Abstract
Microorganisms are very sensitive to environmental change and can be used to gauge anthropogenic impacts and even predict restoration success of degraded environments. Here, we report assessment of bauxite mining activities on soil biogeochemistry and microbial community structure using un-mined and three post-mined sites in Jamaica. The post-mined soils represent a chronosequence, undergoing restoration since 1987, 1997, and 2007. Soils were collected during dry and wet seasons and analyzed for pH, organic matter (OM), total carbon (TC), nitrogen (TN), and phosphorus. The microbial community structure was assessed through quantitative PCR and massively parallel bacterial ribosomal RNA (rRNA) gene sequencing. Edaphic factors and microbial community composition were analyzed using multivariate statistical approaches and revealed a significant, negative impact of mining on soil that persisted even after greater than 20 years of restoration. Seasonal fluctuations contributed to variation in measured soil properties and community composition, but they were minor in comparison to long-term effects of mining. In both seasons, post-mined soils were higher in pH but OM, TC, and TN decreased. Bacterial rRNA gene analyses demonstrated a general decrease in diversity in post-mined soils and up to a 3-log decrease in rRNA gene abundance. Community composition analyses demonstrated that bacteria from the Proteobacteria (α, β, γ, δ), Acidobacteria, and Firmicutes were abundant in all soils. The abundance of Firmicutes was elevated in newer post-mined soils relative to the un-mined soil, and this contrasted a decrease, relative to un-mined soils, in proteobacterial and acidobacterial rRNA gene abundances. Our study indicates long-lasting impacts of mining activities to soil biogeochemical and microbial properties with impending loss in soil productivity.




Similar content being viewed by others
References
Ager D, Evans S, Li H, Lilley AK, van der Gast CK (2009) Anthropogenic disturbance affects the structure of bacterial communities. Environ Microbiol 12:670–678
Andersen JM (1976) An ignition method for determination of total phosphorus in lake sediments. Water Res 10:329–331
Banning NC, Gleeson DB, Grigg AH, Grant CD, Andersen GL, Brodie EL, Murphy DV (2011) Soil microbial community successional patterns during forest ecosystem restoration. Appl Environ Microbiol 77(17):6158–6164
Bray JR, Curtis JT (1957) An ordination of the upland forest communities of southern Wisconsin. Ecological Monographs 27:325–349
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods. doi:10.1038/nmeth.f.303
Castro HF, Classen AT, Austin EE, Norby RJ, Schadt CW (2010) Soil microbial community responses to multiple experimental climate change drivers. Appl Environ Microbiol 76:999–1007
Chambers JC, Wade GL (1990) Evaluating reclamation success: the ecological consideration. General Technical Report NE-164. U.S. Department of Agriculture, Forest Service, Rodnor, PA
Chao A (1984) Nonparametric estimation of the number of classes in a population. Scandinavian J Stat 11:265–270
Chauhan A, Cherrier J, Williams HN (2009) Impact of sideways and bottom up control factors on bacterial community succession over a tidal cycle. Proc Natl Acad Sci USA 106:4301–4306
Chauhan A, Ogram A, Reddy KR (2006) Syntrophic archaeal associations differ with nutrient impact in a freshwater marsh. J Appl Microbiol 100:73–84
Chauhan A, Ogram A, Reddy KR (2004) Syntrophic-methanogenic associations along a nutrient gradient in the Florida Everglades. Appl Environ Microbiol 70:3475–3484
Cleveland CC, Nemergut DR, Schmidt SK, Townsend AR (2007) Increases in soil respiration following labile carbon additions linked to rapid shifts in soil microbial community composition. Biogeochemistry 82:229–240
Coke LB, Weir CC, Hill VG (1987) Environmental impact of bauxite mining and processing in Jamaica. Soc and Econ Stud 36:289–333
DeSantis TZ, Hugenholtz P, Larsen N, Rojas M, Brodie EL, Keller K, Huber T, Dalevi D, Hu P, Andersen GL (2006) Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72:5069–5072
Dowd SE, Sun Y et al (2008) Survey of bacterial diversity in chronic wounds using pyrosequencing, DGGE, and full ribosome shotgun sequencing. BMC Microbiol 8:43
Dunbar J, Takala S, Barns SM, Davis JA, Kuske CR (1999) Levels of bacterial community diversity in four arid soils compared by cultivation and 16S rRNA gene cloning. Appl Environ Microbiol 65:1662–1669
Dyck WJ, Cole DW (1994) Strategies for determining consequences of harvesting and associated practices on long-term productivity. In: Dyck WJ, Cole DW (eds) Impacts of forest harvesting on long-term site productivity. Chapman & Hall, New York, pp 11–40
Fromin N, Hamelin J, Tarnawski S, Roesti D, Jourdain-Miserez K, Forestier N et al (2002) Statistical analysis of denaturing gel electrophoresis (DGE) fingerprinting patterns. Environ Microbiol 4:634–643
