Abstract
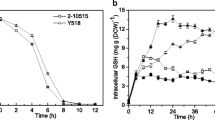
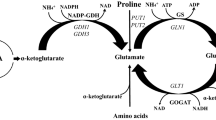
Molecular mechanisms leading to glutathione (GSH) over-accumulation in a Saccharomyces cerevisiae strain produced by UV irradiation-induced random mutagenesis were studied. The mutant accumulated GSH but also cysteine and γ-glutamylcysteine in concentrations that were several fold higher than in its wild-type parent strain under all studied cultivation conditions (chemostat, fed-batch, and turbidostat). Transcript analyses along with shotgun proteome quantification indicated a difference in the expression of a number of genes and proteins, the most pronounced of which were several fold higher expression of CYS3, but also that of GSH1 and its transcriptional activator YAP1. This together with the higher intracellular cysteine concentration is most likely the primary factor underlying GSH over-accumulation in the mutant. Comparative sequencing of GSH1 and the fed-batch experiments with continuous cysteine addition demonstrated that the feedback inhibition of Gsh1p by GSH was still operational in the mutant.





Similar content being viewed by others
References
Alfafara CG, Kanda A, Shioi T, Shimizu H, Shioya S, Suga K (1992a) Effect of amino acids on glutathione production by Saccharomyces cerevisiae. Appl Microbiol Biotechnol 36:538–540
Alfafara CG, Miura K, Shimizu H, Shioya S, Suga K (1992b) Cysteine addition strategy for maximum glutathione production in fed-batch culture of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 37:141–146
Bachhawat AK, Ganguli D, Kaur J, Kasturia N, Thakur A, Kaur H, Kumar A, Yaday A (2009) Glutathione production in yeast. In: Satyanarayana T, Kunze G (eds) Yeast biotechnology: diversity and applications. Springer, Dordrecht, pp 259–280
Bagnyukova TV, Vasylkiv OY, Storey KB, Lushchak VI (2005) Catalase inhibition by amino triazole induces oxidative stress in goldfish brain. Brain Res 1052:180–186
Biterova EI, Barycki JJ (2010) Structural basis for feedback and pharmacological inhibition of Saccharomyces cerevisiae glutamate cysteine ligase. J Biol Chem 285:14459–14466
Delaunay A, Isnard AD, Toledano MB (2000) H2O2 sensing through oxidation of the Yap1 transcription factor. EMBO J 19:5157–5166
Galant NJ, Wang H, Lee DR, Mucsi Z, Setiadi DH, Viskolcz B, Csizmadia IG (2009) Thermodynamic role of glutathione oxidation by peroxide and peroxybicarbonate in the prevention of Alzheimer’s disease and cancer. J Phys Chem A 113:9138–9149
Grant CM, MacIver FH, Dawes IW (1997) Glutathione synthetase is dispensable for growth under both normal and oxidative stress conditions in the yeast Saccharomyces cerevisiae due to an accumulation of the dipeptide γ-glutamylcysteine. Mol Biol Cell 8:1699–1707
Gulshan K, Rovinsky SA, Coleman ST, Moye-Rowley WS (2005) Oxidant-specific folding of Yap1p regulates both transcriptional activation and nuclear localization. J Biol Chem 280:40524–40533
Hamada S, Tanaka H, Sakato K (1986) Process for producing glutathione. US Patent 4(582):801
Hiraishi H, Miyake T, Ono B (2008) Transcriptional regulation of Saccharomyces cerevisiae CYS3 encoding cystathionine γ-lyase. Curr Genet 53:225–234
Ikeno Y, Tanno K, Omori I, Yamada R (1977) Glutathione. JP patent 52(087):296
Kaiser P, Flick K, Wittenberg C, Reed SI (2000) Regulation of transcription by ubiquitination without proteolysis: Cdc34/SCFMet30-mediated inactivation of the transcription factor Met4. Cell 102:303–314
Kasemets K, Nisamedtinov I, Laht T, Abner K, Paalme T (2007) Growth characteristics of Saccharomyces cerevisiae S288C in changing environmental conditions: auxo-accelerostat study. Antonie Leeuwenhoek 92:109–128
Köcher T, Pichler P, Schutzbier M, Stingl C, Kaul A, Teucher N, Hasenfuss G, Penninger JM, Mechtler K (2009) High precision quantitative proteomics using iTRAQ on an LTQ Orbitrap: a new mass spectrometric method combining the benefits of all. J Proteome Res 8:4743–4752
