Abstract
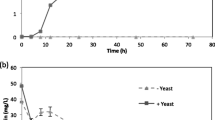
With the increasing demand for biopharmaceutical proteins and industrial enzymes, it is necessary to optimize the production by microbial fermentation or cell cultures. Yeasts are well established for the production of a wide range of recombinant proteins, but there are also some limitations; e.g., metabolic and cellular stresses have a strong impact on recombinant protein production. In this work, we investigated the effect of the specific growth rate on the production of two different recombinant proteins. Our results show that human insulin precursor is produced in a growth-associated manner, whereas α-amylase tends to have a higher yield on substrate at low specific growth rates. Based on transcriptional analysis, we found that the difference in the production of the two proteins as function of the specific growth rate is mainly due to differences in endoplasmic reticulum processing, protein turnover, cell cycle, and global stress response. We also found that there is a shift at a specific growth rate of 0.1 h−1 that influences protein production. Thus, for lower specific growth rates, the α-amylase and insulin precursor-producing strains present similar cell responses and phenotypes, whereas for higher specific growth rates, the two strains respond differently to changes in the specific growth rate.




Similar content being viewed by others
References
Andersen DC, Krummen L (2002) Recombinant protein expression for therapeutic applications. Curr Opin Biotechnol 13(2):117–123
Arvas M, Pakula T, Smit B, Rautio J, Koivistoinen H, Jouhten P, Lindfors E, Wiebe M, Penttilä M, Saloheimo M (2011) Correlation of gene expression and protein production rate—a system wide study. BMC Genomics 12(1):616
Bernales S, Papa FR, Walter P (2006) Intracellular signaling by the unfolded protein response. Annu Rev Cell Dev Biol 22:487
Buchetics M, Dragosits M, Maurer M, Rebnegger C, Porro D, Sauer M, Gasser B, Mattanovich D (2011) Reverse engineering of protein secretion by uncoupling of cell cycle phases from growth. Biotechnol Bioeng 108(10):2403–2412
Castrillo JI, Zeef LA, Hoyle DC, Zhang N, Hayes A, Gardner DCJ, Cornell MJ, Petty J, Hakes L, Wardleworth L (2007) Growth control of the eukaryote cell: a systems biology study in yeast. J Biol 6(2):4
Causton HC, Ren B, Koh SS, Harbison CT, Kanin E, Jennings EG, Lee TI, True HL, Lander ES, Young RA (2001) Remodeling of yeast genome expression in response to environmental changes. Mol Biol Cell 12(2):323–337
Dürrschmid K, Reischer H, Schmidt-Heck W, Hrebicek T, Guthke R, Rizzi A, Bayer K (2008) Monitoring of transcriptome and proteome profiles to investigate the cellular response of E. coli towards recombinant protein expression under defined chemostat conditions. J Biotechnol 135(1):34–44
Fazio A, Jewett M, Daran-Lapujade P, Mustacchi R, Usaite R, Pronk J, Workman C, Nielsen J (2008) Transcription factor control of growth rate dependent genes in Saccharomyces cerevisiae: a three factor design. BMC Genomics 9(1):341
Ferreira B, Calado C, Van Keulen F, Fonseca L, Cabral J, da Fonseca M (2003) Towards a cost effective strategy for cutinase production by a recombinant Saccharomyces cerevisiae: strain physiological aspects. Appl Microbiol Biotechnol 61(1):69–76
Freigassner M, Pichler H, Glieder A (2009) Tuning microbial hosts for membrane protein production. Microb Cell Fact 8(1):69
Fu J, Wilson DB, Shuler ML (1993) Continuous, high level production and excretion of a plasmid-encoded protein by Escherichia coli in a two-stage chemostat. Biotechnol Bioeng 41(10):937–946
Gill R, DeLisa M, Valdes J, Bentley W (2001) Genomic analysis of high-cell-density recombinant Escherichia coli fermentation and “cell conditioning” for improved recombinant protein yield. Biotechnol Bioeng 72(1):85–95
Hackel BJ, Huang D, Bubolz JC, Wang XX, Shusta EV (2006) Production of soluble and active transferrin receptor-targeting single-chain antibody using Saccharomyces cerevisiae. Pharm Res 23(4):790–797
Hardjito L, Greenfield PF, Lee PL (1993) Recombinant protein production via fed-batch culture of the yeast Saccharomyces cerevisiae. Enzyme Microb Technol 15(2):120–126
Hatahet F, Ruddock LW (2009) Modulating proteostasis: peptidomimetic inhibitors and activators of protein folding. Curr Pharm Des 15(21):2488–2507
Haynes CM, Titus EA, Cooper AA (2004) Degradation of misfolded proteins prevents ER-derived oxidative stress and cell death. Mol Cell 15(5):767–776
