Abstract
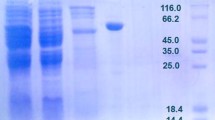
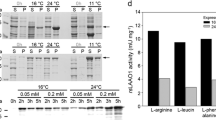
The first hyperthermophilic d-arabitol dehydrogenase from Thermotoga maritima was heterologously purified from Escherichia coli. The protein was purified with and without a Strep-tag. The enzyme exclusively catalyzed the NAD(H)-dependent oxidoreduction of d-arabitol, d-xylitol, d-ribulose, or d-xylulose. A twofold increase of catalytic rates was observed upon addition of Mg2+ or K+. Interestingly, only the tag-less protein was thermostable, retaining 90% of its activity after 90 min at 85 °C. However, the tag-less form of d-arabitol dehydrogenase had similar kinetic parameters compared to the tagged enzyme, demonstrating that the Strep-tag was not deleterious to protein function but decreased protein stability. A single band at 27.6 kDa was observed on SDS-PAGE and native PAGE revealed that the protein formed a homohexamer and a homododecamer. The enzyme catalyzed oxidation of d-arabitol to d-ribulose and therefore belongs to the class of d-arabitol 2-dehydrogenases, which are typically observed in yeast and not bacteria. The product d-ribulose is a rare ketopentose sugar that has numerous industrially applications. Given its thermostability and specificity, d-arabitol 2-dehydrogenase is a desirable biocatalyst for the production of rare sugar precursors.




Similar content being viewed by others
References
Adachi O, Fujii Y, Ghaly MF, Toyama H, Shinagawa E, Matsushita K (2001) Membrane-bound quinoprotein d-arabitol dehydrogenase of Gluconobacter suboxydans IFO 3257: a versatile enzyme for the oxidative fermentation of various ketoses. Biosci Biotechnol Biochem 65:2755–2762
Ahmed Z (2001) Production of natural and rare pentoses using microorganisms and their enzymes. Electron J Biotechnol 4:103–111
Ausubel F (2002) Preparation and analysis of genomic DNA from bacteria. In: Ausubel FM, Kingston RE, Moore DD, Seidman JG, Struhl K (eds) Current protocols in molecular biology, vol 5. Wiley, New York, pp 2–11
Beach JW, Jeong LS, Alves AJ, Pohl D, Kim HO, Chang CN, Doong SL, Schinazi RF, Cheng YC, Chu CK (1992) Synthesis of enantiomerically pure (2′R,5′S)-(-)-1-(2-(hydroxymethyl)oxathiolan-5-yl)cytosine as a potent antiviral agent against hepatitis B virus (HBV) and human immunodeficiency virus (HIV). J Org Chem 57(8):2217–2219
Bertoldo C, Antranikian G (2002) Starch-hydrolyzing enzymes from thermophilic archaea and bacteria. Curr Opin Chem Biol 6(2):151–160
Birken S, Pisano MA (1976) Purification and properties of a polyol dehydrogenase from Cephalosporium chrysogenus. J Bacteriol 125(1):225–232
Blum H, Beier H, Gross HJ (1987) Improved silver staining of plant-proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 8(2):93–99
Bradford MM (1976) Rapid and sensitive method for quantitation of microgram quantities of protein utilizing principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Bronnenmeier K, Kern A, Liebl W, Staudenbauer WL (1995) Purification of Thermotoga maritima enzymes for the degradation of cellulosic materials. Appl Environ Microb 61(4):1399–1407
Bruins ME, Janssen AEM, Boom RM (2001) Thermozymes and their applications—a review of recent literature and patents. Appl Biochem Biotech 90(2):155–186
Charnetzky WT, Mortlock RP (1974) d-arabitol catabolic pathway in Klebsiella aerogenes. J Bacteriol 119(1):170–175
Cheng HR, Jiang N, Shen A, Feng YJ (2005) Molecular cloning and functional expression of D-arabitol dehydrogenase gene from Gluconobacter oxydans in Escherichia coli. FEMS Microbiol Lett 252:35–42
Chiang C, Knight SG (1961) l-Arabinose metabolism by cell-free extracts of Penicillium chrysogenum. Biochim Biophys Acta 46(2):271–278
Conners SB, Mongodin EF, Johnson MR, Montero CI, Nelson KE, Kelly RM (2006) Microbial biochemistry, physiology, and biotechnology of hyperthermophilic Thermotoga species. FEMS Microbiol Rev 30(6):872–905
de Vries RP, Flipphi MJA, Witteveen CFB, Visser J (1994) Characterization of an Aspergillus nidulans L-arabitol dehydrogenase mutant. FEMS Microbiol Lett 123(1–2):83–90
