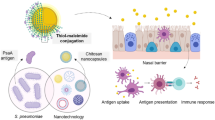
Abstract
Pseudomonas aeruginosa is an opportunistic pathogen that localizes to and colonizes mucosal tissue. Thus, vaccines that elicit a strong mucosal response against P. aeruginosa should be superior to other vaccination strategies. In this study, to stimulate rapid and enhanced mucosal immune responses, mannose-modified chitosan microspheres loaded with the recombinant outer membrane protein OprF190–342-OprI21–83 (FI) (FI-MCS-MPs) of P. aeruginosa were developed as a potent subunit vaccine for mucosal delivery. FI-MCS-MPs were successfully obtained via the tripolyphosphate ionic crosslinking method. Confocal and immunohistochemical analyses indicated that FI-MCS-MPs exhibited the ability to bind the macrophage mannose receptor (MMR, CD206) in vitro and in vivo. After intranasal immunization of mice with FI-MCS-MPs, FI-specific humoral immune responses were detected, measured as local IgM antibody titers in lung tissue slurry; IgA antibody titers in nasal washes, bronchoalveolar lavage (BAL), and intestinal lavage; and systemic IgA and IgG antibody titers in serum. FI-MCS-MPs induced early and high mucosal and systemic humoral antibody responses comparable to those in the group vaccinated with unmodified mannose. High levels of IFN-γ and IL-4 in addition to T lymphocyte subsets induced a mixed Th1/Th2 response in mice immunized with FI-MCS-MPs, resulting in the establishment of cellular immunity. Additionally, when immunized mice were challenged with P. aeruginosa via the nasal cavity, FI-MCS-MPs demonstrated 75 % protective efficacy. Together, these data indicate that mannose-modified chitosan microspheres are a promising subunit delivery system for vaccines against P. aeruginosa infection.








Similar content being viewed by others
References
Asanuma H, Thompson AH, Iwasaki T, Sato Y, Inaba Y, Aizawa C, Kurata T, Tamura S (1997) Isolation and characterization of mouse nasal-associated lymphoid tissue. J Immunol Methods 202(2):123–131
Ausubel FM (1989) Short protocols in molecular biology: a compendium of methods from Current protocols in molecular biology. Greene Pub Associates and Wiley-Interscience, New York
Bassetti M, Villa G, Pecori D (2014) Antibiotic-resistant Pseudomonas aeruginosa: focus on care in patients receiving assisted ventilation. Future Microbiol 9(4):465–474. doi:10.2217/fmb.14.7
Baumann U, Gocke K, Gewecke B, Freihorst J, von Specht BU (2007) Assessment of pulmonary antibodies with induced sputum and bronchoalveolar lavage induced by nasal vaccination against Pseudomonas aeruginosa: a clinical phase I/II study. Respir Res 8:57. doi:10.1186/1465-9921-8-57
Benoudiba F, Toulgoat F, Sarrazin JL (2011) Skull base osteomyelitis. J Radiol 92(11):987–994. doi:10.1016/j.jradio.2011.09.004
Biswal I, Arora BS, Kasana D, Neetushree (2014) Incidence of multidrug resistant pseudomonas aeruginosa isolated from burn patients and environment of teaching institution. J Clin Diagn Res 8(5):DC26–DC29. doi:10.7860/JCDR/2014/7483.4383
Caldas AR, Brandao M, Paula FS, Castro E, Farinha F, Marinho A (2012) Hypertrophic cranial pachymeningitis and skull base osteomyelitis by pseudomonas aeruginosa: case report and review of the literature. J Clin Med Res 4(2):138–144. doi:10.4021/jocmr777w
Chieppa M, Bianchi G, Doni A, Del Prete A, Sironi M, Laskarin G, Monti P, Piemonti L, Biondi A, Mantovani A, Introna M, Allavena P (2003) Cross-linking of the mannose receptor on monocyte-derived dendritic cells activates an anti-inflammatory immunosuppressive program. J Immunol 171(9):4552–4560
Cripps AW, Peek K, Dunkley M, Vento K, Marjason JK, McIntyre ME, Sizer P, Croft D, Sedlak-Weinstein L (2006) Safety and immunogenicity of an oral inactivated whole-cell Pseudomonas aeruginosa vaccine administered to healthy human subjects. Infect Immun 74(2):968–974. doi:10.1128/Iai.74.2.968-974.2006
