Abstract
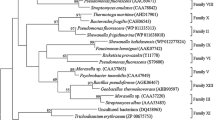
A clone which conferred lipolytic activity at low temperature was identified from a fosmid library constructed from a South China Sea marine sediment sample. The gene responsible, estF, consisted of 1,080 bp that encoded 359 amino acid residues, with a typical N-terminal signal peptide of 28 amino acid residues. A phylogenetic analysis of amino acid sequence with other lipolytic enzymes revealed that EstF and seven closely related putative lipolytic enzymes comprised a unique clade in the phylogenetic tree. Moreover, these hypothetic esterases showed unique conservative sites in the amino acid sequence. The recombinant EstF was overexpressed and purified, and its biochemical properties were partially characterized. The optimal substrate for EstF to hydrolyze among a panel of p-nitrophenyl esters (C2 to C16) was p-nitrophenyl butyrate (C4), with a K m of 0.46 mM. Activity quickly decreased with substrates containing an acyl chain length longer than 10 carbons. We found that EstF was active in the temperature range of 0–60°C, showed the best activity at 50°C, but was unstable at 60°C. It exhibited a high level of activity in the pH range of 7.0–10.0 showing the highest activity at pH 9.0.




Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Arpigny JL, Jaeger KE (1999) Bacterial lipolytic enzymes: classification and properties. Biochem J 343(Pt 1):177–183
Berge JP, Barnathan G (2005) Fatty acids from lipids of marine organisms: molecular biodiversity, roles as biomarkers, biologically active compounds, and economical aspects. Adv Biochem Eng Biotechnol 96:49–125
Blunt JW, Copp BR, Munro MHG, Northcote PT, Prinsep MR (2010) Marine natural products. Nat Prod Rep 27:165–237
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chen IC, Lin WD, Hsu SK, Thiruvengadam V, Hsu WH (2009) Isolation and characterization of a novel lysine racemase from a soil metagenomic library. Appl Environ Microbiol 75:5161–5166
Chu X, He H, Guo C, Sun B (2008) Identification of two novel esterases from a marine metagenomic library derived from South China Sea. Appl Microbiol Biotechnol 80:615–625
Chung GH, Lee YP, Jeohn GH, Yoo OJ, Rhee JS (1991) Cloning and nucleotide sequence of thermostable lipase gene from Pseudomonas fluorescens SIK W1. Agric Biol Chem 55:2359–2365
Chung EJ, Lim HK, Kim JC, Choi GJ, Park EJ, Lee MH, Chung YR, Lee SW (2008) Forest soil metagenome gene cluster involved in antifungal activity expression in Escherichia coli. Appl Environ Microbiol 74:723–730
Das AK, Bellizzi JJ 3rd, Tandel S, Biehl E, Clardy J, Hofmann SL (2000) Structural basis for the insensitivity of a serine enzyme (palmitoyl-protein thioesterase) to phenylmethylsulfonyl fluoride. J Biol Chem 275:23847–23851
Debashish G, Malay S, Barindra S, Joydeep M (2005) Marine enzymes. Adv Biochem Eng Biotechnol 96:189–218
Harvey HR (1989) Cycling of lipids in the marine-environment—role of zooplankton grazing. Abstr Pap Am Chem Soc 197:40-Geoc
Henikoff S, Henikoff JG (1992) Amino acid substitution matrices from protein blocks. Proc Natl Acad Sci USA 89:10915–10919
Hu Y, Zhang G, Li A, Chen J, Ma L (2008) Cloning and enzymatic characterization of a xylanase gene from a soil-derived metagenomic library with an efficient approach. Appl Microbiol Biotechnol 80:823–830
Hu Y, Fu C, Huang Y, Yin Y, Cheng G, Lei F, Lu N, Li J, Ashforth EJ, Zhang L, Zhu B (2010a) Novel lipolytic genes from the microbial metagenomic library of the South China Sea marine sediment. FEMS Microbiol Ecol 72:228–237
Hu Y, Fu C, Yin Y, Cheng G, Lei F, Yang X, Li J, Ashforth EJ, Zhang L, Zhu B (2010b) Construction and preliminary analysis of a deep-sea sediment Metagenomic Fosmid library from Qiongdongnan Basin, South China Sea. Mar Biotechnol (NY) 12(6):719–727
Jensen RG, Hamosh M (1996) Selectivity of lipases - types and determination. Nato Adv Sci Inst Ser 317:17–29
Kim EY, Oh KH, Lee MH, Kang CH, Oh TK, Yoon JH (2009) Novel cold-adapted alkaline lipase from an intertidal flat metagenome and proposal for a new family of bacterial lipases. Appl Environ Microbiol 75:257–260
Kiriakoulakis K, Stutt E, Rowland SJ, Vangriesheim A, Lampitt RS, Wolff GA (2001) Controls on the organic chemical composition of settling particles in the Northeast Atlantic Ocean. Prog Oceanogr 50:65–87
