Abstract
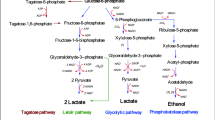
In this study, the behaviour of Lactobacillus fermentum CRL 722 and CRL 251 were evaluated under different pH conditions (pH 6.0, 5.5, 5.0, 4.5) and without pH control. Growth was similar under all conditions assayed except at pH 4.5. These microorganisms were able to eliminate raffinose, a nondigestible α-oligosaccharide (NDO) found in soy products, showing a consumption rate of 0.25 g l−1 h−1 (pH 6.0–5.0). The removal of raffinose was due to the high α-galactosidase (α-gal) activities of these lactic acid bacteria, which was highest at pH 5.5 (5.0 U/ml). The yield of organic acids produced during raffinose consumption was also highest at this pH. The results of this study will allow selection of the optimum growth conditions of L. fermentum with elevated levels of α-gal to be used in the reduction of NDO in soy products when used as starter cultures.




Similar content being viewed by others
References
Adams MR, Hall CJ (1988) Growth inhibition of food-borne pathogens by lactic and acetic acids and their mixtures. Int J Food Sci Technol 23:287–292
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cabo ML, Braber AF, Koenraad PM (2002) Apparent antifungal activity of several lactic acid bacteria against Penicillium discolor is due to acetic acid in the medium. J Food Prot 65:1309–1316
Church FC, Meyers SP, Srinivasan VR (1980) Isolation and characterization of alpha-galactosidase from Pichia guilliermondii. In: Underkofler LA, Wulf ML (eds) Developments in industrial microbiology. Society for Industrial Microbiology. Arlington, Va., pp 339–348
Fuller R (1992) History and development of probiotics. In: Probiotics—the scientific basis. Chapman and Hall, London, pp 1–8
Gardiner GE, Heinemann C, Baroja ML, Bruce AW, Beverman D, Madrenas J, Reid G (2002) Oral administration of the probiotic combination Lactobacillus rhamnosus GR-1 and L. fermentum RC-14 for human intestinal applications. Int Dairy J 12:191–196
Garro MS, Savoy de Giori G, Font de Valdez G, Oliver G (1993) Characterization of alpha-galactosidase from Lactobacillus fermentum. J Appl Bacteriol 75:485–488
Garro MS, Savoy de Giori G, Font de Valdez G, Oliver G (1994) α-d-Galactosidase (EC 3.2.1.22) from Bifidobacterium longum. Lett Appl Microbiol 19:16–19
Garro MS, Font de Valdez G, Oliver G, Savoy de Giori G (1996) Purification of α-galactosidase from Lactobacillus fermentum. J Biotechnol 45:103–109
Garro MS, Font de Valdez G, Oliver G, Savoy de Giori G (1998) Growth characteristics and fermentation products of Streptococcus salivarius subs. thermophilus, Lactobacillus casei and L. fermentum in soymilk. Z Lebensm Unters Forsch A 206:72–75
Garro MS, Font de Valdez G, Oliver G, Savoy de Giori G (1999) Hydrolysis of soya milk oligosaccharides by Bifidobacterium longum CRL 849. Z Lebensm Unters Forsch A 208:57–59
Garro MS, Font de Valdez G, Savoy de Giori G (2003) Temperature effect on activity of Bifidobacterium longum CRL 849 and Lactobacillus fermentum CRL 251 in mixed cultures grown in soymilk. Food Microbiol (in press)
Kandler O (1983) Carbohydrate metabolism in lactic acid bacteria. Antonie Van Leeuwenhoek 49:209–224
Kuriyama S, Mendel LB (1917) The physiological behavior of raffinose. J Biol Chem 31:125–147
Leske KL, Jevne CJ, Coon CN (1993) Effect of oligosaccharide additions on nitrogen-corrected true metabolizable energy of soy protein concentrate. Poult Sci 72:664–668
McIlvaine TC (1921) A buffer solution for colorimetric comparison. J Biol Chem 49:183–186
Mital BK, Steinkraus KH (1975) Utilization of oligosaccharides by lactic acid bacteria during fermentation of soy milk. J Food Sci 40:114–118
Mital BK, Shallenberger RS, Steinkraus KH (1973) Alpha-galactosidase activity of lactobacilli. Appl Microbiol 26:783–788
Montelongo JL, Chassy BM, McCord JD (1993) Lactobacillus salivarius for conversion of soy molasses into lactic acid. J Food Sci 58:863–866
Quigley ME, Hudson GJ, Englyst HN (1999) Determination of resistant short-chain carbohydrates (non-digestible oligosaccharides) using gas-liquid chromatography. Food Chem 65:381–390
Silvestroni A, Connes C, Sesma F, Savoy de Giori G, Piard JC (2002) Characterization of the meA locus for alpha-galactosidase in Lactobacillus plantarum. Appl Environ Microbiol 68:5464–5471
Slominski BA (1994) Hydrolysis of galactooligosaccharides by commercial preparations of alpha-galactosidase and beta-fructofuranose: potential for use as dietary additives. J Sci Food Agric 65:323–330
Stiles ME, Holzapfel WH (1997) Lactic acid bacteria of foods and their current taxonomy. Int J Food Microbiol 36:1–29
Zaika LL (2002) Effect of organic acids and temperature on survival of Shigella flexneri in broth at pH 4. J Food Prot 65:1417–1421
Acknowledgements
This study was partly supported by grants from Consejo Nacional de Investigaciones Científicas y Tecnológicas (CONICET), Agencia Nacional de Promoción Científica y Tecnológica (ANPyCT-FONCYT), and Consejo de Ciencia y Técnica de la Universidad Nacional de Tucumán (CIUNT), Argentina. The authors wish to thank Mr. Oscar Peinado Reviglione for his technical assistance in the HPLC analyses. All experiments comply with the current laws of Argentina.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
LeBlanc, J.G., Garro, M.S. & Savoy de Giori, G. Effect of pH on Lactobacillus fermentum growth, raffinose removal, α-galactosidase activity and fermentation products. Appl Microbiol Biotechnol 65, 119–123 (2004). https://doi.org/10.1007/s00253-003-1532-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-003-1532-z