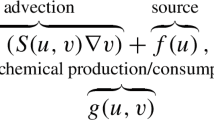Abstract
Leading edge protrusion is one of the critical events in the cell motility cycle and it is believed to be driven by the assembly of the actin network. The concept of dendritic nucleation of actin filaments provides a basis for understanding the organization and dynamics of the actin network at the molecular level. At a larger scale, the dynamic geometry of the cell edge has been described in terms of the graded radial extension model, but this level of description has not yet been linked to the molecular dynamics. Here, we measure the graded distribution of actin filament density along the leading edge of fish epidermal keratocytes. We develop a mathematical model relating dendritic nucleation to the long-range actin distribution and the shape of the leading edge. In this model, a steady-state graded actin distribution evolves as a result of branching, growth and capping of actin filaments in a finite area of the leading edge. We model the shape of the leading edge as a product of the extension of the actin network, which depends on actin filament density. The feedback between the actin density and edge shape in the model results in a cell shape and an actin distribution similar to those experimentally observed. Thus, we explain the stability of the keratocyte shape in terms of the self-organization of the branching actin network.









Similar content being viewed by others
References
Abercrombie M (1980) The crawling movement of metazoan cells. Proc R Soc Lond (Biol) 207:129–147
Abraham VC, Krishnamurthi V, Taylor DL, Lanni F (1999) The actin-based nanomachine at the leading edge of migrating cells, Biophys J 77:1721–1732
Anderson KI, Cross R (2000) Contact dynamics during keratocyte motility. Curr Biol 10:253–260
Bernheim-Groswasser A, Wiesner S, Golsteyn RM, Carlier MF, Sykes S (2002) The dynamics of actin-based motility depend an surface parameters. Nature 417:308–311
Borisy GG, Svitkina TM (2000) Actin machinery: pushing the envelope. Curr Opin Cell Biol 12:104–112
Bottino D, Mogilner A, Roberts T, Stewart M, Oster G (2002) How nematode sperm crawl. J Cell Sci 115:367–384
Bray D (2001) Cell movements. Garland, New York
Cameron LA, Svitkina TM, Vignjevic D, Theriot JA, Borisy GG (2001) Dendritic organization of actin comet tails. Curr Biol 11:130–135
Carlier MF, Pantaloni D (1997) Control of actin dynamics in cell motility. J Mol Biol 269:459–467
Carlsson AE (2001) Growth of branched actin networks against obstacles. Biophys J 81:1907–1923
Condeelis J (1993) Life at the leading edge: the formation of cell protrusions. Annu Rev Cell Biol 9:411–444
Cortese JD, Schwab B, Frieden C, Elson EL (1989) Actin polymerization induces shape change in actin-containing vesicles. Proc Natl Acad Sci USA 86:5773–5777
Dai JW, Sheetz MP (1999) Membrane tether formation from blebbing cells. Biophys J 77:3363–3370
Edelstein-Keshet L, Ermentrout GB (2000) Models for spatial polymerization dynamics of rod-like polymers. J Math Biol 40:64–96
Evans E, Skalak R (1980) Mechanics and thermodynamics of biomembranes. CRC Press, Boca Raton, Fla., USA
Garcia AI (1994) Numerical methods for physicists. Prentice-Hall, Englewood Cliffs, NJ, USA
Gerbal F, Chaikin P, Rabbin Y, Prost J (2000) Elastic model of Listeria propulsion. Biophys J 79:2259–2275
Hartwig JH, Bokoch GM, Carpenter CL, Janmey PA, Taylor LA, Toker A, Stossel TP (1995) Thrombin receptor ligation and activated rac uncap actin filament barbed ends through phosphoinositide synthesis in permeabilized human platelets. Cell 82:643–643
He H, Dembo M (1997) On the mechanics of the first cleavage division of the sea urchin egg. Exp Cell Res 233:252–273