Kono G, Harada M, Sugisaki K, Nishida M (1977) High glutathione-containing yeast. JP Patent 52(125):687
Kuge S, Jones N, Nomoto A (1997) Regulation of yAP-1 nuclear localization in response to oxidative stress. EMBO J 16:1710–1720
Lai J, Lee S, Hsieh C, Hwang C, Liao C (2008) Saccharomyces cerevisiae strains for hyper-producing glutathione and γ-glutamylcysteine and process of use. US Patent 7(371):557
Lee J, Godon C, Lagniel G, Spector D, Garin J, Labarre J, Toledano MB (1999) Yap1 and Skn7 control two specialized oxidative stress response regulons in yeast. J Biol Chem 274:16040–16046
Li Y, Wei G, Chen J (2004) Glutathione: a review on biotechnological production. Appl Microbiol Biotechnol 66:233–242
Liang G, Du G, Chen J (2008) Enhanced glutathione production by using low-pH stress coupled with cysteine addition in the treatment of high cell density culture of Candida utilis. Lett Appl Microbiol 46:507–512
Martin HL, Teismann P (2009) Glutathione–a review on its role and significance in Parkinson’s disease. FASEB J 23:3263–3272
Menant A, Baudouin-Cornu P, Peyraud C, Tyers M, Thomas D (2006) Determinants of the ubiquitin-mediated degradation of the Met4 transcription factor. J Biol Chem 281:11744–11754
Nisamedtinov I, Kevvai K, Orumets K, Rautio JJ, Paalme T (2010) Glutathione accumulation in ethanol-stat fed-batch culture of Saccharomyces cerevisiae with a switch to cysteine feeding. Appl Microbiol Biotechnol 87:175–183
Pócsi I, Prade R, Penninckx M (2004) Glutathione, altruistic metabolite in fungi. Adv Microb Physiol 49:1–76
Rappsilber J, Mann M, Ishihama Y (2007) Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat Protoc 2:1896–1906
Rouillon A, Barbey R, Patton EE, Tyers M, Thomas D (2000) Feedback-regulated degradation of the transcriptional activator Met4 is triggered by the SCFMet30 complex. EMBO J 19:282–294
Soltaninassab SR, Sekhar KR, Meredith MJ, Freeman ML (2000) Multi-faceted regulation of γ-glutamylcysteine synthetase. J Cell Physiol 182:163–170
Sugiyama K, Izawa S, Inoue Y (2000) The Yap1p-dependent induction of glutathione synthesis in heat shock response of Saccharomyces cerevisiae. J Biol Chem 275:15535–15540
Thomas D, Surdin-Kerjan Y (1997) Metabolism of sulfur amino acids in Saccharomyces cerevisiae. Microbiol Mol Biol R 61:503–532
Trotter EW, Grant CM (2003) Non-reciprocal regulation of the redox state of the glutathione-glutaredoxin and thioredoxin systems. EMBO Rep 4:184–188
Wang Z, Tan T, Song J (2007) Effect of amino acids addition and feedback control strategies on the high-cell-density cultivation of Saccharomyces cerevisiae for glutathione production. Process Biochem 42:108–111
Wen S, Zhang T, Tan T (2006) Maximizing production of glutathione by amino acid modulation and high-cell-density fed-batch culture of Saccharomyces cerevisiae. Process Biochem 41:2424–2428
Wheeler GL, Trotter EW, Dawes IW, Grant CM (2003) Coupling of the transcriptional regulation of glutathione biosynthesis to the availability of glutathione and methionine via the Met4 and Yap1 transcription factors. J Biol Chem 278:49920–49928
Xu H, Freitas MA (2009) MassMatrix: a database search program for rapid characterization of proteins and peptides from tandem mass spectrometry data. Proteomics 9:1548–1555
Acknowledgments
The financial support for this research was provided by the Enterprise Estonia project EU22704, Estonian Ministry of Education and Research grant SF0140090s08, and by Estonian Science Foundation grant G7323. We would like to thank Dr. Chris Powell for critical revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nisamedtinov, I., Kevvai, K., Orumets, K. et al. Metabolic changes underlying the higher accumulation of glutathione in Saccharomyces cerevisiae mutants. Appl Microbiol Biotechnol 89, 1029–1037 (2011). https://doi.org/10.1007/s00253-010-2946-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-2946-z