Abstract
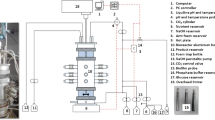
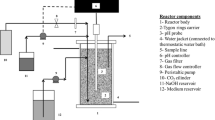
Continuous anaerobic fermentations were performed in a biofilm reactor packed with Poraver® beads. Dilution rates (D) varied between 0.054 and 0.72 h−1, and d-glucose and CO2 gas were used as carbon substrates. Steady-state conditions were shown to be repeatable and independent of the operational history. Production stability was achieved over periods exceeding 80 h at values of D below 0.32 h−1. In these situations, steady-state variation (expressed as fluctuations in NaOH neutralisation flow rates) exhibited a standard deviation of less than 5 % while no indication of biofilm deactivation was detected. The total biomass amount was found to be independent of the dilution rate with an average dry concentration of 23.8 ± 2.9 g L−1 obtained for all runs. This suggests that the attachment area controls the extent of biofilm accumulation. Specific succinic acid (SA) productivities, based on the total biomass amount, exhibited a substantial decrease with decreasing D. An SA volumetric productivity of 10.8 g L−1 h−1 was obtained at D = 0.7 h−1—the highest value reported to date in Actinobacillus succinogenes fermentations. SA yields on glucose increased with decreasing D, with a yield of 0.90 ± 0.01 g g−1 obtained at a D of 0.054 h−1. Production of formic acid approached zero with decreasing D, while the succinic to acetic acid ratio increased with decreasing D, resulting in an increasing SA yield on glucose.







Similar content being viewed by others
References
Balzer GJ, Thakker C, Bennett GN, San KY (2013) Metabolic engineering of Escherichia coli to minimize byproduct formate and improving succinate productivity through increasing NADH availability by heterologous expression of NAD(+)-dependent formate dehydrogenase. Metab Eng 20:1–8. doi:10.1016/j.ymben.2013.07.005
Beauprez JJ, De Mey M, Soetaert WK (2010) Microbial succinic acid production: natural versus metabolic engineered producers. Process Biochem 45:1103–1114. doi:10.1016/j.procbio.2010.03.035
Bechthold I, Bretz K, Kabasci S, Kopitzky R, Springer A (2008) Succinic acid: a new platform chemical for biobased polymers from renewable resources. Chem Eng Technol 31:647–654. doi:10.1002/ceat.200800063
Bozell JJ, Petersen GR (2010) Technology development for the production of biobased products from biorefinery carbohydrates—the US Department of Energy’s “Top 10” revisited. Green Chem 12:539. doi:10.1039/b922014c
Bradfield MFA, Nicol W (2014) Continuous succinic acid production by Actinobacillus succinogenes in a biofilm reactor: steady-state metabolic flux variation. Biochem Eng J 85:1–7. doi:10.1016/j.bej.2014.01.009
Casali S, Gungormusler M, Bertin L, Fava F, Azbar N (2012) Development of a biofilm technology for the production of 1,3-propanediol (1,3-PDO) from crude glycerol. Biochem Eng J 64:84–90. doi:10.1016/j.bej.2011.11.012
Cheng KC, Demirci A, Catchmark JM (2010) Advances in biofilm reactors for production of value-added products. Appl Microbiol Biotechnol 87:445–456. doi:10.1007/s00253-010-2622-3
Corona-González RI, Bories A, González-Álvarez V, Pelayo-Ortiz C (2008) Kinetic study of succinic acid production by Actinobacillus succinogenes ZT-130. Process Biochem 43:1047–1053. doi:10.1016/j.procbio.2008.05.011
Corona-González RI, Bories A, González-Álvarez V, Snell-Castro R, Toriz-González G, Pelayo-Ortiz C (2010) Succinic acid production with Actinobacillus succinogenes ZT-130 in the presence of succinic acid. Curr Microbiol 60:71–77. doi:10.1007/s00284-009-9504-x
Cukalovic A, Stevens CV (2008) Feasibility of production methods for succinic acid derivatives: a marriage of renewable resources and chemical technology. Biofuels Bioprod Biorefin 2:505–529. doi:10.1002/bbb.105
Dagher SF, Ragout AL, Siñeriz F, Bruno-Bárcena JM (2010) Cell immobilization for production of lactic acid: biofilms do it naturally. Adv Appl Microbiol 71:113–148. doi:10.1016/S0065-2164(10)71005-4
