with lytic activity, one of 110 kDa and the other of 99 kDa. The 99-kDa one has high homology to a putative peptidoglycan hydrolase (PGH) enzyme reported in the genome of P. acidilactici 7_4, where two different lytic domains have been identified but not characterized. The aim of this work was the biochemical characterization of the recombinant enzyme of 99 kDa. The enzyme was cloned and expressed successfully and retains its activity against Micrococcus lysodeikticus. It has a higher N-acetylglucosaminidase activity, but the N-acetylmuramoyl-l-alanine amidase can also be detected spectrophotometrically. The protein was then purified using gel filtration chromatography. Antibacterial activity showed an optimal pH of 6.0 and was stable between 5.0 and 7.0. The optimal temperature for activity was 60 °C, and all activity was lost after 1 h of incubation at 70 °C. The number of strains susceptible to the recombinant 99-kDa enzyme was lower than that susceptible to the mixture of the 110- and 99-kDa PGHs of P. acidilactici, a result that suggests synergy between these two enzymes. This is the first PGH from LAB that has been shown to possess two lytic sites. The results of this study will aid in the design of new antibacterial agents from natural origin that can combat foodborne disease and improve hygienic practices in the industrial sector.





Similar content being viewed by others
References
Basu B, Banik A, Das M (2008) Production and characterization of extracellular protease of mutant Aspergillus niger AB100 grown on fish scale. World J Microbiol Biotechnol 24:449–455. doi:10.1007/s11274-007-9492-6
Bradford MM (1976) A rapid and sensitive method for the quantitation of micrograms quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/j.bbrc.2008.07.102
Bublitz M, Polle L, Holland C, Heinz DW, Nimtz M, Schubert WD (2009) Structural basis for autoinhibition and activation of Auto, a virulence-associated peptidoglycan hydrolase of Listeria monocytogenes. Mol Microbiol 71:1509–22. doi:10.1111/j.1365-2958.2009.06619.x
Callewaert L, Walmagh M, Michiels C, Lavigne R (2011) Food applications of bacterial cell wall hydrolases. Curr Opin Biotech 22:164–171. doi:10.1016/j.copbio.2010.10.012
Cheng Q, Fischetti V (2007) Mutagenesis of a bacteriophage lytic enzyme PlyGBS significantly increases its antibacterial activity against group B streptococci. Appl Microbiol Biotechnol 74:1284–1291. doi:10.1007/s00253-006-0771-1
Claperon C, Rozenfeld R, Iturrioz X, Inguimbert N, Okada M, Roques B, Maigret B, Llorens-Cortes C (2008) Asp218 participates with Asp213 to bind a Ca2+ atom into the S1 subsite of aminopeptidase A: a key element for substrate specificity. Biochem J 416:37–46. doi:10.1042/BJ20080471
Corpet F (1988) Multiple sequence alignment with hierarchical clustering. Nucl Acids Res 16:10881–10890. doi:10.1093/nar/16.22.10881
DeWitt T, Grossman AD (2014) The bifunctional cell wall hydrolase CwlT is needed for conjugation of the integrative and conjugative element ICEBs1 in Bacillus subtilis and B. anthracis. J Bacteriol 196:1588–96. doi:10.1128/JB.00012-14
Dziarski R, Gupta D (2006) The peptidoglycan recognition proteins (PGRPs). Genome Biol 7:23. doi:10.1186/gb-2006-7-8-232
Fenton M, Ross RP, McAuliffe O, O’Mahony J, Coffey A (2011) Characterization of the staphylococcal bacteriophage lysin CHAP(K). J Appl Microbiol 111:1025–1035. doi:10.1111/j.1365-2672.2011.05119.x
Ferré F, Clote P (2005) DiANNA: a web server for disulfide connectivity prediction. Nucleic Acids Res 1;33(Web Server issue):W230-2. doi: 10.1093/nar/gki412
Fischetti VA (2010) Bacteriophage endolysins: a novel anti-infective to control Gram-positive pathogens. Int J Med Microbiol 300:357–62. doi:10.1016/j.ijmm.2010.04.002
