Abstract
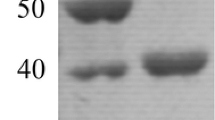
A β-1,4-endoglucanase (Cel5A) was cloned from the genomic DNA of saccharolytic thermophilic eubacterium Thermoanaerobacter tengcongensis MB4 and functionally expressed in Escherichia coli. Substrate specificity analysis revealed that Cel5A cleaves specifically the β-1,4-glycosidic linkage in cellulose with high activity (294 U mg−1; carboxymethyl cellulose sodium (CMC)). On CMC, kinetics of Cel5A was determined (K m 1.39 ± 0.12 g l−1; k cat/K m 1.41 ± 0.13 g−1 s−1). Cel5A displays an activity optimum between 75 and 80 °C. Residues Glu187 and Glu289 were identified as key catalytic amino acids by sequence alignment. Interestingly, derived from a non-halophilic bacterium, Cel5A exhibits high residual activities in molar concentration of NaCl (3 M, 49.3%) and KCl (4 M, 48.6%). In 1 M NaCl, 82% of Cel5A activity is retained after 24 h incubation. Molecular Dynamics studies performed at 0 and 3 M NaCl, correlate the Cel5A stability to the formation of R-COO−···Na+ ···−OOC-R salt bridges within the Cel5A tertiary structure, while activity possibly relates to the number of Na+ ions trapped into the negatively charged active site, involving a competition mechanism between substrate and Na+. Additionally, Cel5A is remarkably resistant in ionic liquids 1-butyl-3-methyllimidazolium chloride (1 M, 54.4%) and 1-allyl-3-methylimidazolium chloride (1 M, 65.1%) which are promising solvents for cellulose degradation and making Cel5A an attractive candidate for industrial applications.







Similar content being viewed by others
References
Abdeev RM, Goldenkova IV, Musiychuk KA, Piruzian ES (2001) Exploring the properties of thermostable Clostridium thermocellum cellulase CelE for the purpose of its expression in plants. Biochemistry (Moscow) 66:808–813
Adsul MG, Terwadkar AP, Varma AJ, Gokhale D (2009) Cellulases from Penicillium janthinellum mutants: solid-state production and their stability in ionic liquids. BioResources 4:1670–1681
Ando S, Ishida H, Kosugi Y, Ishikawa K (2002) Hyperthermostable endoglucanase from Pyrococcus horikoshii. Appl Environ Microbiol 68(1):430–433
Arai T, Araki R, Tanaka A, Karita S, Kimura T, Sakka K, Ohmiya K (2003) Characterization of a cellulase containing a family 30 carbohydrate-binding module (CBM) derived from Clostridium thermocellum CelJ: importance of the CBM to cellulose hydrolysis. J Bacteriol 185:504–512
Arai T, Kosugi A, Chan H, Koukiekolo R, Yukawa H, Inui M, Doi R (2006) Properties of cellulosomal family 9 cellulases from Clostridium cellulovorans. Appl Microbiol Biotechnol 71:654–660
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201
Baker J, McCarley J, Lovett R, Yu C-H, Adney W, Rignall T, Vinzant T, Decker S, Sakon J, Himmel M (2005) Catalytically enhanced endocellulase Cel5A from Acidothermus cellulolyticus. Appl Biochem Biotechnol 121:129–148
Bao Q, Tian Y, Li W, Xu Z, Xuan Z, Hu S, Dong W, Yang J, Chen Y, Xue Y, Xu Y, Lai X, Huang L, Dong X, Ma Y, Ling L, Tan H, Chen R, Wang J, Yu J, Yang H (2002) A complete sequence of the T. tengcongensis genome. Genome Res 12:689–700
Bronnenmeier K, Kern A, Liebl W, Staudenbauer WL (1995) Purification of Thermotoga maritima enzymes for the degradation of cellulosic materials. Appl Environ Microbiol 61:1399–1407
Creuzet N, Frixon C (1983) Purification and characterization of an endoglucanase from a newly isolated thermophilic anaerobic bacterium. Biochimie 65:149–156
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: an N•log(N) method for Ewald sums in large systems. J Chem Phys 98:10089–10092
Davies G, Dauter M, Brzozowski AM, Bjørnvad ME, Andersen KV, Schülein M (1998) Structure of the Bacillus agaradherans family 5 endoglucanase at 1.6 Å and its cellobiose complex at 2.0 Å resolution. Biochemistry 37:1926–1932
Deshpande MV, Eriksson K-E, Göran Pettersson L (1984) An assay for selective determination of exo-1, 4,-β-glucanases in a mixture of cellulolytic enzymes. Anal Biochem 138:481–487
