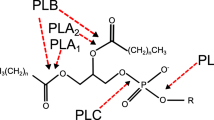
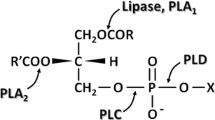
Abstract
Phospholipids are present in all living organisms. They are a major component of all biological membranes, along with glycolipids and cholesterol. Enzymes aimed at modifying phospholipids, namely, phospholipases, are consequently widespread in nature, playing very diverse roles from aggression in snake venom to signal transduction and digestion in humans. In this review, we give a general overview of phospholipases A1, A2, C and D from a sequence and structural perspective and their industrial application. The use of phospholipases in industrial processes has grown hand-in-hand with our ability to clone and express the genes in microbial hosts with commercially attractive amounts. Further, the use in industrial processes is increasing by optimizing the enzymes by protein engineering. Here, we give a perspective on the work done to date to express phospholipases in heterologous hosts and the efforts to optimize them by protein engineering. We will draw attention to the industrial processes where phospholipases play a key role and show how the use of a phospholipase for oil degumming leads to substantial environmental benefits. This illustrates a very general trend: the use of enzymes as an alternative to chemical processes to make products often provides a cleaner solution for the industrial processes. In a world with great demands on non-polluting, energy saving technical solutions—white biotechnology is a strong alternative.


Similar content being viewed by others
References
Aalrust E, Beyer W, Ottofrickenstein H, Penk G, Plainer H, Reiner R (1992) Enzymatic treatment of edible oils, ROEHM GMBH, METALLGESELLSCHAFT AG, [US5264367]
Ackermann EJ, Dennis EA (1995) Mammalian calcium-independent phospholipase A(2). Biochim Biophys Acta 1259:125–136
Albermann K, Kemmner W, Kimpel E, Maier D, Spreafico F, Stock A, Wagner C, Boer DL, Meima RB, Bernhard RM (2003) New polynucleotide encoding a phospholipase, useful in producing dough or baked product of dough. DSM, The Netherlands. PCT International Patent Application WO2003097825
Antikainen NM, Martin SF (2005) Altering protein specificity: techniques and applications. Bioorg Med Chem 13:2701–2716
Aoki J, Nagai Y, Hosono H, Inoue K, Arai H (2002) Structure and function of phosphatidylserine-specific phospholipase A1. Biochim Biophys Acta 1582:26–32
Asahi Kasei Kogyo (1994) Pure microbe producing phospholipase A2 from Streptomyces—is transformed by recombinant plasmid pPLA101, useful for preparing food additive phospholipid from synthetic lecithin. Asahi Kasei Kogyo KK, Japan. Japan Patent Application JP6327468
Bekkers AC, van der Vuurst H, van Willigen G, Akkerman JW, Verheij HM (1995) Targeting of porcine pancreatic phospholipase A2 to human platelets: introduction of an RGD sequence by genetic engineering. Thromb Haemost 74:1138–1144
Bhat MK, Pickersgill RW, Perry BN, Brown RA, Jones ST, Mueller-Harvey I, Sumner IG, Goodenough PW (1993) Modification of the head-group selectivity of porcine pancreatic phospholipase A2 by protein engineering. Biochemistry 32:12203–12208
Bojsen K, Svendsen A, Fuglsang CC, Patkar S, Borch K, Vind J, Petri AG, Glad SS, Budolfsen G, Schroder GSO (2000) Producing a lipolytic enzyme variant useful in baking processes and purification of vegetables oils, comprises altering the parent lipolytic enzyme, preparing a variant and testing its activity on selected ester bond. Novozymes A/S, Denmark. PCT International Patent Application WO2000/32758