Fukushima T, Yao Y, Kitajima T, Yamamoto H, Sekiguchi J (2007) Characterization of new L, D-endopeptidase gene product CwlK (previous YcdD) that hydrolyzes peptidoglycan in Bacillus subtilis. Mol Genet Genomics 278:371–383. doi:10.1007/s00438-007-0255-8
Garcia P, Rodriguez L, Rodriguez A, Martinez B (2010) Food biopreservation: promising strategies using bacteriocins, bacteriophages and endolysins. Trends Food Sci Technol 21:373–382. doi:10.1016/j.tifs.2010.04.010
García-Cano I, Velasco-Pérez L, Rodríguez-Sanoja R, Sánchez S, Mendoza-Hernández G, Llorente-Bousquets A, Farrés A (2011) Detection, cellular localization and antibacterial activity of two lytic enzymes of Pediococcus acidilactici ATCC 8042. J App Microbiol 111:607–615. doi:10.1111/j.1365-2672.2011.05088.x
García-Cano I, Serrano-Maldonado CE, Olvera-García M, Delgado-Arciniega E, Peña-Montes C, Mendoza-Hernández G, Quirasco M (2014) Antibacterial activity produced by Enterococcus spp. isolated from an artisanal Mexican dairy product, Cotija cheese. LWT -Food Sci Tech 59:26–34. doi:10.1016/j.lwt.2014.04.059
Hermoso JA, García JL, García P (2007) Taking aim on bacterial pathogens: from phage therapy to enzybiotics. Curr Opin Microbiol 10:461–72. doi:10.1016/j.mib.2007.08.002
Ho-Shing W, Ming-Ju T (2004) Kinetics of tributyrin hydrolysis by lipase. Enzyme Microb Tech 35:488–493. doi:10.1016/j.enzmictec.2004.08.002
Kibbe WA (2007) OligoCalc: an online oligonucleotide properties calculator. Nucleic Acids Res 35:W43–46. doi:10.1093/nar/gkm234
Koraimann G (2003) Lytic transglycosylases in macromolecular transport systems of Gram-negative bacteria. Cell Mol Life Sci 60:2371–88. doi:10.1007/s00018-003-3056-1
Kyomuhendo P, Myrnes B, Nilsen I (2007) A cold-active salmon goose-type lysozyme with high heat tolerance. Cell Mol Life Sci 64:2841–2847. doi:10.1007/s00018-007-7372-8
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 15:680–685. doi:10.1038/227680a0
Lavigne R, Briers Y, Hertveldt K, Robben J, Volckaert G (2004) Identification and characterization of a highly thermostable bacteriophage lysozyme. Cell Mol Life Sci 61:2753–2759. doi:10.1007/s00018-004-4301-y
Layec S, Decaris B, Leblond-Bourget N (2008) Diversity of Firmicutes peptidoglycan hydrolases and specificities of those involved in daughter cell separation. Res Microbiol 159:507–515. doi:10.1016/j.resmic.2008.06.008
Leclerc D, Asselin A (1989) Detection of bacterial cell wall hydrolases after denaturing gel electrophoresis. Can J Microbiol 35:749–753. doi:10.1139/m89-125
Maidment C, Dyson A, Beard J (2009) A study into measuring the antibacterial activity of lysozyme-containing foods. Nut Food Sci 39:29–35. doi:10.1108/00346650910930798
McCafferty DG, Lessard IA, Walsh CT (1997) Mutational analysis of potential zinc-binding residues in the active site of the enterococcal D-Ala-D-Ala dipeptidase VanX. Biochemistry 36:10498–10505. doi:10.1021/bi970543u
McLaughlan A, Foster S (1997) Characterisation of the peptidoglycan hydrolases of Listeria monocytogenes EGD. FEMS Microbiol Lett 152:149–154. doi:10.1111/j.1574-6968.1997.tb10421.x
Mellroth P, Sandalova T, Kikhney A, Vilaplana F, Hesek D, Lee M, Mobashery S, Normark S, Svergun D, Henriques-Normark B, Achour A (2014) Structural and functional insights into peptidoglycan access for the lytic amidase LytA of Streptococcus pneumoniae. MBio 5:e01120–13. doi:10.1128/mBio.01120-13
Mendes de Souza P, Fernández A, López-Carballo G, Gavara R, Hernández-Muñoz P (2010) Modified sodium caseinate films as releasing carriers of lysozyme. Food Hydrocolloid 24:300–306. doi:10.1016/j.foodhyd.2009.10.005
Mora D, Fortina MG, Parini C, Daffonchio D, Manachini PL (2000) Genomic subpopulations within the species Pediococcus acidilactici detected by multilocus typing analysis: relationships between pediocin AcH/PA-1 producing and non-producing strains. Microbiology 146:2027–2038