da Silva SH, Colombo AL, Blotta MH, Queiroz-Telles F, Lopes JD, de Camargo ZP (2005) Diagnosis of neuroparacoccidioidomycosis by detection of circulating antigen and antibody in cerebrospinal fluid. J Clin Microbiol 43(9):4680–4683. doi:10.1128/JCM. 43.9.4680-4683.2005
Davies J (1996) Bacteria on the rampage. Nature 383(6597):219–220. doi:10.1038/383219a0
de Abreu PM, Farias PG, Paiva GS, Almeida AM, Morais PV (2014) Persistence of microbial communities including Pseudomonas aeruginosa in a hospital environment: a potential health hazard. BMC Microbiol 14:118. doi:10.1186/1471-2180-14-118
del Campo J, Zayas C, Romeu B, Acevedo R, Gonzalez E, Bracho G, Cuello M, Cabrera O, Balboa J, Lastre M (2009) Mucosal immunization using proteoliposome and cochleate structures from Neisseria meningitidis serogroup B induce mucosal and systemic responses. Methods 49(4):301–308. doi:10.1016/j.ymeth.2009.03.025
Driscoll JA, Brody SL, Kollef MH (2007) The epidemiology, pathogenesis and treatment of Pseudomonas aeruginosa infections. Drugs 67(3):351–368. doi:10.2165/00003495-200767030-00003
Dunkley ML, Rajyaguru S, McCue A, Cripps AW, Kyd JM (2003) Pseudomonas aeruginosa-specific IgG1 and IgG2 subclasses in enhancement of pulmonary clearance following passive immunisation in the rat. Fems Immunol Med Mic 39(1):37–44. doi:10.1016/S0928-8244(03)00176-7
Engering AJ, Cella M, Fluitsma DM, Hoefsmit EC, Lanzavecchia A, Pieters J (1997) Mannose receptor mediated antigen uptake and presentation in human dendritic cells. Adv Exp Med Biol 417:183–187
Fishman MA, Perelson AS (1999) Th1/Th2 differentiation and cross-regulation. B Math Biol 61(3):403–436. doi:10.1006/bulm.1998.0074
Gavini E, Hegge AB, Rassu G, Sanna V, Testa C, Pirisino G, Karlsen J, Giunchedi P (2006) Nasal administration of carbamazepine using chitosan microspheres: in vitro/in vivo studies. Int J Pharm 307(1):9–15. doi:10.1016/j.ijpharm.2005.09.013
Glik J, Kawecki M, Gazdzik T, Nowak M (2012) The impact of the types of microorganisms isolated from blood and wounds on the results of treatment in burn patients with sepsis. Pol Przegl Chir 84(1):6–16. doi:10.2478/v10035-012-0002-7
Gocke K, Baumann U, Hagemann H, Gabelsberger J, Hahn H, Freihorst J, von Specht BU (2003) Mucosal vaccination with a recombinant OprF-I vaccine of Pseudomonas aeruginosa in healthy volunteers: comparison of a systemic vs. a mucosal booster schedule. FEMS Immunol Med Microbiol 37(2–3):167–171
Hancock RE, Mutharia LM, Chan L, Darveau RP, Speert DP, Pier GB (1983) Pseudomonas aeruginosa isolates from patients with cystic fibrosis: a class of serum-sensitive, nontypable strains deficient in lipopolysaccharide O side chains. Infect Immun 42(1):170–177
Hansen S, Lehr CM (2012) Nanoparticles for transcutaneous vaccination. Microb Biotechnol 5(2):156–167. doi:10.1111/j.1751-7915.2011.00284.x
Horak D, Babic M, Jendelova P, Herynek V, Trchova M, Pientka Z, Pollert E, Hajek M, Sykova E (2007) D-mannose-modified iron oxide nanoparticles for stem cell labeling. Bioconjug Chem 18(3):635–644. doi:10.1021/bc060186c
Horan TC, White JW, Jarvis WR, Emori TG, Culver DH, Munn VP, Thornsberry C, Olson DR, Hughes JM (1986) Nosocomial infection surveillance, 1984. MMWR CDC Surveill Summ 35(1):17SS–29SS
Horton RM, Hunt HD, Ho SN, Pullen JK, Pease LR (1989) Engineering hybrid genes without the use of restriction enzymes: gene splicing by overlap extension. Gene 77(1):61–68
Illum L, Jabbal-Gill I, Hinchcliffe M, Fisher AN, Davis SS (2001) Chitosan as a novel nasal delivery system for vaccines. Adv Drug Deliv Rev 51(1–3):81–96
Irache JM, Salman HH, Gamazo C, Espuelas S (2008) Mannose-targeted systems for the delivery of therapeutics. Expert Opin Drug Del 5(6):703–724. doi:10.1517/17425247.5.6.703