Borch K, Erlandsen L, Vind J, Svendsen A, Jørgensen CT (2004) New variant polypeptide of fungal lipolytic enzyme, having hydrolytic activity with respect to an ester bond in a polar lipid, useful in dough for making bread and other baked products. Novozymes A/S, Denmark. PCT International Patent Application WO2004/099400
Bornscheuer UT, Kazlauskas RJ (2004) Catalytic promiscuity in biocatalysis: using old enzymes to form new bonds and follow new pathways. Angew Chem Int Ed Engl 43:6032–6040
Budolfsen G, Christiansen L, Spendler T (2000) Lipolytic enzymes in the baking industry. In: Wooton M, Batey IL, Wrigley CW (eds) Cereals 2000. Proceedings of the 11th International ICC Cereals and Bread Congress. Royal Australian Chemical Institute, Melbourne
Campbell JS, Te Bokkel DW, Thatcher KD (2004) Manufacture of modified liquid egg yolk product, used as emulsifier e.g., for sauces, comprises processing phospholipase A-containing microbial fermentate. US Patent Application US6773731
Canaan S, Zadori Z, Ghomashchi F, Bollinger J, Sadilek M, Moreau ME, Tijssen P, Gelb MH (2004) Interfacial enzymology of parvovirus phospholipases A2. J Biol Chem 279:14502–14508
Carriere F, Withers-Martinez C, van Tilbeurgh H, Roussel A, Cambillau C, Verger R (1998) Structural basis for the substrate selectivity of pancreatic lipases and some related proteins. Biochim Biophys Acta 1376:417–432
Castello P, Jollet S, Potus J, Baret JL, Nicolas J (1998) Effect of exogenous lipase on dough lipids during mixing of wheat flours. Cereal Chem 75:595–601
Ciofalo V, Barton N, Kreps J, Coats I, Shanahan D (2006) Safety evaluation of a lipase enzyme preparation, expressed in Pichia pastoris, intended for use in the degumming of edible vegetable oil. Regul Toxicol Pharmacol 45:1–8
Clausen K (2001) Enzymatic oil-degumming by a novel microbial phospholipase. Eur J Lipid Sci Technol 103:333–340
Clausen IG, Patkar SA, Borch K, Halkier T, Barfoed M, Clausen K, Fuglsang CC, Dybdal L (1998) New isolated phospholipase from Fusarium oxysporum—used for e.g. reducing phosphorus content of edible oils, treatment of starch hydrolysates, production of animal feed or in detergent or cleaning compositions. Novozymes A/S, Denmark. PCT International Patent Application WO1998/26057
Dekker N (2000) Outer-membrane phospholipase A: known structure, unknown biological function. Mol Microbiol 35:711–717
Dupureur CM, Yu BZ, Mamone JA, Jain MK, Tsai MD (1992) Phospholipase A2 engineering. The structural and functional roles of aromaticity and hydrophobicity in the conserved phenylalanine-22 and phenylalanine-106 aromatic sandwich. Biochemistry 31:10576–10583
Dutilh CE, Groger W (1981) Improvement of product attributes of mayonnaise by enzymic hydrolysis of egg yolk with phospholipase A2. J Sci Food Agric 32:451–458
Eliason AC, Larsson K (1993) Cereals in bread making. Marcel Dekker, New York
Gan Z, van der Graaf J, Leonard SA, Brooker BE, Parker ML, Schofield JD (1999) The role of wheat proteins and polar lipids in the stabilisation of the foam structure in dough. In: Campbell GM, Webb C, Pandiella SS, Nirajan K (eds) Bubbles in food. Eagan Press, St. Paul, Minnesota, pp 89–93