Dishisha T, Alvarez MT, Hatti-Kaul R (2012) Batch- and continuous propionic acid production from glycerol using free and immobilized cells of Propionibacterium acidipropionici. Bioresour Technol 118:553–562. doi:10.1016/j.biortech.2012.05.079
Fernando S, Adhikari S, Chandrapal C, Murali N (2006) Biorefineries: current status, challenges, and future direction. Energy Fuel 20:1727–1737. doi:10.1021/ef060097w
Gross R, Hauer B, Otto K, Schmid A (2007) Microbial biofilms: new catalysts for maximizing productivity of long-term biotransformations. Biotechnol Bioeng 98:1123–1134. doi:10.1002/bit.21547
Guettler M, Jain M, Rumler D (1996) Method for making succinic acid, bacterial variants for use in the process, and methods for obatining variants. US Patent 5:573,931
Jantama K, Zhang X, Moore JC, Shanmugam KT, Svoronos SA, Ingram LO (2008) Eliminating side products and increasing succinate yields in engineered strains of Escherichia coli C. Biotechnol Bioeng 101:881–893. doi:10.1002/bit.22005
Kim MI, Kim NJ, Shang L, Chang YK, Lee SY, Chang HN (2009) Continuous production of succinic acid using an external membrane cell recycle system. J Microbiol Biotechnol 19:1369–1373. doi:10.4014/jmb.0903.03034
Lin SKC, Du C, Koutinas A, Wang R, Webb C (2008) Substrate and product inhibition kinetics in succinic acid production by Actinobacillus succinogenes. Biochem Eng J 41:128–135. doi:10.1016/j.bej.2008.03.013
Qureshi N, Annous BA, Ezeji TC, Karcher P, Maddox IS (2005) Biofilm reactors for industrial bioconversion processes: employing potential of enhanced reaction rates. Microb Cell Factories 4:24. doi:10.1186/1475-2859-4-24
Rosche B, Li XZ, Hauer B, Schmid A, Buehler K (2009) Microbial biofilms: a concept for industrial catalysis? Trends Biotechnol 27:636–643. doi:10.1016/j.tibtech.2009.08.001
Transparency-Market-Research (2013) Succinic acid market—global industry analysis, size, share, growth, trends and forecast, 2012–2018. http://www.transparencymarketresearch.com/succinic-acid.html. Accessed 19 February 2014
Urbance SE, Pometto AL, DiSpirito AA, Demirci A (2003) Medium evaluation and plastic composite support ingredient selection for biofilm formation and succinic acid production by Actinobacillus succinogenes. Food Biotechnol 17:53–65. doi:10.1081/FBT-120019984
Urbance SE, Pometto AL, DiSpirito AA, Denli Y (2004) Evaluation of succinic acid continuous and repeat-batch biofilm fermentation by Actinobacillus succinogenes using plastic composite support bioreactors. Appl Microbiol Biotechnol 65:664–670. doi:10.1007/s00253-004-1634-2
Van Heerden CD, Nicol W (2013a) Continuous and batch cultures of Escherichia coli KJ134 for succinic acid fermentation: metabolic flux distributions and production characteristics. Microb Cell Factories 12:80. doi:10.1186/1475-2859-12-80
Van Heerden CD, Nicol W (2013b) Continuous succinic acid fermentation by Actinobacillus succinogenes. Biochem Eng J 73:5–11. doi:10.1016/j.bej.2013.01.015
Wee YJ, Yun JS, Kang KH, Ryu HW (2002) Continuous production of succinic acid by a fumarate-reducing bacterium immobilized in a hollow-fiber bioreactor. Appl Biochem Biotechnol 98–100:1093–1104
Werpy T, Petersen G (2004) Top value added chemicals from biomass, Volume 1—results of screening for potential candidates from sugars and synthesis gas. US Department of Energy. http://www1.eere.energy.gov/bioenergy/pdfs/35523.pdf. Accessed 24 February 2014
Zeikus JG, Jain MK, Elankovan P (1999) Biotechnology of succinic acid production and markets for derived industrial products. Appl Microbiol Biotechnol 51:545–552. doi:10.1007/s002530051431
Acknowledgments
The financial assistance of the National Research Foundation (NRF) towards this research is hereby acknowledged. Opinions expressed and conclusions arrived at are those of the author and are not necessarily to be attributed to the NRF.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maharaj, K., Bradfield, M.F.A. & Nicol, W. Succinic acid-producing biofilms of Actinobacillus succinogenes: reproducibility, stability and productivity. Appl Microbiol Biotechnol 98, 7379–7386 (2014). https://doi.org/10.1007/s00253-014-5779-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-5779-3