Jiang HL, Park IK, Shin NR, Kang SG, Yoo HS, Kim SI, Suh SB, Akaike T, Cho CS (2004) In vitro study of the immune stimulating activity of an atrophic [correction of athrophic] rhinitis vaccine associated to chitosan microspheres. Eur J Pharm Biopharm 58(3):471–476. doi:10.1016/j.ejpb.2004.05.006
Jiang HL, Kang ML, Quan JS, Kang SG, Akaike T, Yoo HS, Cho CS (2008) The potential of mannosylated chitosan microspheres to target macrophage mannose receptors in an adjuvant-delivery system for intranasal immunization. Biomaterials 29(12):1931–1939. doi:10.1016/j.biomaterials.2007.12.025
Jiang T, Singh B, Li HS, Kim YK, Kang SK, Nah JW, Choi YJ, Cho CS (2014) Targeted oral delivery of BmpB vaccine using porous PLGA microparticles coated with M cell homing peptide-coupled chitosan. Biomaterials 35(7):2365–2373. doi:10.1016/j.biomaterials.2013.11.073
Johansen HK (1996) Potential of preventing Pseudomonas aeruginosa lung infections in cystic fibrosis patients: experimental studies in animals. APMIS Suppl 63:5–42
Jovic S, Shikhagaie M, Morgelin M, Kjellstrom S, Erjefalt J, Olin AI, Frick IM, Egesten A (2014) Expression of MIG/CXCL9 in cystic fibrosis and modulation of its activities by elastase of Pseudomonas aeruginosa. J Innate Immun. doi:10.1159/000365399
Kang ML, Jiang HL, Kang SG, Guo DD, Lee DY, Cho CS, Yoo HS (2007) Pluronic F127 enhances the effect as an adjuvant of chitosan microspheres in the intranasal delivery of Bordetella bronchiseptica antigens containing dermonecrotoxin. Vaccine 25(23):4602–4610. doi:10.1016/j.vaccine.2007.03.038
Kang ML, Kang SG, Jiang HL, Gu DD, Lee DY, Rayamahji N, Seo YS, Ch CS, Yoo HS (2008) Chitosan microspheres containing Bordetella bronchiseptica antigens as novel vaccine against atrophic rhinitis in pigs. J Microbiol Biotechn 18(6):1179–1185
Keler T, Ramakrishna V, Fanger MW (2004) Mannose receptor-targeted vaccines. Expert Opin Biol Ther 4(12):1953–1962. doi:10.1517/14712598.4.12.1953
Knapp B, Shaw A, Hundt E, Enders B, Kupper HA (1988) A histidin alanine rich recombinant antigen protects Aotus monkeys from P. falciparum infection. Behring Inst Mitt 82:349–359
Knapp B, Hundt E, Lenz U, Hungerer KD, Gabelsberger J, Domdey H, Mansouri E, Li Y, von Specht BU (1999) A recombinant hybrid outer membrane protein for vaccination against Pseudomonas aeruginosa. Vaccine 17(13–14):1663–1666
Krause A, Whu WZ, Xu Y, Joh J, Crystal RG, Worgall S (2011) Protective anti-Pseudomonas aeruginosa humoral and cellular mucosal immunity by AdC7-mediated expression of the P. aeruginosa protein OprF. Vaccine 29(11):2131–2139. doi:10.1016/j.vaccine.2010.12.087
Larbig M, Mansouri E, Freihorst J, Tummler B, Kohler G, Domdey H, Knapp B, Hungerer KD, Hundt E, Gabelsberger J, von Specht BU (2001) Safety and immunogenicity of an intranasal Pseudomonas aeruginosa hybrid outer membrane protein F-I vaccine in human volunteers. Vaccine 19(17–19):2291–2297
Lycke N (2012) Recent progress in mucosal vaccine development: potential and limitations. Nat Rev Immunol 12(8):592–605. doi:10.1038/Nri3251
Mansouri E, Gabelsberger J, Knapp B, Hundt E, Lenz U, Hungerer KD, Gilleland HE, Staczek J, Domdey H, von Specht BU (1999) Safety and immunogenicity of a Pseudomonas aeruginosa hybrid outer membrane protein F-I vaccine in human volunteers. Infect Immun 67(3):1461–1470
Mansouri E, Blome-Eberwein S, Gabelsberger J, Germann G, von Specht BU (2003) Clinical study to assess the immunogenicity and safety of a recombinant Pseudomonas aeruginosa OprF-OprI vaccine in burn patients. FEMS Immunol Med Microbiol 37(2–3):161–166
Mayr UB, Kudela P, Atrasheuskaya A, Bukin E, Ignatyev G, Lubitz W (2012) Rectal single dose immunization of mice with Escherichia coli O157:H7 bacterial ghosts induces efficient humoral and cellular immune responses and protects against the lethal heterologous challenge. Microb Biotechnol 5(2):283–294. doi:10.1111/j.1751-7915.2011.00316.x