Morner S, Braun V (1984) Murein hydrolase (N-acetyl-muramyl-L-alanine amidase) in human serum. Arch Microbiol 140:171–177. doi:10.1007/BF00454921
Muralidharan V, Goldberg DE (2013) Asparagine repeats in Plasmodium falciparum proteins: good for nothing? PLoS Pathog 9, e1003488. doi:10.1371/journal.ppat.1003488
Nakimbugwe D, Masschalck B, Deckers D, Callewaert L, Aertsen A, Michiels CW (2006) Cell wall substrate specificity of six different lysozymes and lysozyme inhibitory activity of bacterial extracts. FEMS Microbiol Lett 259:41–6. doi:10.1111/j.1574-6968.2006.00240.x
Sabala I, Jonsson I, Tarkowski A, Bochtler M (2012) Anti-staphylococcal activities of lysostaphin and LytM catalytic domain. BMC Microbiol 12:97. doi:10.1186/1471-2180-12-97
Schmelcher M, Waldherr F, Loessner M (2012) Listeria bacteriophage peptidoglycan hydrolases feature high thermoresistance and reveal increased activity after divalent metal cation substitution. Appl Microbiol Biotechnol 93:633–643. doi:10.1007/s00253-011-3372-6
Son B, Yun J, Lim JA, Shin H, Heu S, Ryu S (2012) Characterization of LysB4, an endolysin from the Bacillus cereus-infecting bacteriophage B4. BMC Microbiol 15:12–33. doi:10.1186/1471-2180-12-33
Sugahara K, Yokoi K, Nakamura Y, Nishino T, Yamakawa A, Taketo A, Kodaira K (2007) Mutational and biochemical analyses of the endolysin LysgaY encoded by the Lactobacillus gasseri JCM 1131T phage φgaY. Gene 404:41–52. doi:10.1016/j.gene.2007.08.023
Szweda P, Schielmann M, Kotlowski R, Gorczyca G, Zalewska M, Milewski S (2012) Peptidoglycan hydrolases-potential weapons against Staphylococcus aureus. Appl Microbiol Biotechnol 96:1157–1174. doi:10.1007/s00253-012-4484-3
Turner M, Waldherr F, Loessner M, Giffard P (2007) Antimicrobial activity of lysostaphin and a Listeria monocytogenes bacteriophage endolysin produced and secreted by lactic acid bacteria. Syst Appl Microbiol 30:58–67. doi:10.1016/j.syapm.2006.01.013
Vollmer W, Joris B, Charlier P, Foster S (2008) Bacterial peptidoglycan (murein) hydrolases. FEMSMicrobiol Rev 32:259–286. doi:10.1111/j.1574-6976.2007.00099.x
Xu Q, Chiu HJ, Farr CL, Jaroszewski L, Knuth MW, Miller MD, Lesley SA, Godzik A, Elsliger MA, Deacon AM, Wilson IA (2014) Structures of a bifunctional cell wall hydrolase CwlT containing a novel bacterial lysozyme and an NlpC/P60 DL-endopeptidase. J Mol Biol 426:169–84. doi:10.1016/j.jmb.2013.09.011
Yoke-Ming W, Yu H, Kumar S, Yoshiko M, Keiji N (2015) Optimization of Poly(N-isopropylacrylamide) as an artificial amidase. Biomacromolecules 16:411–421. doi:10.1021/bm501671r
Yoshimura G, Komatsuzawa H, Hayashi I, Fujiwara T, Yamada S, Nakano Y, Tomita Y, Kozai K, Sugai M (2006) Identification and molecular characterization of an N-acetylmuraminidase, Aml, involved in Streptococcus mutans cell separation. Microbiol Immunol 50:729–4. doi:10.1007/s12275-012-2187-1
Zhang X (2012) Diversity analysis of peptidoglycan hydrolases in lactic acid bacteria and their phages. Int Dairy J 25:60–65. doi:10.1016/j.idairyj.2012.02.003
Zoll S, Pätzold B, Schlag M, Götz F, Kalbacher H, Stehle T (2010) Structural basis of cell wall cleavage by a staphylococcal autolysin. PLoS Pathog 6:e1000807. doi:10.1371/journal.ppat.1000807
Acknowledgments
This work was partially funded by PAPIIT-DGAPA-UNAM IN210606. I.G.-C. received a scholarship from CONACyT 208631. We appreciate English review by American Journal Experts.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
García-Cano, I., Campos-Gómez, M., Contreras-Cruz, M. et al. Expression, purification, and characterization of a bifunctional 99-kDa peptidoglycan hydrolase from Pediococcus acidilactici ATCC 8042. Appl Microbiol Biotechnol 99, 8563–8573 (2015). https://doi.org/10.1007/s00253-015-6593-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6593-2