Hoffmann F, Rinas U (2000) Kinetics of heat-shock response and inclusion body formation during temperature-induced production of basic fibroblast growth factor in high-cell-density cultures of recombinant Escherichia coli. Biotechnol Prog 16(6):1000–1007
Hou J, Tyo K, Liu Z, Petranovic D, Nielsen J (2012a) Engineering of vesicle trafficking improves heterologous protein secretion in Saccharomyces cerevisiae. Metab Eng 14(2):120–127
Hou J, Tyo K, Liu Z, Petranovic D, Nielsen J (2012b) Metabolic engineering of recombinant protein production by Saccharomyces cerevisiae. FEMS Yeast Res 12(5):491–510
Idiris A, Tohda H, Kumagai H, Takegawa K (2010) Engineering of protein secretion in yeast: strategies and impact on protein production. Appl Microbiol Biotechnol 86(2):403–417
Kim IK, Roldão A, Siewers V, Nielsen J (2012) A systems-level approach for metabolic engineering of yeast cell factories. FEMS Yeast Res 12(2):228–248
Langer ES (2012) Biomanufacturing outsourcing outlook. BioPharm International 25(2):15–16
Liu Z, Tyo K, Martínez J, Petranovic D, Nielsen J (2012) Different expression systems for production of recombinant proteins in Saccharomyces cerevisiae. Biotechnol Bioeng 109(5):1259–1268
Lunter G, Goodson M (2011) Stampy: A statistical algorithm for sensitive and fast mapping of Illumina sequence reads. Genome Res 21(6):936–939
Moye-Rowley WS (2002) Transcription factors regulating the response to oxidative stress in yeast. Antioxid Redox Sign 4(1):123–140
Nancib N, Boudrant J (1992) Effect of growth rate on stability and gene expression of a recombinant plasmid during continuous culture of Escherichia coli in a non-selective medium. Biotechnol Lett 14(8):643–648
Nurse P (2003) Systems biology: understanding cells. Nature 424(6951):883–883
Oliveira AP, Patil KR, Nielsen J (2008) Architecture of transcriptional regulatory circuits is knitted over the topology of bio-molecular interaction networks. BMC Syst Biol 2(1):17
Oliveira C, Teixeira JA, Lima N, Da Silva NA, Domingues L (2007) Development of stable flocculent Saccharomyces cerevisiae strain for continuous Aspergillus niger beta-galactosidase production. J Biosci Bioeng 103(4):318–324. doi:10.1263/jbb.103.318
Patil KR, Nielsen J (2005) Uncovering transcriptional regulation of metabolism by using metabolic network topology. Proc Nati Acad Sci USA 102(8):2685–2689
Pincus D, Chevalier MW, Aragón T, Van Anken E, Vidal SE, El-Samad H, Walter P (2010) BiP binding to the ER-stress sensor Ire1 tunes the homeostatic behavior of the unfolded protein response. PLoS Biol 8(7):e1000415. doi:10.1371/journal.pbio.1000415
Regenberg B, Grotkjær T, Winther O, Fausbøll A, Åkesson M, Bro C, Hansen LK, Brunak S, Nielsen J (2006) Growth-rate regulated genes have profound impact on interpretation of transcriptome profiling in Saccharomyces cerevisiae. Genome Biol 7(11):R107
Schröder M (2008) Engineering eukaryotic protein factories. Biotechnol Lett 30(2):187–196
Seresht AK, Palmqvist EA, Olsson L (2011) The impact of phosphate scarcity on pharmaceutical protein production in S. cerevisiae: linking transcriptomic insights to phenotypic responses. Microb Cell Fact 10(1):104
Thomas JG, Baneyx F (2000) ClpB and HtpG facilitate de novo protein folding in stressed Escherichia coli cells. Mol Microbiol 36(6):1360–1370
Tyo K, Liu Z, Petranovic D, Nielsen J (2012) Imbalance of heterologous protein folding and disulfide bond formation rates yields runaway oxidative stress. BMC Biol 10:16
Verripsab T, Duboc P, Visser C, Sagt C (2000) From gene to product in yeast: production of fungal cutinase. Enzyme Microb Technol 26(9–10):812–818
Acknowledgments
We thank Dr. Intawat Nookaew and Ph.D. student Leif Väremo from Chalmers University of Technology for kindly providing the PIANO software for microarray analysis. This work is financially supported by the European Research Council ERC project INSYSBIO (grant no. 247013), the Novo Nordisk Foundation, the Chalmers Foundation, and the Knut and Alice Wallenberg Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 104 kb)
Rights and permissions
About this article
Cite this article
Liu, Z., Hou, J., Martínez, J.L. et al. Correlation of cell growth and heterologous protein production by Saccharomyces cerevisiae . Appl Microbiol Biotechnol 97, 8955–8962 (2013). https://doi.org/10.1007/s00253-013-4715-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-4715-2