McNeela EA, O’Connor D, Jabbal-Gill I, Illum L, Davis SS, Pizza M, Peppoloni S, Rappuoli R, Mills KHG (2000) A mucosal vaccine against diphtheria: formulation of cross reacting material (CRM197) of diphtheria toxin with chitosan enhances local and systemic antibody and Th2 responses following nasal delivery. Vaccine 19(9–10):1188–1198. doi:10.1016/S0264-410x(00)00309-1
McNeela EA, Jabbal-Gill I, Illum L, Pizza M, Rappuoli R, Podda A, Lewis DJ, Mills KH (2004) Intranasal immunization with genetically detoxified diphtheria toxin induces T cell responses in humans: enhancement of Th2 responses and toxin-neutralizing antibodies by formulation with chitosan. Vaccine 22(8):909–914. doi:10.1016/j.vaccine.2003.09.012
Mishell BB, Shiigi SM (1980) Selected methods in cellular immunology. W. H. Freeman, San Francisco
Park SA, Cho SS, Kwak GJ (2014) Factors influencing ventilator-associated pneumonia in cancer patients. Asian Pac J Cancer Prev 15(14):5787–5791
Podschun R, Ullmann U (1998) Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin Microbiol Rev 11(4):589–603
Podschun R, Acktun H, Okpara J, Linderkamp O, Ullmann U, Borneff-Lipp M (1998) Isolation of Klebsiella planticola from newborns in a neonatal ward. J Clin Microbiol 36(8):2331–2332
Pruitt BA Jr, McManus AT, Kim SH, Goodwin CW (1998) Burn wound infections: current status. World J Surg 22(2):135–145
Ramakrishna V, Treml JF, Vitale L, Connolly JE, O’Neill T, Smith PA, Jones CL, He LZ, Goldstein J, Wallace PK, Keler T, Endres MJ (2004) Mannose receptor targeting of tumor antigen pmel17 to human dendritic cells directs anti-melanoma T cell responses via multiple HLA molecules. J Immunol 172(5):2845–2852
Robinson HL (1997) Nucleic acid vaccines: an overview. Vaccine 15(8):785–787
Roitt IM, Delves PJ (1992) Encyclopedia of immunology. Academic, London
Roitt IM, Brostoff J, Male DK (2001) Immunology, 6th edn. Mosby Inc, Edinburgh
Sallusto F, Cella M, Danieli C, Lanzavecchia A (1995) Dendritic cells use macropinocytosis and the mannose receptor to concentrate macromolecules in the major histocompatibility complex class II compartment: downregulation by cytokines and bacterial products. J Exp Med 182(2):389–400
Salman HH, Gamazo C, Campanero MA, Irache JM (2006) Bioadhesive mannosylated nanoparticles for oral drug delivery. J Nanosci Nanotechnol 6(9–10):3203–3209
Samonis G, Vardakas KZ, Kofteridis DP, Dimopoulou D, Andrianaki AM, Chatzinikolaou I, Katsanevaki E, Maraki S, Falagas ME (2014) Characteristics, risk factors and outcomes of adult cancer patients with extensively drug-resistant Pseudomonas aeruginosa infections. Infection 42(4):721–728. doi:10.1007/s15010-014-0635-z
Sedaghat B, Stephenson R, Toth I (2014) Targeting the mannose receptor with mannosylated subunit vaccines. Curr Med Chem 21(30):3405–3418
Sharma A, Krause A, Worgall S (2011) Recent developments for Pseudomonas vaccines. Hum Vaccin 7(10):999–1011. doi:10.4161/hv.7.10.16369
Six A, Bellier B, Thomas-Vaslin V, Klatzmann D (2012) Systems biology in vaccine design. Microb Biotechnol 5(2):295–304. doi:10.1111/j.1751-7915.2011.00321.x
Smith DB, Johnson KS (1988) Single-step purification of polypeptides expressed in Escherichia coli as fusions with glutathione S-transferase. Gene 67(1):31–40
Sorichter S, Baumann U, Baumgart A, Walterspacher S, von Specht BU (2009) Immune responses in the airways by nasal vaccination with systemic boosting against Pseudomonas aeruginosa in chronic lung disease. Vaccine 27(21):2755–2759. doi:10.1016/j.vaccine.2009.03.010
Tan MC, Mommaas AM, Drijfhout JW, Jordens R, Onderwater JJ, Verwoerd D, Mulder AA, van der Heiden AN, Ottenhoff TH, Cella M, Tulp A, Neefjes JJ, Koning F (1997) Mannose receptor mediated uptake of antigens strongly enhances HLA-class II restricted antigen presentation by cultured dendritic cells. Adv Exp Med Biol 417:171–174
