Abstract
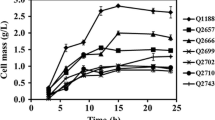
The effect of gene knockout on metabolism in the pflA−, pflB−, pflC−, and pflD− mutants of Escherichia coli was investigated. Batch cultivations of the pfl − mutants and their parent strain were conducted using glucose as a carbon source. It was found that pflA− and pflB− mutants, but not pflC− and pflD− mutants, produced large amounts of d-lactate from glucose under the microaerobic condition, and the maximum yield was 73%. In order to investigate the metabolic regulation mechanism, we measured enzyme activities for the following eight enzymes: glucose 6-phosphate dehydrogenase, 6-phosphogluconate dehydrogenase, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), pyruvate kinase, lactate dehydrogenase (LDH), phosphoenolpyruvate carboxylase, acetate kinase, and alcohol dehydrogenase. Intracellular metabolite concentrations of glucose 6-phosphate, fructose 1,6-bisphosphate, phosphoenolpyruvate, pyruvate, acetyl coenzyme A as well as ATP, ADP, AMP, NADH, and NAD+ were also measured. It was shown that the GAPDH and LDH activities were considerably higher in pflA− and pflB− mutants, which implies coupling between NADH production and consumption between the two corresponding reactions. The urgent energy requirement was shown by the lower ATP/AMP level due to both oxygen limitation and pfl gene knockout, which promoted significant stepping-up of glycolysis when using glucose as a carbon source. It was shown that the demand for energy is more important than intracellular redox balance, thus excess NADH produced through GAPDH resulted in a significantly higher intracellular NADH/NAD+ ratio in pfl − mutants. Consequently, the homolactate production was achieved to meet the requirements of the redox balance and the energy production through glycolysis. The effect of using different carbon sources such as gluconate, pyruvate, fructose, and glycerol was investigated.







Similar content being viewed by others
References
Abdel-Hamid AM, Attwood MM, Guest JR (2001) Pyruvate oxidase contributes to the aerobic growth efficiency of Escherichia coli. Microbiology 147:1483–1498
Alexeeva S, de Kort B, Sawers G, Hellingwere KJ, Teixeira de Mattos MJ (2000) Effects of limited aeration and of the ArcAB system on intermediary pyruvate catabolism in Escherichia coli. J Bacteriol 182:4934–4940
Bergmeyer HU (1984) Methods of enzymatic analysis, vol 6, 3rd edn. VCH, Weinheim, Germany
Bergmeyer HU (1985) Methods of enzymatic analysis, vol 7, 3rd edn. VCH, Weinheim, Germany
Böck A, Sawers G (1996) Fermentation. In: Neidhardt FC, Curtiss R III, Ingraham JL, Lin ECC, Low KB, Magasanik B, Reznikoff WS, Riley M, Schacchter M, Umbarger HE (eds) Escherichia coli and Salmonella cellular and molecular biology, 2nd edn. ASM, Washington D.C.
Boynton ZL, Bennett GN, Rudolph FB, (1994) Intracellular concentrations of coenzyme A and its derivatives from Clostridium acetobutylicum ATCC824 and their roles in enzyme regulation. Appl Environ Microbiol 60:39–44
Brown TDK, Jones-Mortimer MC, Kornberg HL (1977) The enzymic interconversion of acetate and acetyle-coenzyme A in Escherichia coli. J Gen Microbiol 102:327–336
Christel G, Pascal L, Nic DL, Muriel C (1997) Control of the shift from homolactic acid to mixed-acid fermentation in Lactococcus lactis: predominant role of the NADH/NAD+ ratio. J Bacteriol 179:5282–5287
Clark DP (1989) The fermentation pathways of Escherichia coli. FEMS Microbiol Rev 63:223–234
Datsenko KA, Wanner BL (2000) One step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A 97:6640–6645
Drapal N, Sawers G (1995) Promoter 7 of the Escherichia coli pfl operon is a major determinant in the anaerobic regulation of experession by ArcA. J Bacteriol 177:5338–5341
Even S, Garrigues C, Loubiere P, Lindley ND, Cocaign-Bousquet M (1999) Pyruvate metabolism in Lactococcus lactis is dependent upon glyceraldehyde-3-phosphate dehydrogenase activity. Metab Eng 1:198–205
Garrigues C, Loubiere P, Nic D, Lindley ND, Cocaign-Bousquet M (1997) Control of the shift from homolactic acid to mixed-acid fermentation in Lactococcus lactis: Predominant role of the NADH/NAD+ ratio. J Bacteriol 179:5282–5287
Gokarn RR, Altman E, Eiteman MA (1998) Metabolic analysis of Escherichia coli in the presence of and absence of pyruvate carboxylase. Biotechnol Lett 20:795–798
