Abstract
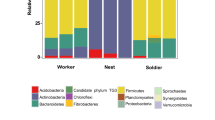
Termites are intimately tied to the microbial world, as they utilize their gut microbiome for the conversion of plant cellulose into necessary nutrients. Subterranean termites must also protect themselves from the vast diversity of harmful microbes found in soil. However, not all soil microbes are harmful, such as Streptomyces and methanotrophic bacteria that some species of termites harbor in complex nest structures made of fecal material. The eastern subterranean termite, Reticulitermes flavipes, has a simple nest structure consisting of fecal lined galleries. We tested the hypothesis that R. flavipes maintains a select microbial community in its nests to limit the penetration of harmful soilborne pathogens and favor the growth of beneficial microbes. Using Illumina sequencing, we characterized the bacterial and fungal communities in the surrounding soil, in the nest galleries, and on the cuticle of workers. We found that the galleries provide a more beneficial microbial community than the surrounding soil. Bacterial and fungal diversity was highest in the soil, lower in the galleries, and least on the cuticle. Bacterial communities clustered together according to the substrate from which they were sampled, but this clustering was less clear in fungal communities. Most of the identified bacterial and fungal taxa were unique to one substrate, but the soil and gallery communities had very similar phylum-level taxonomic profiles. Notably, the galleries of R. flavipes also harbored both the potentially beneficial Streptomyces and the methanotrophic Methylacidiphilales, indicating that these microbial associations in fecal material pre-date the emergence of complex fecal nest structures. Surprisingly, several pathogenic groups were relatively abundant in the galleries and on the cuticle, suggesting that pathogens may accumulate within termite nests over time while putatively remaining at enzootic level during the lifetime of the colony.





Similar content being viewed by others
Data Availability
Metadata and raw reads of 16S rRNA gene and ITS gene amplicons have been deposited in the Open Science Framework database, https://osf.io (https://doi.org/10.17605/OSF.IO/4CSN2).
References
Wilson EO (1990) Success and dominance in ecosystems: the case of the social insects. In: Kinne O (ed) Excellence in ecology, vol 2. Ecology Institute, Oldendorf/Luhe, Federal Republic of Germany
Keller S, Zimmermann G, Wilding N, Collins N, Hammond P, Webber J (1989) Mycopathogens of soil insects. Insect-Fungus Interact. 239–270
Oi DH, Pereira RM (1993) Ant behavior and microbial pathogens (Hymenoptera: Formicidae). Fla. Entomol. 76:63–74
Schmid-Hempel P (1998) Parasites in social insects. Princeton University Press, Princeton
Vega FE, Goettel MS, Blackwell M, Chandler D, Jackson MA, Keller S, Koike M, Maniania NK, Monzon A, Ownley BH (2009) Fungal entomopathogens: new insights on their ecology. Fungal Ecol. 2(4):149–159
Chouvenc T, Su N-Y, Grace JK (2011) Fifty years of attempted biological control of termites–analysis of a failure. Biol. Control 59(2):69–82
Evans HC, Elliot SL, Hughes DP (2011) Hidden diversity behind the zombie-ant fungus Ophiocordyceps unilateralis: four new species described from carpenter ants in Minas Gerais, Brazil. PLoS One 6(3):e17024
Hughes DP, Andersen SB, Hywel-Jones NL, Himaman W, Billen J, Boomsma JJ (2011) Behavioral mechanisms and morphological symptoms of zombie ants dying from fungal infection. BMC Ecol. 11(1):13
Bignell DE (2006) Termites as soil engineers and soil processors. In: König HVA (ed) Intestinal microorganisms of termites and other invertebrates. Springer, Berlin Heidelberg, pp 183–220
