Abstract
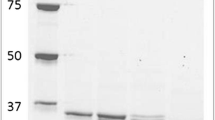
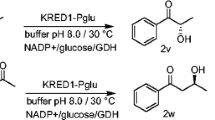
Two genes encoding recombinant cytosolic oxidoreductases from Gluconobacter oxydans, gox0313 and gox0646, were heterologously expressed in Escherichia coli and the resulting proteins were purified and characterized. GOX0313 was identified as a medium-chain alcohol dehydrogenase, whereas GOX0646 was classified as a ketocarbonyl reductase. GOX0313 had a broad substrate spectrum and oxidized various primary alcohols. However, GOX0313 had a preference for substrate reduction, reducing many aldehydes and α-diketones. In contrast, GOX0646 had a narrow substrate spectrum and reduced α-diketones, preferring short-chain ketocarbonyls. Both enzymes regio- and stereospecifically reduced α-diketones to the corresponding (S)-hydroxy ketone, as shown by NMR. These products are difficult to produce chemically, requiring complicated protecting group chemistry. Furthermore, hydroxy ketones find industrial application in the production of pheromones, fragrances, flavors, and pharmaceuticals. Hence, these enzymes are interesting biocatalysts for the production of enantiomerically pure building blocks that are difficult to prepare chemically.



Similar content being viewed by others
References
Adachi O, Moonmangmee D, Toyama H, Yamada M, Shinagawa E, Matsushita K (2003) New developments in oxidative fermentation. Appl Microbiol Biotechnol 60:643–653
Andersson A, Jordan D, Schneider G, Lindqvist Y (1996) Crystal structure of the ternary complex of 1,3,8-trihydroxynaphthalene reductase from Magnaporthe grisea with NADPH and an active-site inhibitor. Structure 4:1161–1170
Armarego WL (1979) Hydrogen transfer from 4-R and 4-S (4–3 H) NADH in the reduction of d, l-cis-6,7-dimethyl-6,7 (8 H) dihydropterin with dihydropteridine reductase from human liver and sheep liver. Biochem Biophys Res Commun 89:246–249
Arnold LJ, You KS, Allison WS, Kaplan NO (1976) Determination of hydride transfer stereospecificity of nicotinamide adenine-dinucleotide linked oxidoreductases by proton magnetic-resonance. Biochemistry 15:4844–4849
Ausubel FM (2002) Preparation and analysis of genomic DNA from bacteria. In: Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Struhl K (eds) Current protocols in molecular biology, vol 5. Wiley, New York, pp 2–11
Bel-Rhlid R, Fauve A, Veschambre H (1989) Synthesis of the pheromone components of the grape borer Xylotrechus pyrrhoderus by microbiological reduction of an α-diketone. J Org Chem 54:3221–3223
Benner SA, Nambiar KP, Chambers GK (1985) A stereochemical imperative in dehydrogenases—new data and criteria for evaluating function-based theories in bioorganic chemistry. J Am Chem Soc 107:5513–5517
Betz G, Warren JC (1968) Reaction mechanism and stereospecificity of 20 β-hydroxysteroid dehydrogenase. Arch Biochem Biophys 128:745–752
Blum H, Beier H, Gross HJ (1987) Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 8:93–99
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cannio R, Rossi M, Bartolucci S (1994) A few amino acid substitutions are responsible for the higher thermostability of a novel NAD(+)-dependent bacillar alcohol dehydrogenase. Eur J Biochem 222(2):345–352
Ceccarelli C, Liang ZX, Strickler M, Prehna G, Goldstein BM, Klinman JP, Bahnson BJ (2004) Crystal structure and amide H/D exchange of binary complexes of alcohol dehydrogenase from Bacillus stearothermophilus: insight into thermostability and cofactor binding. Biochemistry 43:5266–5277
Chen J, Schenker S, Frosto TA, Henderson GI (1998) Inhibition of cytochrome c oxidase activity by 4-hydroxynonenal (HNE). Role of HNE adduct formation with the enzyme subunits. Biochim Biophys Acta 1380:336–344
Coppola GM, Schuster HF (1997) α-Hydroxy acids in enantioselective synthesis. Wiley-VCH, Weinheim