Gramatikova S, Hazlewood G, Lam D, Barton NR, Sturgis BG, Robertson DE, Li J, Kreps JA, Fielding R, Brown RC, Vasavada A, Tan X, Badillo A, van Hoek WP, Janssen G, Isaac C, Burk MJ (2005) NewMut isolated or recombinant polypeptide having phospholipase activity, useful for degumming oil, caustic refining of a phospholipid-containing composition, purifying a phytosterol or a triterpene, or refining a crude oil. Diversa, USA. PCT International Patent Application WO2005086900
Heikinheimo P, Goldman A, Jeffries C, Ollis DL (1999) Of barn owls and bankers: a lush variety of alpha/beta hydrolases. Structure 7:R141–R146
Heinz DW, Essen LO, Williams RL (1998) Structural and mechanistic comparison of prokaryotic and eukaryotic phosphoinositide-specific phospholipases C. J Mol Biol 275:635–650
Hirabayashi T, Murayama T, Shimizu T (2004) Regulatory mechanism and physiological role of cytosolic phospholipase A2. Biol Pharm Bull 27:1168–1173
Hiramatsu T, Sonoda H, Takanezawa Y, Morikawa R, Ishida M, Kasahara K, Sanai Y, Taguchi R, Aoki J, Arai H (2003) Biochemical and molecular characterization of two phosphatidic acid-selective phospholipase A1s, mPA-PLA1alpha and mPA-PLA1beta. J Biol Chem 278:49438–49447
Inoue S, Ota S (1984) Composition for improving elasticity of bread and cereals—contains phospholipase. Kyowa Hakko Kogyo KK, Japan. European Patent Application EP109244
Jaeger KE, Dijkstra BW, Reetz MT (1999) Bacterial biocatalysts: molecular biology, three-dimensional structures, and biotechnological applications of lipases. Annu Rev Microbiol 53:315–351
Janssen MJ, Burghout PJ, Verheij HM, Slotboom AJ, Egmond MR (1999) Introduction of a C-terminal aromatic sequence from snake venom phospholipases A2 into the porcine pancreatic isozyme dramatically changes the interfacial kinetics. Eur J Biochem 263:782–788
Katan M (1998) Families of phosphoinositide-specific phospholipase C: structure and function. Biochim Biophys Acta 1436:5–17
Kauffmann I, Schmidt-Dannert C (2001) Conversion of Bacillus thermocatenulatus lipase into an efficient phospholipase with increased activity towards long-chain fatty acyl substrates by directed evolution and rational design. Protein Eng 14:919–928
Kawai S (2004) Characterization of diacylglycerol oil mayonnaise emulsified using phospholipase A2-treated egg yolk. J Am Oil Chem Soc 81:993–998
Kazlauskas RJ (2005) Enhancing catalytic promiscuity for biocatalysis. Curr Opin Chem Biol 9:195–201
King TP, Kochoumian L, Joslyn A (1984) Wasp venom proteins: phospholipase-A1 and B. Arch Biochem Biophys 230:1–12
Krog N (1971) Amylose complexing effect on food grade emulsifiers. Starch/Stärke 23:206–210
Lappas M, Rice GE (2004) Phospholipase A2 isozymes in pregnancy and parturition. Prostaglandins Leukot Essent Fatty Acids 70:87–100
Leiros I, Secundo F, Zambonelli C, Servi S, Hough E (2000) The first crystal structure of a phospholipase D. Structure 8:655–667
Liscovitch M, Czarny M, Fiucci G, Tang X (2000) Phospholipase D: molecular and cell biology of a novel gene family. Biochem J 345:401–415
Liu XH, Zhu HX, Huang BH, Rogers J, Yu BZ, Kumar A, Jain MK, Sundaralingam M, Tsai MD (1995) Phospholipase A(2) engineering—probing the structural and functional roles of N-terminal residues with site-directed mutagenesis, X-ray, and NMR. Biochemistry 34:7322–7334
Loeffler F, Jungschaffer G, Khanh QN, Schuster E, Sproessler B, Wolf S, Loffler F, Sprossler B, Khanh Q, Luefler F (1998) New two-chain form of Aspergillus lyso-phospholipase—useful for degumming plant oils, as baking aid and generally for hydrolysis of phospholipase. Röhm GMBH and AB Enzymes GMBH, Germany. PCT International Patent Application WO1998/31790