Jouquet P, Dauber J, Lagerlöf J, Lavelle P, Lepage M (2006) Soil invertebrates as ecosystem engineers: intended and accidental effects on soil and feedback loops. Appl. Soil Ecol. 32(2):153–164
Chouvenc T, Elliott ML, Su N-Y (2011) Rich microbial community associated with the nest material of Reticulitermes flavipes (Isoptera: Rhinotermitidae). Fla. Entomol. 94(1):115–116
Chouvenc T, Bardunias P, Efstathion CA, Chakrabarti S, Elliott ML, Giblin-Davis R, Su N-Y (2013) Resource opportunities from the nest of dying subterranean termite (Isoptera: Rhinotermitidae) colonies: a laboratory case of ecological succession. Ann. Entomol. Soc. Am. 106(6):771–778
Cremer S, Armitage SA, Schmid-Hempel P (2007) Social immunity. Curr. Biol. 17(16):R693–R702
Cremer S, Pull CD, Fuerst MA (2018) Social immunity: emergence and evolution of colony-level disease protection. Annu. Rev. Entomol. 63:105–123
Liu L, Zhao X-Y, Tang Q-B, Lei C-L, Huang Q-Y (2019) The mechanisms of social immunity against fungal infections in eusocial insects. Toxins 11(5):244
Brown Jr WL (1968) An hypothesis concerning the function of the metapleural glands in ants. Am. Nat. 102(924):188–191
Hölldobler B, Engel-Siegel H (1984) On the metapleural gland of ants. Psyche: A Journal of Entomology 91(3–4):201–224
Ortius-Lechner D, Maile R, Morgan ED, Boomsma JJ (2000) Metapleural gland secretion of the leaf-cutter ant Acromyrmex octospinosus: new compounds and their functional significance. J. Chem. Ecol. 26(7):1667–1683
Rosengaus RB, Guldin MR, Traniello JF (1998) Inhibitory effect of termite fecal pellets on fungal spore germination. J. Chem. Ecol. 24(10):1697–1706
Chouvenc T, Efstathion CA, Elliott ML (1770) Su N-Y (2013) extended disease resistance emerging from the faecal nest of a subterranean termite. Proc. R. Soc. B Biol. Sci. 280:20131885
Bulmer MS, Bachelet I, Raman R, Rosengaus RB, Sasisekharan R (2009) Targeting an antimicrobial effector function in insect immunity as a pest control strategy. Proc. Natl. Acad. Sci. U. S. A. 106(31):12652–12657
Bulmer MS, Crozier RH (2004) Duplication and diversifying selection among termite antifungal peptides. Mol. Biol. Evol. 21(12):2256–2264
Hamilton C, Lay F, Bulmer MS (2011) Subterranean termite prophylactic secretions and external antifungal defenses. J. Insect Physiol. 57(9):1259–1266
Lamberty M, Zachary D, Lanot R, Bordereau C, Robert A, Hoffmann JA, Bulet P (2001) Insect immunity. Constitutive expression of a cysteine-rich antifungal and a linear antibacterial peptide in a termite insect. J. Biol. Chem. 276:4085–4092
Higashi M, Abe T, Burns TP (1992) Carbon—nitrogen balance and termite ecology. Proc. R. Soc. B Biol. Sci. 249(1326):303–308
Eggleton P (2010) An introduction to termites: biology, taxonomy and functional morphology. In: Bignell DE, Roisin Y, Lo N (eds) Biology of termites: a modern synthesis. Springer, Berlin, pp 1–26
Vargo EL (2003) Hierarchical analysis of colony and population genetic structure of the eastern subterranean termite, Reticulitermes flavipes, using two classes of molecular markers. Evol 57(12):2805–2818
DeHeer CJ, Vargo EL (2004) Colony genetic organization and colony fusion in the termite Reticulitermes flavipes as revealed by foraging patterns over time and space. Mol. Ecol. 13(2):431–441
DeHeer C, Kutnik M, Vargo E, Bagneres A (2005) The breeding system and population structure of the termite Reticulitermes grassei in Southwestern France. Heredity 95(5):408–415
Kozich JJ, Westcott SL, Baxter NT, Highlander SK, Schloss PD (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 79(17):5112–5120
White T, Bruns T, Lee S, Taylor J, Innis M, Gelfand D, Sninsky J (1990) PCR protocols: a guide to methods and applications.