Crout HG, Morrey SM (1983) Synthesis of (R)- and (S)-acetoin (3-hydroxybutan-2-one). J Chem Soc, Perkin Trans 1:2435–2440
De Ley J, Gillis M, Swings J (1984) The genus Gluconobacter. In: Krieg NR, Holt JG (eds) Bergey’s manual of systematic bacteriology, vol 1. Williams and Wilkins, Baltimore, pp 267–278
DeMaster EG, Shirota FN, Nagasawa HT (1986) Role of propiolaldehyde and other metabolites in the pargyline inhibition of rat-liver aldehyde dehydrogenase. Biochem Pharmacol 35:1481–1489
Deppenmeier U, Ehrenreich A (2009) Physiology of acetic acid bacteria in light of the genome sequence of Gluconobacter oxydans. J Mol Microbiol Biotechnol 16:69–80
Deppenmeier U, Hoffmeister M, Prust C (2002) Biochemistry and biotechnological applications of Gluconobacter strains. Appl Microbiol Biotechnol 60:233–242
Do Nascimento KH, Davies DD (1975) Stereospecificity of sequential nicotinamide-adenine dinucleotide-dependent oxidoreductases in relation to evolution of metabolic sequences. Biochem J 149:553–557
Drauz K, Waldman H (eds) (2002) Enzyme catalysis in organic synthesis, vol 1-III. Wiley-VCH, Weinheim, Germany
Fuganti C, Grasselli P (1982) Synthesis of the C14 chromanyl moiety of natural α-tocopherol (vitamin E). J Chem Soc Chem Commun 1982(4):205–206
Gala D, DiBedetto DJ, Clark JE, Murphy BL, Schuhmacher D, Steinman M (1996) Preparations of antifungal Sch 42427/SM 9164: Preparative chromatographic resolution, and total asymmetric synthesis via enzymatic preparation of chiral α-hydroxy arylketones. Tetrahedron Lett 37:611–614
Hall DR, Cork A, Phythian SJ, Chittamuru S, Jayarama BK, Venkatesha MG, Sreedharan K, Vinod Kumar PK, Seetharama HG, Naidu R (2006) Identification of components of male-produced pheromone of coffee white stemborer, Xylotrechus quadripes. J Chem Ecol 32:195–219
Hanke T, Richhardt J, Polen T, Sahm H, Bringer S, Bott M (2012) Influence of oxygen limitation, absence of the cytochrome bc1 complex and low pH on global gene expression in Gluconobacter oxydans 621 H using DNA microarray technology. J Biotechnol 157(3):359–372
Johansen L, Larsen SH, Størmer FC (1973) Diacetyl (acetoin) reductase from Aerobacter aerogenes—kinetic studies of reduction of diacetyl to acetoin. Eur J Biochem 34:97–99
Kavanagh KL, Jörnvall H, Persson B, Oppermann U (2008) The SDR superfamily: functional and structural diversity within a family of metabolic and regulatory enzymes. Cell Mol Life Sci 65:3895–3906
Laemmli UK (1970) Cleavage of structural proteins during the assemblyof the head of bacteriophage T4. Nature 227:680–685
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) ClustalW and ClustalX version 2. Bioinformatics 23(21):2947–2948
Larsen SH, Størmer FC (1973) Diacetyl (acetoin) reductase from Aerobacter aerogenes—kinetic mechanism and regulation by acetate of reversible reduction of acetoin to 2,3-butanediol. Eur J Biochem 34:100–106
Lee JK, Koo BS, Kim SY, Hyun HH (2003) Purification and characterization of a novel mannitol dehydrogenase from a newly isolated strain of Candida magnoliae. Appl Environ Microbiol 69:4438–4447
Levin I, Meiri G, Peretz M, Burstein Y, Frolow F (2004) The ternary complex of Pseudomonas aeruginosa alcohol dehydrogenase with NADH and ethylene glycol. Protein Sci 13(6):1547–1556
Moore JC, Pollard DJ, Kosjek B, Devine PN (2007) Advances in the enzymatic reduction of ketones. Acc Chem Res 40:1412–1419
Mori K, Otsuka T (1985) Synthesis of (2S,3S)-2,3-octanediol and (S)-2-hydroxy-3-octanone, the male sex pheromone of the grape borer Xylotrechus pyrrhoderus. Tetrahedron 41:553–556
Mott JE, Grant RA, Ho YS, Platt T (1985) Maximizing gene expression from plasmid vectors containing the λPL promoter: strategies for overproducing transcription termination factor ρ. Proc Natl Acad Sci U S A 82:88–92
Neale AD, Scopes RK, Kelly JM, Wettenhall RE (1986) The two alcohol dehydrogenases of Zymomonas mobilis. Purification by differential dye ligand chromatography, molecular characterisation and physiological roles. Eur J Biochem 154(1):119–124