Dyrløv Bendtsen J, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340:783–795
Fauth U, Romaniec MP, Kobayashi T, Demain AL (1991) Purification and characterization of endoglucanase Ss from Clostridium thermocellum. Biochem J 279:67–73
Fukuchi S, Yoshimune K, Wakayama M, Moriguchi M, Nishikawa K (2003) Unique amino acid composition of proteins in halophilic bacteria. J Mol Biol 327:347–357
Gouet P, Robert X, Courcelle E (2003) ESPript/ENDscript: extracting and rendering sequence and 3D information from atomic structures of proteins. Nucleic Acids Res 31:3320–3323
Haki GD, Rakshit SK (2003) Developments in industrially important thermostable enzymes: a review. Bioresour Technol 89:17–34
Hess B, Kutzner C, van der Spoel D, Lindahl E (2008) GROMACS 4: algorithms for highly efficient, load-balanced, and scalable molecular simulation. J Chem Theory Comput 4:435–447
Hirasawa K, Uchimura K, Kashiwa M, Grant W, Ito S, Kobayashi T, Horikoshi K (2006) Salt-activated endoglucanase of a strain of alkaliphilic Bacillus agaradhaerens. Anton Leeuw Int J G 89:211–219
Honda H, Naito H, Taya M, Iijima S, Kobayashi T (1987) Cloning and expression in Escherichia coli of a Thermoanaerobacter cellulolyticus gene coding for heat-stable β-glucanase. Appl Microbiol Biotechnol 25:480–483
Hoover WG (1985) Canonical dynamics: equilibrium phase-space distributions. Phys Rev A 31:1695–1697
Huang XP, Monk C (2004) Purification and characterization of a cellulase (CMCase) from a newly isolated thermophilic aerobic bacterium Caldibacillus cellulovorans gen. nov., sp. nov. World J Microbiol Biotechnol 20:85–92
Kakiuchi M, Isui A, Suzuki K, Fujino T, Fujino E, Kimura T, Karita S, Sakka K, Ohmiya K (1998) Cloning and DNA sequencing of the genes encoding Clostridium josui scaffolding protein CipA and cellulase CelD and identification of their gene products as major components of the cellulosome. J Bacteriol 180:4303–4308
Kim JO, Park SR, Lim WJ, Ryu SK, Kim MK, An CL, Cho SJ, Park YW, Kim JH, Yun HD (2000) Cloning and characterization of thermostable endoglucanase (Cel8Y) from the hyperthermophilic Aquifex aeolicus VF5. Biochem Biophys Res Commun 279:420–426
Larkin M, Blackshields G, Brown N, Chenna R, McGettigan P, McWilliam H, Valentin F, Wallace I, Wilm A, Lopez R, Thompson J, Gibson T, Higgins D (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Letunic I, Doerks T, Bork P (2009) SMART 6: recent updates and new developments. Nucleic Acids Res 37:229–232
Lill MA, Helms V (2001) Molecular dynamics simulation of proton transport with quantum mechanically derived proton hopping rates (Q-HOP MD). J Chem Phys 115:7993–8005
Limauro D, Cannio R, Fiorentino G, Rossi M, Bartolucci S (2001) Identification and molecular characterization of an endoglucanase gene, celS, from the extremely thermophilic archaeon Sulfolobus solfataricus. Extremophiles 5:213–219
Liu L, Chen H (2006) Enzymatic hydrolysis of cellulose materials treated with ionic liquid [BMIM] Cl. Chin Sci Bull 51:2432–2436
Madern D, Ebel C, Zaccai G (2000) Halophilic adaptation of enzymes. Extremophiles 4:91–98
Maki M, Leung KT, Qin W (2009) The prospects of cellulase-producing bacteria for the bioconversion of lignocellulosic biomass. Int J Biol Sci 5:500–516
McCarter JD, Withers SG (1994) Mechanisms of enzymatic glycoside hydrolysis. Curr Opin Struct Biol 4:885–892
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Ng TK, Zeikus JG (1981) Purification and characterization of an endoglucanase (1, 4-beta-D-glucan glucanohydrolase) from Clostridium thermocellum. Biochem J 199:341–350
Ng IS, Li C-W, Yeh Y-F, Chen P, Chir J-L, Ma C-H, Yu S-M, T-h H, Tong C-G (2009) A novel endo-glucanase from the thermophilic bacterium Geobacillus sp. 70PC53 with high activity and stability over a broad range of temperatures. Extremophiles 13:425–435
Nielsen H, Engelbrecht J, Brunak S, von Heijne G (1997) Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng Des Sel 10:1–6