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet C, Al-Ghalith GA, Alexander H, Alm EJ, Arumugam M, Asnicar F (2018) QIIME 2: reproducible, interactive, scalable, and extensible microbiome data science. PeerJ Prepr,
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJA, Holmes SP (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat. Methods 13(7):581–583
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30(4):772–780
Hamady M, Lozupone C, Knight R (2010) Fast UniFrac: facilitating high-throughput phylogenetic analyses of microbial communities including analysis of pyrosequencing and PhyloChip data. ISME J 4(1):17–27
Pruesse E, Quast C, Knittel K, Fuchs BM, Ludwig W, Peplies J, Glöckner FO (2007) SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 35(21):7188–7196
Kõljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AF, Bahram M, Bates ST, Bruns TD, Bengtsson-Palme J, Callaghan TM (2013) Towards a unified paradigm for sequence-based identification of fungi. Mol. Ecol. 22(21):5271–5277
Sung G-H, Hywel-Jones NL, Sung J-M, Luangsa-ard JJ, Shrestha B, Spatafora JW (2007) Phylogenetic classification of Cordyceps and the clavicipitaceous fungi. Stud. Mycol. 57:5–59
Chouvenc T, Su NY, Robert A (2009) Inhibition of Metarhizium anisopliae in the alimentary tract of the eastern subterranean termite Reticulitermes flavipes. J. Invertebr. Pathol. 101:130–136
Aguero CM, Eyer PA, Vargo EL (2020) Increased genetic diversity from colony merging in termites does not improve survival against a fungal pathogen. Sci. Rep. 10:1–9
Brune A (2014) Symbiotic digestion of lignocellulose in termite guts. Nat. Rev. Microbiol. 12(3):168–180
Brune A, Dietrich C (2015) The gut microbiota of termites: digesting the diversity in the light of ecology and evolution. Annu. Rev. Microbiol. 69:145–166
Rouland-Lefèvre C (2000) Symbiosis with fungi. In: Takuya ABD, Higashi M (eds) Termites: evolution, sociality, symbioses, ecology. Kluwer Academic Publishers, Dordrecht, pp 289–306
Nobre T, Rouland-Lefèvre C, Aanen DK (2010) Comparative biology of fungus cultivation in termites and ants. In: Bignell DE, Roisin Y, Lo N (eds) Biology of termites: a modern synthesis. Springer-Verlag, Berlin, pp 193–210
Waksman SA, Lechevalier HA (1962) The Actinomycetes. Vol. III. Antibiotics of Actinomycetes
Goodfellow M, Williams S (1983) Ecology of Actinomycetes. Annu. Rev. Microbiol. 37(1):189–216
Kaltenpoth M (2009) Actinobacteria as mutualists: general healthcare for insects? Trends Microbiol. 17(12):529–535
Visser AA, Nobre T, Currie CR, Aanen DK, Poulsen M (2012) Exploring the potential for Actinobacteria as defensive symbionts in fungus-growing termites. Microb. Ecol. 63(4):975–985
Benndorf R, Guo H, Sommerwerk E, Weigel C, Garcia-Altares M, Martin K, Hu H, Küfner M, de Beer Z, Poulsen M (2018) Natural products from Actinobacteria associated with fungus-growing termites. Antibiotics 7(3):83
Currie CR, Scott JA, Summerbell RC, Malloch D (1999) Fungus-growing ants use antibiotic-producing bacteria to control garden parasites. Nature 398(6729):701–704
Little AE, Currie CR (2007) Symbiotic complexity: discovery of a fifth symbiont in the attine ant–microbe symbiosis. Biol. Lett. 3(5):501–504
Aanen DK, Eggleton P (2017) Symbiogenesis: beyond the endosymbiosis theory? J. Theor. Biol. 434:99–103
Wood T (1988) Termites and the soil environment. Biol. Fertil. Soils 6(3):228–236
Arango A, Carlson CM, Currie CR, McDonald BR, Book AJ, Green F, Lebow NK, Raffa KF (2016) Antimicrobial activity of actinobacteria isolated from the guts of subterranean termites. Environ. Entomol. 45:1415–1423
Chouvenc T, Elliott ML, Šobotník J, Efstathion CA, Su N-Y (2018) The termite fecal nest: a framework for the opportunistic acquisition of beneficial soil Streptomyces (Actinomycetales: Streptomycetaceae). Environ. Entomol. 47:1431–1439