Drueckhammer DG, Hennen WJ, Pederson RL, Barbas CF, Gautheron CM, Krach T, Wong CH (1991) Enzyme catalysis in synthetic carbohydrate-chemistry. Synth Stuttgart 7:499–525
Fernandes S, Tuohy MG, Murray PG (2010) Cloning, heterologous expression, and characterization of the xylitol and L-arabitol dehydrogenase genes, Texdh and Telad, from the thermophilic fungus Talaromyces emersonii. Biochem Genet 48(5–6):480–495
Givry S, Bliard C, Duchiron F (2007) Selective ketopentose analysis in concentrate carbohydrate syrups by HPLC. Carbohyd Res 342(6):859–864
Granström TB, Izumori K, Leisola M (2007) A rare sugar xylitol. Part II: biotechnological production and future applications of xylitol. Appl Microbiol Biot 74(2):273–276
Groer GJ, Haslbeck M, Gessner A (2009) Expression and purification of soluble E-Syt2: low protein stability impedes tag removal. J Chromatogr B Analyt Technol Biomed Life Sci 877(16–17):1643–1650
Hallborn J, Walfridsson M, Penttila M, Keranen S, Hahnhagerdal B (1995) A short-chain dehydrogenase gene from Pichia stipitis having D-arabinitol dehydrogenase activity. Yeast 11(9):839–847
Hernandez-Rocamora VM, Maestro B, Molla-Morales A, Sanz JM (2008) Rational stabilization of the C-LytA affinity tag by protein engineering. Protein Eng Des Sel 21(12):709–720
Horwath RO (1984). Process for oxidizing L-sorbitol to l-fructose. US Patent 4467033
Huber R, Langworthy TA, Konig H, Thomm M, Woese CR, Sleytr UB, Stetter KO (1986) Thermotoga maritima sp. nov. represents a new genus of unique extremely thermophilic eubacteria growing up to 90°C. Arch Microbiol 144(4):324–333
Jiang Y, Zhou Q, Wu K, Li XQ, Shao WL (2006) A highly efficient method for liquid and solid cultivation of the anaerobic hyperthermophilic eubacterium Thermotoga maritima. FEMS Microbiol Lett 259(2):254–259
Kawarasaki Y, Yamada Y, Ichimori M, Shinbata T, Kohda K, Nakano H, Yamane T (2003) Stabilization of affinity-tagged recombinant protein during/after its production in a cell-free system using wheat-germ extract. J Biosci Bioeng 95(3):209–214
Klevstrand R, Nordal A (1950) A spraying reagent for paper chromatograms which is apparently specific for ketoheptoses. Acta Chem Scand 4(8):1320–1320
Knapp R (2010) Sugar: world production supply and distribution. Foreign Agricultural Service Washington DC, United States Department of Agriculture
Laemmli UK (1970) Cleavage of structural proteins during assembly of head of bacteriophage-T4. Nature 227(5259):680–685
Link T, Lohaus G, Heiser I, Mendgen K, Hahn M, Voegele RT (2005) Characterization of a novel NADP(+)-dependent D-arabitol dehydrogenase from the plant pathogen Uromyces fabae. Biochem J 389:289–295
Ma T, Pai SB, Zhu YL, Lin JS, Cheng YC, Chu CK (1996) Structure-activity relationships of 2′-fluoro-L-pyrimidine nucleosides as anti-HBV agents. J Med Chem 39(14):2835–2843
Morita M, Sawa E, Yamaji K, Sakai T, Natori T, Koezuka Y, Fukushima H, Akimoto K (1996) Practical total synthesis of (2S,3S,4R)-1-O-(alpha-D-galactopyranosyl)-N-hexacosanoyl-2-amino-1, 3, 4-octadecanetriol, the antitumorial and immunostimulatory alpha-galactosylceramide, KRN7000. Biosci Biotech Bioch 60(2):288–292
Mott JE, Grant RA, Ho YS, Platt T (1985) Maximizing gene-expression from plasmid vectors containing the lambda PL promoter: strategies for overproducing transcription termination factor rho. Proc Natl Acad Sci USA 82(1):88–92
Nelson KE, Clayton RA, Gill SR, Gwinn ML, Dodson RJ, Haft DH, Hickey EK, Peterson LD, Nelson WC, Ketchum KA, McDonald L, Utterback TR, Malek JA, Linher KD, Garrett MM, Stewart AM, Cotton MD, Pratt MS, Phillips CA, Richardson D, Heidelberg J, Sutton GG, Fleischmann RD, Eisen JA, White O, Salzberg SL, Smith HO, Venter JC, Fraser CM (1999) Evidence for lateral gene transfer between archaea and bacteria from genome sequence of Thermotoga maritima. Nature 399(6734):323–329
Neuberger MS, Patterson RA, Hartley BS (1979) Purification and properties of Klebsiella aerogenes D-arabitol dehydrogenase. Biochem J 183(1):31–42