Huang M, Yang C, Schafer DA, Cooper JA, Higgs HN, Zigmond SH (1999) Cdc42-induced actin filaments are protected from capping protein. Curr Biol 9:979–982
Janmey PA, Hvidt S, Kas J, Lerche D, Maggs A, Sackmann E, Schliwa M, Stossel TP (1994) The mechanical properties of actin gels, elastic modulus and filament motions. J Biol Chem 269:32503–32513
Lee J, Ishiara A, Jacobson K (1993a) The fish epidermal keratocyte as a model system for the study of cell locomotion. In: Jones G, Wigley C, Warn R (eds) Cell behavior: adhesion and motility. Company of Biologists, Cambridge, UK, pp 73–89
Lee J, Ishihara A, Theriot J, Jacobson K (1993b) Principles of locomotion for simple-shaped cells. Nature 362:467–471
Logan JD (1997) Applied mathematics. Wiley, New York
Loisel TP, Boujemaa R, Pantaloni D, Carlier MF (1999) Reconstitution of actin-based motility of listeria and shigella using pure proteins. Nature 401:613–616
Maly IV, Borisy GG (2001) Self-organization of a propulsive actin network as an evolutionary process. Proc Natl Acad Sci USA 98:11324–11329
McGrath JL, Osborn EA, Tardy YS, Dewey CF, Hartwig JH (2000) Regulation of the actin cycle in vivo by actin filament severing. Proc Natl Acad Sci USA 97:6532–6537
Mitchison TJ, Cramer LP (1996) Actin-based cell motility and cell locomotion. Cell 84:371–379
Miyata H, Nishiyama S, Akashi KI, Kinosita K (1999) Protrusive growth from giant liposomes driven by actin polymerization. Proc Natl Acad Sci USA 96:2048–2053
Mogilner A, Edelstein-Keshet L (2002) Regulation of actin dynamics in rapidly moving cells: a quantitative analysis. Biophys J 83:1237–1258
Mogilner A, Oster G (1996) Cell motility driven by actin polymerization. Biophys J 71:3030–3045
Pantaloni D, Clainche CL, Carlier MF (2001) Mechanism of actin-based motility. Science 292:1502–1506
Pollard TD, Blanchoin L, Mullins RD (2000) Molecular mechanisms controlling actin filament dynamics in nonmuscle cells. Annu Rev Biophys Biomol Struct 29:545–576
Rinnerthaler G, Herzog M, Klappacher M, Kunka H, Small JV (1991) Leading edge movement and ultrastructure in mouse macrophages. J Struct Biol 106:1–16
Rottner K, Behrendt B, Small JV, Wehland J (1999) Vasp dynamics during lamellipodia protrusion. Nat Cell Biol 1:321–322
Schafer DA, Jennings PB, Cooper JA (1996) Dynamics of capping protein and actin assembly in vitro: uncapping barbed ends by polyphosphoinositides. J Cell Biol 135:169–179
Small JV (1994) Lamellipodia architecture: actin filament turnover and the lateral flow of actin filaments during motility. Semin Cell Biol 5:157–163
Small JV, Herzog M, Anderson K (1995) Actin filament organization in the fish keratocyte lamellipodium. J Cell Biol 129:1275–1286
Svitkina TM, Borisy GG (1999) Arp2/3 complex and actin depolymerizing factor/cofilin in dendritic organization and treadmilling of actin filament array in lamellipodia. J Cell Biol 145:1009–1026
Svitkina TM, Verhovsky AB, McQuade KM, Borisy GG (1997) Analysis of the actin-myosin 11 system in fish epidermal keratocytes: mechanism of cell body translocation. J Cell Biol 139:397–415
Theriot JA, Mitchison TJ (1991) Actin microfilament dynamics in locomoting cells. Nature 352:126–131
Welch MD, Mallavarapu A, Rosenblatt J, Mitchison TJ (1997) Actin dynamics in vivo. Curr Opin Cell Biol 9:54–61
Acknowledgements
The work is supported by Swiss Science Foundation grant 31-61589 to H.P.G., A.B.V., and J.J.M. A.M. is supported by a UCD Chancellor's Fellowship, a NSF grant and a NIH GLUE grant "Cell Migration Consortium". We are grateful to Gary G. Borisy, Tatyana Svitkina, Gaudenz Danuser, Eric Cytrynbaum, and Angela Gallegos for fruitful discussions.