Sanderson M (1996) Biomass of termites and their emissions of methane and carbon dioxide: a global database. Glob. Biogeochem. Cycles 10(4):543–557
Kirschke S, Bousquet P, Ciais P, Saunois M, Canadell JG, Dlugokencky EJ, Bergamaschi P, Bergmann D, Blake DR, Bruhwiler L (2013) Three decades of global methane sources and sinks. Nat. Geosci. 6(10):813–823
Brune A (2018) Methanogens in the digestive tract of termites. In: JHP H (ed) (Endo) symbiotic methanogenic Archaea. Microbiology monographs 19. Springer, Berlin Heidelberg, pp. 81–101
Nauer PA, Hutley LB, Arndt SK (2018) Termite mounds mitigate half of termite methane emissions. Proc. Natl. Acad. Sci. 115(52):13306–13311 201809790
Bourguignon T, Lo N, Šobotník J, Ho SY, Iqbal N, Coissac E, Lee M, Jendryka MM, Sillam-Dussès D, Krížková B, Roisin Y, Evans TA (2017) Mitochondrial phylogenomics resolves the global spread of higher termites, ecosystem engineers of the tropics. Mol. Biol. Evol. 34(3):589–597
Bucek A, Šobotník J, He S, Shi M, Mc Mahon DP, Holmes EC, Roisin Y, Lo N, Bourguignon T (2019) Evolution of termite symbiosis informed by transcriptome-based phylogenies. Curr. Biol. 29(21):3728–3734
Loreto R, Hughes DP (2016) Disease dynamics in ants: a critical review of the ecological relevance of using generalist fungi to study infections in insect societies. In: advances in genetics, vol 94. Elsevier, pp 287-306
Rosengaus RB, Maxmen AB, Coates LE, Traniello JF (1998) Disease resistance: a benefit of sociality in the dampwood termite Zootermopsis angusticollis (Isoptera: Termopsidae). Behav. Ecol. Sociobiol. 44(2):125–134
Chouvenc T, Efstathion CA, Elliott ML, Su N-Y (2012) Resource competition between two fungal parasites in subterranean termites. Naturwissenschaften 99(11):949–958
Myles TG (2002) Observations on mites Acari associated with the eastern subterranean termite, Reticulitermes flavipes Isoptera: Rhinotermitidae. Sociobiology 40(2):277–280
Wang C, Powell JE, O'Connor BM (2002) Mites and nematodes associated with three subterranean termite species (Isoptera: Rhinotermitidae). Fla. Entomol. 85(3):499–506
Matsuura K (2006) Termite-egg mimicry by a sclerotium-forming fungus. Proc. R. Soc. B Biol. Sci. 273(1591):1203–1209
Mitaka Y, Mori N, Matsuura K (2018) A termite fungistatic compound, mellein, inhibits entomopathogenic fungi but not egg-mimicking termite ball fungi. Appl. Entomol. Zool. 54:39–46 1–8
Yashiro T, Matsuura K (2007) Distribution and phylogenetic analysis of termite egg-mimicking fungi “termite balls” in Reticulitermes termites. Ann. Entomol. Soc. Am. 100(4):532–538
Matsuura K, Tanaka C, Nishida T (2000) Symbiosis of a termite and a sclerotium-forming fungus: sclerotia mimic termite eggs. Ecol. Res. 15(4):405–414
Hughes DP, Pierce NE, Boomsma JJ (2008) Social insect symbionts: evolution in homeostatic fortresses. Trends Ecol. Evol. 23(12):672–677
Acknowledgments
We thank Alan Byboth and the Sam Houston State University Center for Biological Field Studies for providing us access to their site to collect termite and soil samples.
Funding
This research was supported by the Urban Entomology Endowment at Texas A&M University.
Author information
Authors and Affiliations
Contributions
CMA, PAE, TLC, and ELV designed the study. CMA collected and analyzed the data. CMA, PAE, TLC, and ELV wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Supplementary Information
ESM 1
(PDF 926 kb)
Rights and permissions
About this article
Cite this article
Aguero, C.M., Eyer, PA., Crippen, T.L. et al. Reduced Environmental Microbial Diversity on the Cuticle and in the Galleries of a Subterranean Termite Compared to Surrounding Soil. Microb Ecol 81, 1054–1063 (2021). https://doi.org/10.1007/s00248-020-01664-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-020-01664-w