Klibanov AM (2001) Improving enzymes by using them in organic solvents. Nature 409:241–246
Kumar S, Nei M, Dudley J, Tamura K (2008) MEGA: a biologist-centric software for evolutionary analysis of DNA and protein sequences. Brief Bioinform 9:299–306
Lang DA, Dijkstra BW (1998) Structural investigations of the regio- and enantioselectivity of lipases. Chem Phys Lipids 93:115–122
LeCleir GR, Buchan A, Maurer J, Moran MA, Hollibaugh JT (2007) Comparison of chitinolytic enzymes from an alkaline, hypersaline lake and an estuary. Environ Microbiol 9:197–205
Lee SW, Won K, Lim HK, Kim JC, Choi GJ, Cho KY (2004) Screening for novel lipolytic enzymes from uncultured soil microorganisms. Appl Microbiol Biotechnol 65:720–726
Lee MH, Lee CH, Oh TK, Song JK, Yoon JH (2006) Isolation and characterization of a novel lipase from a metagenomic library of tidal flat sediments: evidence for a new family of bacterial lipases. Appl Environ Microbiol 72:7406–7409
Lorenz P, Liebeton K, Niehaus F, Eck J (2002) Screening for novel enzymes for biocatalytic processes: accessing the metagenome as a resource of novel functional sequence space. Curr Opin Biotechnol 13:572–577
Nielsen H, Engelbrecht J, Brunak S, von Heijne G (1997) A neural network method for identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Int J Neural Syst 8:581–599
Rhee JK, Ahn DG, Kim YG, Oh JW (2005) New thermophilic and thermostable esterase with sequence similarity to the hormone-sensitive lipase family, cloned from a metagenomic library. Appl Environ Microbiol 71:817–825
Russell NJ, Nichols DS (1999) Polyunsaturated fatty acids in marine bacteria—a dogma rewritten. Microbiol-Sgm 145:767–779
Schloss PD, Handelsman J (2003) Biotechnological prospects from metagenomics. Curr Opin Biotechnol 14:303–310
Sharma S, Khan FG, Qazi GN (2010) Molecular cloning and characterization of amylase from soil metagenomic library derived from Northwestern Himalayas. Appl Microbiol Biotechnol 86:1821–1828
Singh A, Singh Chauhan N, Thulasiram HV, Taneja V, Sharma R (2010) Identification of two flavin monooxygenases from an effluent treatment plant sludge metagenomic library. Bioresour Technol 101:8481–8484
Steele HL, Jaeger KE, Daniel R, Streit WR (2009) Advances in recovery of novel biocatalysts from metagenomes. J Mol Microbiol Biotechnol 16:25–37
Streit WR, Schmitz RA (2004) Metagenomics—the key to the uncultured microbes. Curr Opin Microbiol 7:492–498
Streit WR, Daniel R, Jaeger KE (2004) Prospecting for biocatalysts and drugs in the genomes of non-cultured microorganisms. Curr Opin Biotechnol 15:285–290
Verger R (1997) ‘Interfacial activation’ of lipases: facts and artifacts. Trends Biotechnol 15:32–38
Voget S, Leggewie C, Uesbeck A, Raasch C, Jaeger KE, Streit WR (2003) Prospecting for novel biocatalysts in a soil metagenome. Appl Environ Microbiol 69:6235–6242
Waschkowitz T, Rockstroh S, Daniel R (2009) Isolation and characterization of metalloproteases with a novel domain structure by construction and screening of metagenomic libraries. Appl Environ Microbiol 75:2506–2516
Ye M, Li G, Liang WQ, Liu YH (2010) Molecular cloning and characterization of a novel metagenome-derived multicopper oxidase with alkaline laccase activity and highly soluble expression. Appl Microbiol Biotechnol 87:1023–1031
Acknowledgments
We thank Dr. Simon Shih for helpful discussions. This work was supported in part by grants from the National 863 Project (2007AA09Z443), the Key Project for International Cooperation (2007DFB31620), the National Natural Science Foundation of China (30973665, 30700015, 30873129, 30901849, 30911120484, 81011120046, and 30911120483), the National Key Technology R&D Program (2007BAI26B02), the CAS Pillar Program (KSCX2-YW-R-164), and from the Important National Science & Technology Specific Projects (2008ZX09401-05, 2009ZX09302-004). L. Z. received funding from the Hundred Talents Program.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Chengzhang Fu and Yongfei Hu contributed equally to this article.
Rights and permissions
About this article
Cite this article
Fu, C., Hu, Y., Xie, F. et al. Molecular cloning and characterization of a new cold-active esterase from a deep-sea metagenomic library. Appl Microbiol Biotechnol 90, 961–970 (2011). https://doi.org/10.1007/s00253-010-3079-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-3079-0