Abstract
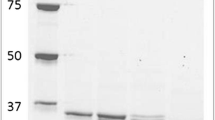
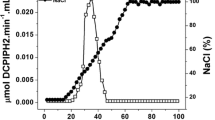
Two cytosolic nicotinamide adenine dinucleotide phosphate-dependent aldehyde reductases, Gox1899 and Gox2253, from Gluconobacter oxydans 621H were overproduced and purified from Escherichia coli. The purified proteins exhibited subunit masses of 26.4 (Gox1899) and 36.7 kDa (Gox2253). Both proteins formed homo-octamers exhibiting native masses of 210 and 280 kDa, respectively. The substrate spectra, optimal reaction conditions, and kinetic constants were determined for Gox1899 and Gox2253. Both enzymes efficiently catalyzed the reduction of medium/long-chain aldehydes. However, Gox1899 had a wider substrate spectrum and was more catalytically efficient. The best activity with Gox1899 was found for aliphatic aldehydes of C6-C10. In contrast, Gox2253 had a limited substrate spectrum and reduced octanal, nonanal, and decanal. Both enzymes were unable to oxidize primary alcohols. Aldehyde removal may be of particular importance for Gluconobacter because the membrane-bound alcohol dehydrogenase rapidly oxidizes short to long-chain alcohols, and large quantities of aldehydes could enter the cell, making detoxification necessary.


Similar content being viewed by others
References
Adachi O, Tayama K, Shinagawa E, Matsushita K, Ameyama M (1978) Purification and characterization of particulate alcohol dehydrogenase from Gluconobacter suboxydans. Agric Biol Chem 42:2045–2205
Adams CW, Fornwald JA, Schmidt FJ, Rosenberg M, Brawneri ME (1988) Gene organization and structure of the Streptomyces lividans gal operon. J Bacteriol 170:203–212
Ausubel FM (2002) Preparation and analysis of genomic DNA from bacteria. In: Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Struhl K (eds) Current protocols in molecular biology, vol 5. Wiley, New York, pp 2–11
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chen J, Schenker S, Frosto TA, Henderson GI (1998) Inhibition of cytochrome c oxidase activity by 4-hydroxynonenal (HNE). Role of HNE adduct formation with the enzyme subunits. Biochim Biophys Acta 1380:336–344
De Ley J, Gillis M, Swings J (1984) The genus Gluconobacter. In: Krieg NR, Holt JG (eds) Bergey’s manual of systematic bacteriology, vol 1. Williams and Wilkins, Baltimore, MD, pp 267–278
DeMaster EG, Shirota FN, Nagasawa HT (1986) Role of propiolaldehyde and other metabolites in the pargyline inhibition of rat-liver aldehyde dehydrogenase. Biochem Pharmacol 35:1481–1489
Esterbauer H, Zollner H, Scholz N (1975) Reaction of glutathione with conjugated carbonyls. Z Naturforsch 30:466–473
Grimshaw CE (1992) Aldose reductase: model for a new paradigm of enzymatic perfection in detoxification catalysts. Biochemistry 31:10139–10145
Jakoby WB, Ziegler DM (1990) The enzymes of detoxication. J Biol Chem 265:20715–20718
Ji C, Mirvish SS, Nickols J, Ishizaki H, Lee MJ, Yang CS (1989) Formation of hydroxy derivatives, aldehydes, and nitrite from n-nitrosomethyl-n-amylamine by rat liver microsomes and by purified cytochrome P-450 IIB1. Cancer Res 49:5299–5304
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Larroy C, Fernández MR, Gonzáles E, Parés X, Biosca JA (2002) Characterization of the Saccharomyces cerevisiae YMR318C (ADH6) gene product as a broad specificity NADPH-dependent alcohol dehydrogenase: relevance in aldehyde reduction. Biochem J 361:163–172
Maness PC, Smolinski S, Blake DM, Huang Z, Wolfrum EJ, Jacoby WA (1999) Bactericidal activity of photocatalytic TiO2 reaction: toward an understanding of its killing mechanism. Appl Environ Microbiol 65:4094–4098
Marinello AJ, Bansal SK, Paul B, Koser PL, Love J, Struck RF, Gurtoo HL (1984) Metabolism and binding of cyclophosphamide and its metabolite acrolein to rat hepatic microsomal cytochrome P-450. Cancer Res 44:4615–4621
Maxwell ES (1957) The enzymic interconversion of uridine diphosphogalactose and uridine diphosphoglucose. J Biol Chem 229:139–151
