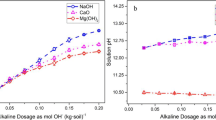
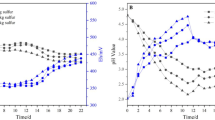
Abstract
Contaminated soil from a historical industrial site and containing sulfide ore ashes and aromatic hydrocarbons underwent sequential leaching by 0.5 M citrate and microbial treatments. Heavy metals leaching was with the following efficiency scale: Cu (58.7%) > Pb (55.1%) > Zn (44.5%) > Cd (42.9%) > Cr (26.4%) > Ni (17.7%) > Co (14.0%) > As (12.4%) > Fe (5.3%) > Hg (1.1%) and was accompanied by concomitant removal of organic contaminants (about 13%). Leached metals were concentrated into an iron gel, produced during ferric citrate fermentation by the metal-resistant strain BAS-10 of Klebsiella oxytoca. Concomitantly, the acidic leached soil was bioaugmented with Allescheriella sp. DABAC 1, Stachybotrys sp. DABAC 3, Phlebia sp. DABAC 9, Pleurotus pulmonarius CBS 664.97, and Botryosphaeria rhodina DABAC P82. B. rhodina was most effective, leading to a significant depletion of the most abundant contaminants, including 7-H-benz[DE]anthracene-7-one, 9,10-anthracene dione and dichloroaniline isomers, and to a marked detoxification as assessed by the mortality test with the Collembola Folsomia candida Willem. The overall degradation activities of B. rhodina and P. pulmonarius appeared to be significantly enhanced by the preliminary metal removal.




Similar content being viewed by others
References
Abraitis PK, Pattrick RAD, Kelsall GH, Vaughan DJ (2004) Acid leaching and dissolution of major sulphide ore minerals: processes and galvanic effects in complex systems. Mineral Mag 68:343–351
Asim RK, Abhijit R, Nirmolendu R (1987) Structure of the capsular polysaccharide of Klebsiella serotype K 40. Carbohydr Res 165:77–86
Baldi F, Minacci A, Pepi M, Scozzafava A (2001) Gel sequestration of heavy metals by Klebsiella oxytoca isolated from iron mat. FEMS Microbiol Ecol 36:169–174
Bennett JW, Faison BD (1997) Use of fungi in bioremediation. In: Hurst CJ (ed) Manual of environmental microbiology. ASM, Washington, USA, pp 758–765
Berselli S, Milone G, Canepa P, Di Gioia D, Fava F (2004) Effect of cyclodextrins, humic substances and rhamnolipids on the washing of a historically contaminated soil and on the aerobic bioremediation of the resulting effluents. Biotechnol Bioeng 88:111–120
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Choinowski T, Blodig W, Winterhalter KH, Piontek K (1999) The crystal structure of lignin peroxidase at 1.7 Å resolution reveals a hydroxy group on the C-beta of tryptophan 171: a novel radical site formed during the redox cycle. J Mol Biol 286:809–827
Concas A, Ardau C, Cristini A, Zuddas P, Cao G (2006) Mobility of heavy metals from tailings to stream waters in a mining activity contaminated site. Chemosphere 63:244–253
Conte P, Agretto A, Spaccini R, Piccolo A (2005) Soil remediation: humic acids as natural surfactants in the washings of highly contaminated soils. Environ Pollut 135:515–522
D’Annibale A, Ricci M, Leonardi V, Quaratino D, Mincione E, Petruccioli M (2005) Degradation of aromatic hydrocarbons by white-rot fungi in a historically contaminated soil. Biotechnol Bioeng 90:723–731
D’Annibale A, Rosetto F, Leonardi V, Federici F, Petruccioli M (2006) Role of autochthonous filamentous fungi in bioremediation of a soil historically contaminated with aromatic hydrocarbons. Appl Environ Microbiol 72:28–36
EPA (Environmental Protection Agency)-USA (1986) Method 7380—Iron (atomic absorption, direct aspiration) (Revision 0)
EPA (Environmental Protection Agency)-USA (1994a) Method 3051—Microwave assisted acid digestion of sediments, sludges, soils, and oils (Revision 0)
EPA (Environmental Protection Agency)-USA (1994b) Method 6020—Inductively coupled plasma-mass spectrometry (Revision 0)
Ettler V, Komárková M, Jehlička J, Coufal P, Hradil D, Machovič V, Delorme F (2004) Leaching of lead metallurgical slag in citric solutions—implications for disposal and weathering in soil environments. Chemosphere 57:567–577
Fava F, Bertin L, Marchetti L (2002) Studio di “biotrattabilità” del suolo ACNA A5 136 B. In: Canepa P (ed) Progetto Sisifo—Soil Remediation Series no. 1. Poligrafica Venezia, pp 73–80
Fenice M, Giovannozzi Sermanni G, Federici F, D’Annibale A (2003) Submerged and solid-state production of laccase and manganese-peroxidase production by Panus tigrinus on olive-mill wastewater-based media. J Biotechnol 100:77–85