MacMurray TA, Morrison WR (1970) Composition of wheat-flour lipids. J Sci Food Agric 21:520–528
Macphee CH, Nelson JJ, Zalewski A (2005) Lipoprotein-associated phospholipase A2 as a target of therapy. Curr Opin Lipidol 16:442–446
Macritch F, Gras PW (1973) Role of flour lipids in baking. Cereal Chem 50:292–302
McDermott M, Wakelam MJ, Morris AJ (2004) Phospholipase D. Biochem Cell Biol 82:225–253
Nebesny E, Rosicka J, Pierzgalski T (1998) Enzymatic hydrolysis of wheat starch into glucose. Starch/Stärke 50:337–341
Nevalainen KMH, Te’o VSJ, Bergquist PL (2005) Heterologous protein expression in filamentous fungi. Trends Biotechnol 23:468–474
Nielsen PM (2000) Producing cheese for products such as pizza, involves treating cheese milk or its fraction with phospholipases and producing cheese from cheese milk which is also obtained by reversing process. Novozymes A/S, Denmark. PCT International Patent Application WO2000/54601
Nielsen EW (2004) Principles of cheese production. In: Hui YH, Meunier-Goddik L, Solvejg Hansen Å, Josephsen J, Nip WK, Stanfield PS, Toldra F (eds) Handbook of food and beverage fermentation technology. Marcel Dekker, New York, pp 221–239
Noel JP, Bingman CA, Deng TL, Dupureur CM, Hamilton KJ, Jiang RT, Kwak JG, Sekharudu C, Sundaralingam M, Tsai MD (1991) Phospholipase A2 engineering. X-ray structural and functional evidence for the interaction of lysine-56 with substrates. Biochemistry 30:11801–11811
Ollis DL et al (1992) The alpha/beta hydrolase fold. Protein Eng 5:197–211
Oxenbøll KM (2005) Life cycle assessment on enzymatic degumming of soybean oil. Novozymes A/S Internal Report. (Critically reviewed by Prof. Henrik Wenzel from the Technical University of Denmark)
Patkar SA, Higgins D, Fatum TM, Vind J, Madkor S, Sørensen TL (2004) New variants of fungal phospholipases having a lower lipase activity on triglycerides than the parent enzyme, useful in cheese-making processes. Novozymes A/S, Denmark; Novozymes, USA; Christian Hansen A/S, Denmark. PCT International Patent Application WO2004/111216
Phillis JW, O’Regan MH (2003) The role of phospholipases, cyclooxygenases, and lipoxygenases in cerebral ischemic/traumatic injuries. Crit Rev Neurobiol 15:61–90
Poulsen CH, Søe JB (1997) Effect and functionality of lipases in dough and bread. In: Angelino SAGF, Hamer RJ, van Hartingsveldt W, Heidekamp F, van der Lugt JP (eds) First European symposium on enzymes and grain processing. TNO Nutrition and Food Research Institute, The Netherlands, pp 204–214
Poulsen CH, Søe JB, Rasmussen P, Madrid SM, Zargahi MR (1998) New Aspergillus tubigensis lipase—used as dough additive to reduce pore size in the crumb, increase pore homogeneity and increase gluten index. Danisco A/S, Denmark. PCT International Patent Application WO1998/45453
Sankyo (1998) Aspergillus-derived phospholipase A1 gene—used for the recombinant production of phospholipase A of high purity and in a high yield. Sankyo, Japan. Japan Patent Application JP10155493
Reimerdes EH, Franke K, Sell M (2004) Influencing functional properties of egg yolk by using phospholipases. Presentation given at the Conference on Food Structure and Food Quality on October 3–7, 2004 at Cork, Ireland. http://www.aocs.org/member/division/fsff/reimerdesernsth.pdf