Author information
Authors and Affiliations
Corresponding author
Appendix: asymptotic analysis of the model equations
Appendix: asymptotic analysis of the model equations
Scaling and non-dimensionalization
Introducing the non-dimensionalized variables x/L, γt and p/(β/γ), we can write the model equations in the case of the steady locomotion in the form:
Here, for simplicity, we keep the same notations for non-dimensional variables. In what follows, we investigate the steady-state solutions of these equations, i.e. when the time derivative is equal to zero. We also concentrate on the case of the flat leading edge, f"(x)=0. In this last case, without loss of generality we assume that v ±=1, which can be justified by re-scaling the protrusion velocity (f"(x)=0).
Slow rate of capping
In this case (γ≪V/L, ε≫1), the scale of the actin density is defined by the ratio of the branching rate β over the effective rate of fiber disappearance due to the lateral flow, V/L. In the non-dimensional system this means that the scale of actin density is ~1/ε. Substituting this new scale into Eqs. (12) and (13) and dropping the negligible capping term (the third term on the right-hand side), we obtain the following steady-state equations:
Let us introduce the function s=p +−p –. Adding and subtracting Eqs. (15), we obtain the equations:
for the local model. The solutions of these equations can be found easily by first solving the first equation of (16), and then the second one, and by using the conditions that s(x) is an odd function, p(x) is an even function, and p(1)=s(1):
The dimensional form of these solutions is given by Eq. (8). For the global model, adding and subtracting Eqs. (15), we obtain:
Differentiating these equations a second time, we find:
Using symmetry properties of functions s(x) and p(x), we have:
Using the condition p(1)=s(1), we find that 1/P=π/4. Finally, integrating p(x) from −1 to 1, we obtain the value of A:
The dimensional form of these solutions is given by Eq. (9).
Fast rate of capping
In this case (γ≫V/L, ε≪1), singular perturbation theory can be used to investigate the local model (Eq. 12). Simple analysis shows that away from two boundary layers of width O(ε) at the spatial domain's edges, the constant "outer" solution, p ±(x)=0.5, p(x)=1, gives the leading order approximation. Near the boundaries, re-scaling the spatial variable, x→εx, leads to a non-linear system, which cannot be solved analytically. A qualitative analysis shows that the corresponding "inner" solution for p(x) decreases from p=1 inside the spatial interval to p=O(ε) at the boundaries.
Unlike the local model, the global model (Eq. 13) is not characterized by the boundary layers and has an approximate analytical solution in this limit. Adding and subtracting Eqs. (13), we find:
Differentiating these equations a second time, we derive:
Solving these equations and using symmetry properties of functions s(x) and p(x) gives:
Substituting these solutions into Eq. (22) and using the condition p(1)=s(1), we find that \( \bar P \approx 1 - (\pi ^2 \varepsilon ^2 /8) \), κ≈(πε/2), A≈π/4 and B≈π2ε/16. Thus, the non-dimensional approximate solution of the local model in this limit is p=(π/4)cos(πx/2). The dimensional form of the solutions of the global and local models in the fast capping rates limit are given by Eq. (10).
Rights and permissions
About this article
Cite this article
Grimm, H.P., Verkhovsky, A.B., Mogilner, A. et al. Analysis of actin dynamics at the leading edge of crawling cells: implications for the shape of keratocyte lamellipodia. Eur Biophys J 32, 563–577 (2003). https://doi.org/10.1007/s00249-003-0300-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-003-0300-4