Niehaus F, Bertoldo C, Kahler M, Antranikian G (1999) Extremophiles as a source of novel enzymes for industrial application. Appl Microbiol Biot 51(6):711–729
Nüss D, Goettig P, Magler I, Denk U, Breitenbach M, Schneider PB, Brandstetter H, Simon-Nobbe B (2010) Crystal structure of the NADP-dependent mannitol dehydrogenase from Cladosporium herbarum: implications for oligomerisation and catalysis. Biochimie 92(8):985–993
Oppermann U, Filling C, Hult M, Shafqat N, Wu XQ, Lindh M, Shafqat J, Nordling E, Kallberg Y, Persson B, Jornvall H (2003) Short-chain dehydrogenases/reductases (SDR): the 2002 update. Chem Biol Interact 143:247–253
Oshitari T, Shibasaki M, Yoshizawa T, Tomita M, Takao K, Kobayashi S (1997) Synthesis of 2-O-(3-O-carbamoyl-alpha-D-mannopyranosyl)-l-gulopyranose: sugar moiety of antitumor antibiotic bleomycin. Tetrahedron 53(32):10993–11006
Prakash O, Jaiswal N (2010) alpha-Amylase: an ideal representative of thermostable enzymes. Appl Biochem Biotech 160(8):2401–2414
Quong MW, Miyada CG, Switchenko AC, Goodman TC (1993) Identification, purification, and characterization of a D-arabinitol-specific dehydrogenase from Candida tropicalis. Biochem Bioph Res Co 196(3):1323–1329
Salusjärvi T, Povelainen M, Hvorslev N, Eneyskaya EV, Kulminskaya AA, Shabalin KA, Neustroev KN, Kalkkinen N, Miasnikov AN (2004) Cloning of a gluconate/polyol dehydrogenase gene from Gluconobacter suboxydans IFO 12528, characterisation of the enzyme and its use for the production of 5-ketogluconate in a recombinant Escherichia coli strain. Appl Microbiol Biot 65(3):306–314
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Schweiger P, Volland S, Deppenmeier U (2007) Overproduction and characterization of two distinct aldehyde-oxidizing enzymes from Gluconobacter oxydans 621H. J Mol Microb Biotech 13(1–3):147–155
Schweiger P, Gross H, Deppenmeier U (2010) Characterization of two aldo-keto reductases from Gluconobacter oxydans 621H capable of regio- and stereoselective alpha-ketocarbonyl reduction. Appl Microbiol Biotechnol 87(4):1415–1426
Scott A (2002) Danisco aims to ramp up sales of “pharmaceutical” sugars. Chem Week 164(46):43
Song SH, Ahluwalia N, Leduc Y, Delbaere LTJ, Vieille C (2008) Thermotoga maritima TM0298 is a highly thermostable mannitol dehydrogenase. Appl Microbiol Biot 81(3):485–495
Stein R, Gross W, Schnarrenberger C (1997) Characterization of a xylitol dehydrogenase and a D-arabitol dehydrogenase from the thermo- and acidophilic red alga Galdieria sulphuraria. Planta 202(4):487–493
Takagi Y, Nakai K, Tsuchiya T, Takeuchi T (1996) A 5′-(trifluoromethyl)anthracycline glycoside: synthesis of antitumor-active 7-0-(2,6-dideoxy-6,6,6-trifluoro-alpha-L-lyxo-hexopyranosyl) adriamycinone. J Med Chem 39(8):1582–1588
Weimberg R (1962) Mode of formation of D-arabitol by Saccharomyces mellis. Biochem Bioph Res Co 8(6):442–445
Winterhalter C, Liebl W (1995) Two extremely thermostable xylanases of the hyperthermophilic bacterium Thermotoga maritima MSB8. Appl Environ Microb 61(5):1810–1815
Wohlgemuth R (2010) Asymmetric biocatalysis with microbial enzymes and cells. Curr Opin Microbiol 13(3):283–292
Wong B, Murray JS, Castellanos M, Croen KD (1993) D-arabitol metabolism in Candida albicans: studies of the biosynthetic pathway and the gene that encodes NAD-dependent D-arabitol dehydrogenase. J Bacteriol 175(19):6314–6320
Acknowledgments
This project was supported by funds from Bundesministerium für Bildung und Forschung (BMBF, project no. 0315632A).
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at https://doi.org/10.1007/s00253-011-3250-2
Rights and permissions
About this article
Cite this article
Kallnik, V., Schultz, C., Schweiger, P. et al. Properties of recombinant Strep-tagged and untagged hyperthermophilic D-arabitol dehydrogenase from Thermotoga maritima . Appl Microbiol Biotechnol 90, 1285–1293 (2011). https://doi.org/10.1007/s00253-011-3187-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3187-5