Rhee J, Song J, Park TI, Lee DA (2002) Novel thermostable phospholipase TA3 and TA13 useful in various biological processes, pharmaceutical, cosmetics and food industries, is a mutant of phospholipase A1 derived from Serratia sp. strain MK1. Korea Advanced Institute for Science and Technology, Republic of Korea. US Patent Application US2002/086355
Roberts IN, Jeenes DJ, MacKenzie DA, Wilkinson AP, Sumner IG, Archer DB (1992) Heterologous gene expression in Aspergillus niger of a glucoamylase-porcine pancreatic prophospholipase A2 fusion protein is secreted and processed to yield mature enzyme. Gene 122:155–161
Ross AS, Macritchie F (1995) Interactions of wheat proteins, carbohydrates and lipids. In: Goankar AG (ed) Ingredient interactions: effects on food quality. Marcel Dekker, New York, pp 321–356
Sato T et al (1997) Serine phospholipid-specific phospholipase A that is secreted from activated platelets. A new member of the lipase family. J Biol Chem 272:2192–2198
Scandella CJ, Kornberg A (1971) Membrane-bound phospholipase A1 purified from Escherichia coli. Biochemistry 10:4447–4456
Sekar K, Yu BZ, Rogers J, Lutton J, Liu XH, Chen X, Tsai MD, Jain MK, Sundaralingam M (1997) Phospholipase A(2) engineering. Structural and functional roles of the highly conserved active site residue aspartate-99. Biochemistry 36:3104–3114
Sekharudu C, Ramakrishnan B, Huang B, Jiang RT, Dupureur CM, Tsai MD, Sundaralingam M (1992) Crystal structure of the Y52F/Y73F double mutant of phospholipase A2: increased hydrophobic interactions of the phenyl groups compensate for the disrupted hydrogen-bonds of the tyrosines. Protein Sci 1:1585–1594
Servi S (1999) Phospholipases as synthetic catalysts. Top Curr Chem 200:127–158
Shiba Y, Ono C, Fukui F, Watanabe I, Serizawa N, Gomi K, Yoshikawa H (2001) High-level secretory production of phospholipase A1 by Saccharomyces cerevisiae and Aspergillus oryzae. Biosci Biotechnol Biochem 65:94–101
Shimizu T, Ohto T, Kita Y (2006) Cytosolic phospholipase A2: Biochemical properties and physiological roles. IUBMB Life 58:328–333
Si JQ (1996) New enzymes for the baking industry. Food Tech Eur 3:60–64
Simons JW, Adams H, Cox RC, Dekker N, Gotz F, Slotboom AJ, Verheij HM (1996) The lipase from Staphylococcus aureus. Expression in Escherichia coli, large-scale purification and comparison of substrate specificity to Staphylococcus hyicus lipase. Eur J Biochem 242:760–769
Six DA, Dennis EA (2000) The expanding superfamily of phospholipase A(2) enzymes: classification and characterization. Biochim Biophys Acta 1488:1–19
Song JK, Kim MK, Rhee JS (1999) Cloning and expression of the gene encoding phospholipase A1 from Serratia sp. MK1 in Escherichia coli. J Biotechnol 72:103–114
Song JK, Chung B, Oh YH, Rhee JS (2002) Construction of DNA-shuffled and incrementally truncated libraries by a mutagenic and unidirectional reassembly method: changing from a substrate specificity of phospholipase to that of lipase. Appl Environ Microbiol 68:6146–6151
Soragni E, Bolchi A, Balestrini R, Gambaretto C, Percudani R, Bonfante P, Ottonello S (2001) A nutrient-regulated, dual localization phospholipase A(2) in the symbiotic fungus Tuber borchii. EMBO J 20:5079–5090
Stringer MA, Fatum TM, Patkar SA (2004) Novel phospholipase polypeptide having 80% identity with amino acid sequence of Tuber albidum, Verticillium tenerum or Aspergillus oryzae, useful for producing cheese, preparing dough and reducing content of phosphorus in vegetable oil. Novozymes A/S, Denmark; Christian Hansen A/S, Denmark. PCT International Patent Application WO2004/097012