George P, Kvaratskhelia M, Dilworth MJ, Thorneley RNF (1999) Reversible alkaline inactivation of lignin peroxidase involves the release of both the distal and proximal site calcium ions and bishistidine co-ordination of the haem. Biochem J 344:237–244
Gianfreda L, Rao AM (2004) Potential of extra-cellular enzymes in remediation of polluted soils: a review. Enzyme Microb Technol 35:339–354
Glusker JP (1980) Citrate conformation and chelation: enzymatic implication. Acad Chem Res 13:345–352
Guetta O, Milas M, Rinaudo M (2003) Structure and properties of a bacterial polysaccharide from a Klebsiella Strain (ATCC 12657). Biomacromolecules 4:1372–1379
Leclerc N, Meux E, Lecuire JM (2002) Hydrometallurgical recovery of zinc and lead from electric arc furnace dust using mononitrilotriacetate anion and hexahydrated ferric chloride. J Hazard Mater 91:257–270
Masaphy S, Levanon D, Henis Y, Venkateswarlu K, Kelly SL (1995) Microsomal and cytosolic cytochrome P450 mediated benzo(a)pyrene hydroxylation in Pleurotus pulmonarius. Biotechnol Lett 17:969–974
Mori T, Kondo R (2002) Oxidation of dibenzo-p-dioxin, dibenzofuran, biphenyl, and diphenyl ether by the white-rot fungus Phlebia lindtneri. Appl Microbiol Biotechnol 60:200–205
Novotny C, Erbanov0a P, Ŝaŝek V, Kubatova A, Cajthaml T, Lang E, Krahl J, Zadrazil F (1999) Extracellular oxidative enzyme production and PAH removal in soil by exploratory mycelium of white rot fungi. Biodegradation 10:159–168
Peters RW (1999) Chelant extraction of heavy metals from contaminated soils. J Hazard Mater 66:151–210
Pointing SB (2001) Feasibility of bioremediation by white-rot fungi. Appl Microbiol Biotechnol 57:20–33
Reed BE, Carriere PC, Moore R (1996) Flushing of a Pb II contaminated soil using HCl, EDTA, and CaCl2. J Environ Eng 122:48–50
Ŝaŝek V (2003) Why mycoremediations have not yet come into practice. In: Sasek V, Glaser JA, Baveye Ph (eds) The utilization of bioremediation to reduce soil contamination: problems and solution. Kluwer, Netherlands, pp 247–266
Schwab AP, He Y, Banks MK (2005) The influence of organic ligands on the retention of lead in soil. Chemosphere 61:856–866
Singh AV, Gupta V, Menaria KL (2001) Beneficiation studies on the flotation of pyritic sulphide ore of zinc from Rajpura Dariba Mines, Dariba, (India). J Surf Sci Technol 17:93–98
Singh N, Sarkar AK, Ramchandran R, Lal K (2003) Determination of arsenic in fly ash and sulphide ore by flame atomic absorption spectrometry using hydride generator. Asian J Chem 15:1327–1330
Sutherland GRJ, Schick Zapanta L, Tien M, Aust SD (1997) Role of calcium in maintaining the heme environment of manganese peroxidase. Biochemistry 36:3654–3662
Thöming J, Stichnote H, Mangold S, Calmano W (2000) Hydrometallurgical approaches to soil remediation-process optimization applying heavy metal speciation. Land Contam Reclam 8:19–31
Tuor U, Winterhalter K, Fiechter A (1995) Enzymes of white-rot fungi involved in lignin degradation and ecological determinants in wood decay. J Biotechnol 41:1–7
Verdín J, Pogni R, Baeza A, Baratto MC, Basosi R, Vázquez-Duhalt R (2006) Mechanism of versatile peroxidase inactivation by Ca2+ depletion. Biophys Chem 121:163–170
Vinals J, Balart MJ, Roca A (2002) Inertization of pyrite cinders and co-inertization with electric arc furnace flue dusts by pyroconsolidation at solid state. Waste Manag 22:773–782
Viollier E, Inglett PW, Hunter K, Roychoudhury AN, Van Cappellen P (2000) The ferrozine method revisited: Fe(II)/Fe(III) determination in natural waters. Appl Geochem 15:785–790
Acknowledgments
The authors wish to thank the Interuniversitary Consortium “The Chemistry for the Environment” (I.N.C.A.) that supported, with the contribution of Italian Ministry of Environment, this work within the “Sisifo” Project (“ACNA-Bonifica di siti contaminati”). Further thanks are due to Prof. G. Cao (I.N.C.A. Laboratory, Cagliari, Italy) for providing diffractometric analyses of the ACNA soil.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
D’Annibale, A., Leonardi, V., Federici, E. et al. Leaching and microbial treatment of a soil contaminated by sulphide ore ashes and aromatic hydrocarbons. Appl Microbiol Biotechnol 74, 1135–1144 (2007). https://doi.org/10.1007/s00253-006-0749-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0749-z