Mott JE, Grant RA, Ho YS, Platt T (1985) Maximizing gene expression from plasmid vectors containing the λ PL promoter: strategies for overproducing transcription termination factor ρ. Proc Natl Acad Sci U S A 82:88–92
Neudecker T, Eder E, Deininger D, Henschler D (1991) Mutagenicity of 2-methylacrolein, 2-ethylacrolein and 2-propylacrolein in Salmonella typhimurium TA100. A comparative study. Mutat Res 264:193–196
Pérez JM, Calderón IL, Arenas FA, Fuentes DE, Pradenas GA, Fuentes EL, Sandoval JM, Castro ME, Elías AO, Vásquez CC (2007) Bacterial toxicity of potassium tellurite: unveiling an ancient enigma. PLoS ONE 2:e211
Pérez JM, Arenas FA, Pradenas GA, Sandoval JM, Vásquez CC (2008) Escherichia coli YqhD exhibits aldehyde reductase activity and protects from the harmful effect of lipid peroxidation-derived aldehydes. J Biol Chem 283:7346–7353
Persson B, Krook M, Jörnvall H (1991) Characteristics of short-chain alcohol dehydrogenases and related enzymes. Eur J Biochem 200:537–543
Prust C, Hoffmeister M, Liesegang H, Wiezer A, Fricke WF, Ehrenreich A, Gottschalk G, Deppenmeier U (2005) Complete genome sequence of the acetic acid bacterium Gluconobacter oxydans. Nat Biotechnol 23:195–200
Raner GM, Chiang EW, Vaz AD, Coon MJ (1997) Mechanism-based inactivation of cytochrome P450 2B4 by aldehydes: relationship to aldehyde deformylation via a peroxyhemiacetal intermediate. Biochemistry 36:4895–4902
Salusjärvi T, Povelainen M, Hvorslev N, Eneyskaya EV, Kulminskaya AA, Shabalin KA, Neustroev KN, Kalkkinen N, Miasnikov AN (2004) Cloning of a gluconate/polyol dehydrogenase gene from Gluconobacter suboxydans IFO 12528, characterisation of the enzyme and its use for the production of 5-ketogluconate in a recombinant Escherichia coli strain. Appl Microbiol Biotechnol 65:306–314
Schweiger P, Volland S, Deppenmeier U (2007) Overproduction and characterization of two distinct aldehyde-oxidizing enzymes from Gluconobacter oxydans 621H. J Mol Microbiol Biotechnol 13:147–155
Schweiger P, Gross H, Wesener S, Deppenmeier U (2008) Vinyl ketone reduction by three distinct Gluconobacter oxydans 621H enzymes. Appl Microbiol Biotechnol 80:995–1006
Semchyshyn H, Bagnyukova T, Storey K, Lushchak V (2005) Hydrogen peroxide increases the activities of soxRS regulon enzymes and the levels of oxidized proteins and lipids in Escherichia coli. Cell Biol Int 29:898–902
Skerra A (1994) Use of the tetracycline promoter for the tightly regulated production of a murine antibody fragment in Escherichia coli. Gene 151:131–135
Szweda LI, Uchida K, Tsai L, Stadtman ER (1993) Inactivation of glucose-6-phosphate dehydrogenase by 4-hydroxy-2-nonenal. Selective modification of an active-site lysine. J Biol Chem 268:3342–3347
Thoden JB, Hegeman AD, Wesenberg G, Chapeau MC, Frey PA, Holden HM (1997) Structural analysis of UDP-sugar binding to UDP-galactose 4-epimerase from Escherichia coli. Biochemistry 36:6294–6304
Uchida K, Stadtman ER (1993) Covalent attachment of 4-hydroxynonenal to glyceraldehyde-3-phosphate dehydrogenase. A possible involvement of intra- and intermolecular cross-linking reaction. J Biol Chem 268:6388–6393
Yang IY, Hossain M, Miller H, Khullar S, Johnson F, Grollman A, Moriya M (2001) Responses to the major acrolein-derived deoxyguanosine adduct in Escherichia coli. J Biol Chem 276:9071–9076
Yoon SJ, Park JE, Yang JH, Park JW (2002) OxyR regulon controls lipid peroxidation-mediated oxidative stress in Escherichia coli. J Biochem Mol Biol 35:297–301
Acknowledgements
This project was supported by funds from Bundesministerium für Bildung und Forschung; Germany (BMBF; Project-No 0313751P).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schweiger, P., Deppenmeier, U. Analysis of aldehyde reductases from Gluconobacter oxydans 621H. Appl Microbiol Biotechnol 85, 1025–1031 (2010). https://doi.org/10.1007/s00253-009-2154-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2154-x