Nosé S (1984) A molecular dynamics method for simulations in the canonical ensemble. Mol Phys 52:255–268
Ogura J, Toyoda A, Kurosawa T, Chong AL, Chohnan S, Masaki T (2006) Purification, characterization, and gene analysis of cellulase (Cel8A) from Lysobacter sp. IB-9374. Biosci Biotechnol Biochem 70:2420–2428
Parrinello M, Rahman A (1980) Crystal structure and pair potentials: a molecular-dynamics study. Phys Rev Lett 45:1196
Pottkämper J, Barthen P, Ilmberger N, Schwaneberg U, Schenk A, Schulte M, Ignatiev N, Streit WR (2009) Applying metagenomics for the identification of bacterial cellulases that are stable in ionic liquids. Green Chem 11:957–965
Schülein M (2000) Protein engineering of cellulases. Biochim Biophys Acta Enzymol 1543:239–252
Schultz J, Milpetz F, Bork P, Ponting CP (1998) SMART, a simple modular architecture research tool: identification of signaling domains. Proc Nat Acad Sci USA 95:5857–5864
Schwarz WH, Bronnenmeier K, Gräbnitz F, Staudenbauer WL (1987) Activity staining of cellulases in polyacrylamide gels containing mixed linkage beta-glucans. Anal Biochem 164:72–77
Shepherd MG, Cole AL, Tong CC, Willis AW, Scott TK (1988) Cellulases from Thermoascus aurantiacus. In: methods in enzymology. Elsevier, San Diego, pp 300–307
Shirai T, Ishida H, J-i N, Yamane T, Ozaki K, Hakamada Y, Ito S (2001) Crystal structure of alkaline cellulase K: insight into the alkaline adaptation of an industrial enzyme. J Mol Biol 310:1079–1087
Singh J, Batra N, Sobti RC (2004) Purification and characterisation of alkaline cellulase produced by a novel isolate, Bacillus sphaericus JS1. J Ind Microbiol Biotechnol 31:51–56
Van der Spoel D, Lindahl E, Hess B, Groenhof G, Mark AE, Berendsen HJC (2005) GROMACS: fast, flexible, and free. J Comput Chem 26:1701–1718
Varrot A, Schulein M, Davies GJ (2000) Insights into ligand-induced conformational change in Cel5A from Bacillus agaradhaerens revealed by a catalytically active crystal form. J Mol Biol 297:819–828
Voget S, Steele HL, Streit WR (2006) Characterization of a metagenome-derived halotolerant cellulase. J Biotechnol 126:26–36
Voth GA (2006) Computer simulation of proton solvation and transport in aqueous and biomolecular systems. Acc Chem Res 39:143–150
Wang Q, Tull D, Meinke A, Gilkes NR, Warren RA, Aebersold R, Withers SG (1993) Glu280 is the nucleophile in the active site of Clostridium thermocellum CelC, a family A endo-beta-1, 4-glucanase. J Biol Chem 268:14096–14102
Wu J, Zhang J, Zhang H, He J, Ren Q, Guo M (2004) Homogeneous acetylation of cellulose in a new ionic liquid. Biomacromolecules 5:266–268
Xue Y, Xu Y, Liu Y, Ma Y, Zhou P (2001) Thermoanaerobacter tengcongensis sp. nov., a novel anaerobic, saccharolytic, thermophilic bacterium isolated from a hot spring in Tengcong, China. Int J Syst Evol Microbiol 51:1335–1341
Acknowledgement
This research was supported by the grant of Natural Science Foundation of China (No.30621005) and Ministry of Sciences and Technology of China (973 Program: 2007CB707801 and 2009CB724705, 863 Programs: 2006AA020201 and 2007AA021306). The authors would like to express our sincere gratitude to Deutscher Akademischer Austausch Dienst (DAAD) for its valuable support, the Cluster of Excellence Tailor Made Fuels from Biomass (TMFB, RWTH Aachen University), BioNoCo (Biocatalysis using non-conventional media, Deutsche Forschungsgemeinschaft) and the computational resources offered by the RZ (Rechen Zentrum, Super Computer Center RWTH Aachen). We would like to thank Mr. Wolfgang Glory and Mr. Caesar Garfield for their assistance in experiments.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 275 kb)
Rights and permissions
About this article
Cite this article
Liang, C., Xue, Y., Fioroni, M. et al. Cloning and characterization of a thermostable and halo-tolerant endoglucanase from Thermoanaerobacter tengcongensis MB4. Appl Microbiol Biotechnol 89, 315–326 (2011). https://doi.org/10.1007/s00253-010-2842-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-2842-6