Suzuki S, Yamatoya H, Sakai M, Kataoka A, Furushiro M, Kudo S (2001) Oral administration of soybean lecithin transphosphatidylated phosphatidylserine improves memory impairment in aged rats. J Nutr 131:2951–2956
Tanaka T, Kimura S, Ota Y (1988) Secretion of the proenzyme and active bovine pancreatic phospholipase A2 enzyme by Saccharomyces cerevisiae: design and use of a synthetic gene. Gene 64:257–264
Tsutsumi N, Sasaki Y, Rey MW, Zaretsky E, Spendler T, Vind J, Golightly EJ (2002) New Fusarium venenatum, F. sulphureum, Acremonium berkeleyanum, F. culmorum or F. solani lipolytic enzyme encoding polynucleotides, encoding lipolytic enzymes which are useful for preparing dough, bread and cakes. Novozymes A/S, Denmark; Novozymes, USA. WO2002/00852
Tyndall JD, Sinchaikul S, Fothergill-Gilmore LA, Taylor P, Walkinshaw MD (2002) Crystal structure of a thermostable lipase from Bacillus stearothermophilus P1. J Mol Biol 323:859–869
Ulbrich-Hofmann R (2000) Phospholipases used in lipid transformation. In: Bornscheuer UT (ed) Enzymes in lipid modification. Wiley-VCH, Weinheim, pp 219–262
Ulbrich-Hofmann R, Lerchner A, Oblozinsky M, Bezakova L (2005) Phospholipase D and its application in biocatalysis. Biotechnol Lett 27:535–544
Unilever (1976) De-waxing triglyceride oils-by adding a strong acid or anhydride, followed by water and removing the sludge obtained. Unilever, UK. US Patent Application US4049686
van Dam AF (1978) Stabilised Emulsion comprising phospholipo-protein. UNIVELER [UK1525929]
van den Bergh CJ, Bekkers AC, De Geus P, Verheij HM, De Haas GH (1987) Secretion of biologically active porcine prophospholipase A2 by Saccharomyces cerevisiae. Use of the prepro sequence of the alpha-mating factor. Eur J Biochem 170:241–246
van Kampen MD, Egmond MR (2000) Directed evolution: from a staphylococcal lipase to a phospholipase. Eur J Lipid Sci Technol 102:717–726
van Oort MG, Deveer AM, Dijkman R, Tjeenk ML, Verheij HM, De Haas GH, Wenzig E, Gotz F (1989) Purification and substrate specificity of Staphylococcus hyicus lipase. Biochemistry 28:9278–9285
Vind J, Patkar SA (2005) Preparing modified polypeptide comprises providing a sequence encoding a parent polypeptide comprising at least two Cys residues forming a disulfide bond. Novozymes A/S, Denmark. WO2005/024012
Wenzel H, Hauchild M, Alting L (2000) Environmental Assessment of products. Volume 1: methodology, tools and case studies in product development. Dordrecht, The Netherlands: Kluwer Academic Publishers
Winstead MV, Balsinde J, Dennis EA (2000) Calcium-independent phospholipase A(2): structure and function. Biochim Biophys Acta 1488:28–39
Yoshinori M (1997) Structural and functional changes of hen’s egg yolk low-density lipoproteins with phospholipase A2. J Agric Food Chem 45:4558–4563
Zhu HX, Dupureur CM, Zhang XY, Tsai MD (1995) Phospholipase A2 engineering. The roles of disulfide bonds in structure, conformational stability, and catalytic function. Biochemistry 34:15307–15314
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00253-007-0986-9
Rights and permissions
About this article
Cite this article
De Maria, L., Vind, J., Oxenbøll, K.M. et al. Phospholipases and their industrial applications. Appl Microbiol Biotechnol 74, 290–300 (2007). https://doi.org/10.1007/s00253-006-0775-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0775-x