Tan JQ, Rouse SL, Li DF, Pye VE, Vogeley L, Brinth AR, El Arnaout T, Whitney JC, Howell PL, Sansom MSP, Caffrey M (2014) A conformational landscape for alginate secretion across the outer membrane of Pseudomonas aeruginosa. Acta Crystallogr D 70:2054–2068. doi:10.1107/S1399004714001850
Taranejoo S, Janmaleki M, Rafienia M, Kamali M, Mansouri M (2011) Chitosan microparticles loaded with exotoxin A subunit antigen for intranasal vaccination against Pseudomonas aeruginosa: an in vitro study. Carbohyd Polym 83(4):1854–1861. doi:10.1016/j.carbpol.2010.10.051
Tummler B, Bosshammer J, Breitenstein S, Brockhausen I, Gudowius P, Herrmann C, Herrmann S, Heuer T, Kubesch P, Mekus F, Romling U, Schmidt KD, Spangenberg C, Walter S (1997) Infections with Pseudomonas aeruginosa in patients with cystic fibrosis. Behring Inst Mitt (98):249–55
Varshosaz J, Sadrai H, Alinagari R (2004) Nasal delivery of insulin using chitosan microspheres. J Microencapsul 21(7):761–774. doi:10.1080/02652040400015403
Velasco E, Thuler LC, Martins CA, Dias LM, Goncalves VM (1997) Nosocomial infections in an oncology intensive care unit. Am J Infect Control 25(6):458–462
Vincenti S, Quaranta G, De Meo C, Bruno S, Ficarra MG, Carovillano S, Ricciardi W, Laurenti P (2014) Non-fermentative gram-negative bacteria in hospital tap water and water used for haemodialysis and bronchoscope flushing: prevalence and distribution of antibiotic resistant strains. Sci Total Environ 499C:47–54. doi:10.1016/j.scitotenv.2014.08.041
von Specht BU, Knapp B, Muth G, Broker M, Hungerer KD, Diehl KD, Massarrat K, Seemann A, Domdey H (1995) Protection of immunocompromised mice against lethal infection with Pseudomonas aeruginosa by active or passive immunization with recombinant P. aeruginosa outer membrane protein F and outer membrane protein I fusion proteins. Infect Immun 63(5):1855–1862
Wang N, Wang T, Zhang M, Chen R, Niu R, Deng Y (2014) Mannose derivative and lipid A dually decorated cationic liposomes as an effective cold chain free oral mucosal vaccine adjuvant-delivery system. Eur J Pharm Biopharm. doi:10.1016/j.ejpb.2014.04.007
Yuki Y, Kiyono H (2009) Mucosal vaccines: novel advances in technology and delivery. Expert Rev Vaccines 8(8):1083–1097. doi:10.1586/Erv.09.61
Zaharoff DA, Rogers CJ, Hance KW, Schlom J, Greiner JW (2007) Chitosan solution enhances both humoral and cell-mediated immune responses to subcutaneous vaccination. Vaccine 25(11):2085–2094. doi:10.1016/j.vaccine.2006.11.034
Zhai RL, Xu HY, Wang YL, Qin ZM, Jiang SJ (2007) Study on immune efficacy of Newcastle disease chitosan microsphere vaccine. Wei Sheng Wu Xue Bao 47(4):692–696
Zhou X, Liu B, Yu X, Zha X, Zhang X, Chen Y, Wang X, Jin Y, Wu Y, Chen Y, Shan Y, Chen Y, Liu J, Kong W, Shen J (2007) Controlled release of PEI/DNA complexes from mannose-bearing chitosan microspheres as a potent delivery system to enhance immune response to HBV DNA vaccine. J Control Release 121(3):200–207. doi:10.1016/j.jconrel.2007.05.018
Acknowledgments
This study was supported by the National High Technology Research and Development Program of China (No. 2011AA10A210). The authors thank Ye Yuan for help with chemically synthesized statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
(PDF 120 kb)
Rights and permissions
About this article
Cite this article
Cui, Z., Han, D., Sun, X. et al. Mannose-modified chitosan microspheres enhance OprF-OprI-mediated protection of mice against Pseudomonas aeruginosa infection via induction of mucosal immunity. Appl Microbiol Biotechnol 99, 667–680 (2015). https://doi.org/10.1007/s00253-014-6147-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-6147-z