Abstract
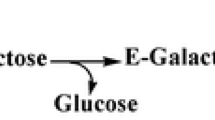
Recently, a new gene encoding β-glucuronidase from Streptococcus equi subsp. zooepidemicus (SEZ) was identified and expressed in Escherichia coli. In this paper, the characterization of the enzyme is described. Specific enzyme activity was 120,000 U/mg purified protein at 37°C and pH = 7.0. The temperature and pH value, at which the enzyme has the highest specific activity, were determined and were found to be approximately 52°C and 5.6, respectively. The mutant strain SEZ glcHis was designed for the efficient isolation of β-glucuronidase from S. equi subsp. zooepidemicus. It was observed that the specific activity of β-glucuronidase in the cytoplasmic extract of a mutated strain was about 45% lower than in the cytoplasmic extract of a wild-type strain. The specific activity of purified β-glucuronidase from SEZ glcHis was four times as low as β-glucuronidase purified from E. coli. Comparing the specific activity of purified streptococcal β-glucuronidase from E. coli with E. coli β-glucuronidase (the enzyme with the highest specific activity was supplied by Sigma), the former is 1.8 higher than the latter.





Similar content being viewed by others
References
Akao T (1999) Purification and characterization of glycyrrhetic acid monoglucuronide beta-D-glucuronidase in Eubacterium sp. GLH. Biol Pharm Bull 22:80–82
Beaud D, Tailliez P, Anba-Mondoloni J (2005) Genetic characterization of the β-glucuronidase enzyme from a human intestinal bacterium, Ruminococcus gnavus. Microbiology 151:2323–2330
Buehler HJ, Katzman PA, Doisy EA (1951) Studies on beta-glucuronidase from E. coli. Proc Soc Exp Biol Med 76:672–676
Delvin E, Gianetto R (1970) The purification of lysosomal rat-liver beta glucuronidase. Biochim Biophys Acta 220:93–100
Elvin CM, Thompson PR, Argall ME, Hendry P, Stamford NPJ, Lilley PE, Dixon NE (1990) Modified bacteriophage lambda promoter vectors for overproduction of proteins in Escherichia coli. Gene 87:123–126
Gutierrez JA, Crowley PJ, Brown DB, Hillman JD, Youngman P, Bleiweis AS (1996) Insertional mutagenesis and recovery genes of Streptococcus mutans by using transposon Tn917: preliminary characterization of mutants displaying acid sensitivity and nutritional requirements. J Bacteriol 178:4166–4175
Hoshi H, Nakagawa H, Nishiguchi S, Iwata K, Niikura K, Monde K, Nishimura S-I (2004) An engineered hyaluronan synthase. J Biol Chem 279:2341–2349
Jacox RF (1951) Beta-glucuronidase production by beta hemolytic streptococci. J Clin Invest 30:652
Jacox RF (1953) Streptococcal β-glucuronidase. J Bacteriol 65:700–705
Jefferson RA, Burgess SM, Hirsh D (1986) Beta-glucuronidase from Escherichia coli as a gene-fusion marker. Proc Natl Acad Sci USA 83:8447–8451
Krahulec J, Krahulcova J (2005) Increase in hyaluronic acid production by Streptococcus equi subsp. zooepidemicus strain deficient in beta-glucuronidase in laboratory conditions. Appl Microbiol Biotechnol 15:1–8
Lin ChW, Orcutt ML, Fishman WH (1975) Purification and characterization of mouse kidney β-glucuronidase. J Biol Chem 250:4737–4743
McCue PM, Wilson WD (1989) Equine mastitis—a review of 28 cases. Equine Vet J 21:351–353
Nimrod A (1998) Method of producing high molecular weight sodium hyaluronate by fermentation of streptococcus. US Patent 4,780,414
Plapp BV, Cole D (1966) Purification and characterization of bovine liver beta-glucuronidase. Arch Biochem Biophys 116:193–206
Russell WM, Klaenhammer TR (2001) Identification and cloning of gusA, encoding a new β-glucuronidase from Lactobacillus gasseri ADH. Appl Environ Microbiol 67:1253–1261
Stahl PD, Touster O (1971) Beta-glucuronidase of rat liver lysosomes. Purification, properties, subunits. J Biol Chem 246:5398–5406
Wilson KJ, Hughes SG, Jefferson RA (1992) The Escherichia coli gus operon: induction and expression of the gus operon in E. coli and the occurrence and use of GUS in other bacteria. In: Gallagher SR (ed) GUS protocols: using the GUS gene as a reporter of gene expression. Academic, San Diego, CA
Acknowledgement
This work was supported by CPN spol. s r.o., Dolní Dobrouč 401, Czech Republic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krahulec, J., Krahulcová, J. Characterization of the new β-glucuronidase from Streptococcus equi subsp. zooepidemicus . Appl Microbiol Biotechnol 74, 1016–1022 (2007). https://doi.org/10.1007/s00253-006-0745-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0745-3