Gokarn RR, Eiteman MA, Altman E (2000) Metabolic analysis of Escherichia coli in the presence and absence of carboxylating enzymes phosphoenolpyruvate carboxylase and pyruvate carboxylase. Appl Environ Microbiol 66:1844–1850
Gunsalus RP (1992) Control of electron flow in Escherichia coli: Coordinated transcription of respiratory pathway genes. J Bacteriol 174:7096–7074
Hong SH, Lee SY (2001) Metabolic flux analysis for succinic acid production by recombinant Escherichia coli with amplified malic enzyme activity. Biotechnol Bioeng 74:89–95
Koebmann BJ, Westerhoff HV, Snoep JL, Nilsson D, Jensen PR (2002) The glycolytic flux in Escherichia coli is controlled by the demand for ATP. J Bacteriol 184:3909–3916
Kumari S, Beatty CM, Browning DF, Busby SJW, Simel EJ, Hovel-Miner G, Wolfe AJ (2000) regulation of acetyl coenzyme A synthetase in Escherichia coli. J Bacteriol 182:4173–4179
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Moritz B, Striegel K, de Graaf AA, Sahm H (2000) Kinetic properties of the glucose-6-phosphate and 6-phosphogluconate dehydrogenase from Corynebacterium glutamicum and their application for predicting pentose phosphate pathway flux in vivo. Eur J Biochem 267:3442–3452
Neves AR, Ramos A, Costa H, van Swam II, Hugenholtz J, Kleerebezem M, de Vos W, Santos H (2002) Effect of different NADH oxidase levels on glucose metabolism by Lactococcus lactis: Kinetics of intracellular metabolite pools determined by in vivo nuclear magnetic resonance. Appl Environ Microbiol 68:6332–6342
Pascal MC, Chippaux M, Abou-Jaoudé A, Blaschkowski HP, Knappe J (1981) Mutants of Escherichia coli K12 with defects in anaerobic pyruvate metabolism. J Gen Microbiol 124:35–42
Peng L, Shimizu K (2003) Global metabolic regulation analysis for Escherichia coli K12 based on protein expressino by 2-dimensional electrophoresis and enzyme activity measurement. Appl Microbiol Biotechnol 61:163–178
Riondet C, Cachon R, WachéY, Alcaraz G, Diviès C (2000) Extracellular oxidoreduction potential modifies carbon and electron flow in Escherichia coli. J Bacteriol 182:620–626
Rödel W, Plaga W, Frank R, Knappe J (1988) Primary structures of Escherichia coli pyruvate formate-lyase and pyruvate-formate-lyase-activating enzyme deduced from the DNA nucleotide sequences. Eur J Biochem 177:153–158
Schaefer U, Boos W, Takors R, Weuster-Botz D (1999) Automated sampling device for monitoring intracellular metabolite dynamics. Anal Biochem 270 88–96
Tseng CP, Albrecht J, Gunsalus RP (1996) Effect of microaerophilic cell growth conditions on expression of the aerobic (cyoABCDE and cydAB) and anaerobic (narGHJI frdABCD and dmsABC) respiratory pathway genes in Escherichia coli. J Bacteriol 178:1094–1098
Vemuri GN, Eiteman MA, Altman E (2002) Effects of growth mode and pyruvate carboxylase on succinic acid production by metabolically engineered strains of Escherichia coli. Appl Environ Microbiol 68:1715–1727
Williamson JR, Corkey BE (1969) Assays of intermediates of the citric acid cycle and related compounds by flourometric enzyme methods In: Lowenstein JM (ed) Citric acid cycle, vol XIII of methods in enzymology. Academic, New York, pp 434–513
Yang YT, Bennett GN, San KY (2001) The effects of feed and intracellular pyruvate levels on the redistribution of metabolic fluxes in Escherichia coli. Metab Eng 3:115–123
Zhou S, Causey TB, Hasona A, Shanmugam KT, Ingram LO (2003) Production of optically pure d-lactic acid in mineral salts medium by metabolically engineered Escherichia coli W3110. Appl Environ Microbiol 69:399–407
Acknowledgements
This work was supported in part by a grant from New Energy and Industrial Technology Development Organization of the Ministry of Economy, Trade, and Industry of Japan (Development of Technological Infrastructure for Industrial Bioprocess Project). It is acknowledged that the mutant strains were kindly provided by Drs. T. Baba and H. Mori of Keio University. It is also acknowledged that Ms. Mika Tanaka of Kyushu Institute of Technology found the interesting fermentation phenomenon for the case of pflB− mutant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, J., Shimizu, K. The effect of pfl gene knockout on the metabolism for optically pure d-lactate production by Escherichia coli . Appl Microbiol Biotechnol 64, 367–375 (2004). https://doi.org/10.1007/s00253-003-1499-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-003-1499-9