Palomo C, Oiarbide M, García JM (2012) α-Hydroxy ketones as useful templates in asymmetric reactions. Chem Soc Rev 41:4150–4164
Pauly S (1989) Permeability and diffusion data. In: Bandrup J, Immergut EH (eds) Polymer Handbook, 3rd edn. Wiley, New York, pp 435–449
Prust C, Hoffmeister M, Liesegang H, Wiezer A, Fricke WF, Ehrenreich A, Gottschalk G, Deppenmeier U (2005) Complete genome sequence of the acetic acid bacterium Gluconobacter oxydans. Nat Biotechnol 23:195–200
Rhainds M, Lan CC, King S, Gries R, Mo LZ, Gries G (2001) Pheromone communication and mating behaviour of coffee white stem borer, Xylotrechus quadripes Chevrolat (Coleoptera: Cerambycidae). Appl Entomol Zool 36:299–309
Saebel CE, Neureuther JM, Siemann S (2010) A spectrophotometric method for the determination of zinc, copper, and cobalt ions in metalloproteins using Zincon. Anal Biochem 397:218–226
Sakoda H, Imanaka T (1992) Cloning and sequencing of the gene coding for alcohol dehydrogenase of Bacillus stearothermophilus and rational shift of the optimum pH. J Bacteriol 174(4):1397–1402
Salusjärvi T, Povelainen M, Hvorslev N, Eneyskaya EV, Kulminskaya AA, Shabalin KA, Neustroev KN, Kalkkinen N, Miasnikov AN (2004) Cloning of a gluconate/polyol dehydrogenase gene from Gluconobacter suboxydans IFO 12528, characterisation of the enzyme and its use for the production of 5-ketogluconate in a recombinant Escherichia coli strain. Appl Microbiol Biotechnol 65:306–314
Schedel M (2000) Regioselective oxidation of aminosorbitol with Gluconobacter oxydans, key reaction in the industrial 1-deoxynojirimycin synthesis. In: Kelly DR (ed) Biotechnology, vol 8b. Wiley-VCH, Weinheim, pp 295–308
Schweiger P, Gross H, Wesener S, Deppenmeier U (2008) Vinyl ketone reduction by three distinct Gluconobacter oxydans 621H enzymes. Appl Microbiol Biotechnol 80:955–1006
Schweiger P, Gross H, Deppenmeier U (2010) Characterization of two aldo-keto reductases from Gluconobacter oxydans 621H capable of regio- and stereoselective α-ketocarbonyl reduction. Appl Microbiol Biotechnol 87(4):1415–1426
Silber P, Chung H, Gargiulo P, Schulz H (1974) Purification and properties of a diacetyl reductase from Escherichia coli. J Bacteriol 118(3):919–927
Steiner P, Sauer U (2001) Proteins induced during adaptation of Acetobacter aceti to high acetate concentrations. Appl Environ Microbiol 67:5474–5481
Uchida K, Stadtman ER (1993) Covalent attachment of 4-hydroxynonenal to glyceraldehyde-3-phosphate dehydrogenase. A possible involvement of intra- and intermolecular cross-linking reaction. J Biol Chem 268:6388–6393
Vasic-Racki D (2000) Industrial biotransformation. In: Liese A, Seelbach K, Wandrey C (eds). Wiley-VCH, Weinheim Germany
Vidal I, Gonzalez J, Bernardo A, Martin R (1988) Purification and characterization of diacetyl-reducing enzymes from Staphylococcus aureus. Biochem J 251:461–466
Willis LB, Walker GC (1998) Identification of the Rhizobium meliloti alcohol dehydrogenase gene (adhA) and heterologous expression in Alcaligenes eutrophus. Biochim Biophys Acta 1384(2):197–203
Yang IY, Hossain M, Miller H, Khullar S, Johnson F, Grollman A, Moriya M (2001) Responses to the major acrolein-derived deoxyguanosine adduct in Escherichia coli. J Biol Chem 276:9071–9076
Acknowledgments
This project was supported by funds from Bundesministerium für Bildung und Forschung (BMBF, project no. 0315632A).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 770 kb)
Rights and permissions
About this article
Cite this article
Schweiger, P., Gross, H., Zeiser, J. et al. Asymmetric reduction of diketones by two Gluconobacter oxydans oxidoreductases. Appl Microbiol Biotechnol 97, 3475–3484 (2013). https://doi.org/10.1007/s00253-012-4395-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4395-3