Gardner JH, Bell DT (2007) Bauxite mining restoration by Alcoa World Alumina Australia in Western Australia: social, political, historical, and environmental contexts. Restoration Ecology 15:S3–S10
Gihring TM, Green SJ, Schadt CW (2011) Massively parallel rRNA gene sequencing exacerbates the potential for biased community diversity comparisons due to variable library sizes. doi:10.1111/j.1462-2920.2011.02550.x
Greenberg WA, Wilding LP (2007) Pre- and post-mined bauxite soils of Jamaica: physical and chemical properties. Soil Sci Soc Am J 71:181–188
Griffiths BS, Bonkowski M, Roy J, Ritz K (2001) Functional stability, substrate utilization and biological indicators of soils following environmental impacts. Appl Soil Ecol 16:49–61
Griffiths BS, Hallett PD, Kuan HL, Gregory AS, Watts CW, Whitmore AP (2008) Functional resilience of soil microbial communities depends on both soil structure and microbial community composition. Biol Fertil Soils 44:745–754
Hanke S, Schuler K (1995) Alternative monetary regimes for Jamaica. Study for the Private Sector Organization of Jamaica (PSOJ)
Harris J (2009) Soil microbial communities and restoration ecology: facilitators or followers? Science 325:573–574
Hartman WH, Richardson CJ, Vilgalys R, Bruland GL (2008) Environmental and anthropogenic controls over bacterial communities in wetland soils. PNAS 105:17842–17847
Hogg BM (1966) Yield decline on sugar cane on part of Worthy Park Estate. Proceedings of the Jamaica Association of Sugarcane Technologists
Jasper DA (2007) Beneficial soil microorganisms of the Jarrah forest and their recovery in bauxite Southwestern Australia. Restoration Ecology 15:S74–S84
Kersters K, De Vos P, Gillis M, Swings J, Vandamme P, Stackebrandt E (2006) Introduction to the Proteobacteria. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The prokaryotes: a handbook on the biology of bacteria, vol 5, 3rd edn. Springer, New York, pp 3–37
Kovach WL (1999) MVSP—a multivariate statistical package for windows, version 3.0, Kovach Computing Services, Pentraeth, Wales, UK
Kuske CR, Barns SM, Bush JD (1997) Diverse uncultivated bacterial groups from soils of the arid Southwestern United States that are present in many geographic regions. Appl Environ Microbiol 63:3614–3621
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, New York
Lewis D, White JR, Wafula D, Athar R, Williams HN, Chauhan A (2010) Soil functional diversity analysis of a bauxite mined restoration chronosequence. Microb Ecol 59:710–723
Lozupone C, Knight R (2005) UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71:8228–8235
Magurran AE, Phillip DAT (2001) Implications of species loss in freshwater fish assemblages. Ecography 24:645–650
McCaig AE, Glover LA, Prosser JI (1999) Molecular analysis of bacterial community structure and diversity in unimproved and improved upland grass pastures. Appl Environ Microbiol 65:1721–1730
Morgan GW (1974) Crop productivity as affected by depth of topsoil spread for reclaiming bauxite mined lands in Jamaica. Tropical Agric 51:332–346
Mummey DL, Stahl PD, Buyer JS (2002) Soil microbiological properties 20 years after surface mine reclamation: spatial analysis of rehabilitated and undisturbed sites. Soil Biol Biochem 34:1717–1725
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Nadkarni MA, Martin EF, Jacques NA, Hunter N (2002) Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiology 148:257–266
Pielou EC (1975) Ecological diversity. Wiley, New York
Quaiser A, Ochsenreiter T, Lanz C, Schuster SC, Treusch AH, Eck J, Schleper C (2003) Acidobacteria form a coherent but highly diverse group within the bacterial domain: evidence from environmental genomics. Mol Microbiol 50:563–575
Quirino BF, Pappas GJ, Tagliaferro AC, Collevatti RG, Neto EL, da Silva MRSS, Bustamante MMC, Krüger RH (2009) Molecular phylogenetic diversity of bacteria associated with soil of the savanna-like Cerrado vegetation. Microbiol Res 164:59–70
Sait M, Davis KER, Janssen PH (2006) Effect of pH on isolation and distribution of members of subdivision 1 of the phylum Acidobacteria occurring in soil. Appl Environ Microbiol 72:1852–1857
Scholten JJ, Andriesse W (1986) Morphology, genesis and classification of three soils over limestone. Jamaica Geoderma 39:1–40
Shannon CE, Weaver W (1949) In: The mathematical theory of communication. University of Illinois Press, Urbana
Spain AM, Krumholz LR, Elshahed MS (2009) Abundance, composition, diversity and novelty of soil Proteobacteria. ISME J 3:992–1000
Spain AV, Hinz DA, Ludwig JA, Tibbett M, Tongway DJ (2006) Mine closure and ecosystem development—Alcan Gove bauxite mine. Northern Territory, Australia, ISBN 978-0-9756756-6-3. Proceedings of the first international seminar on mine closure 2006 In: First international seminar on mine closure, Perth, Western Australia, Australia, 13-15 September 2006
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Tarlera S, Jangid K, Ivester AH, Whitman WB, Williams MA (2008) Microbial community succession and bacterial diversity in soils during 77 000 years of ecosystem development. FEMS Microbiol Ecol 64:129–140
Tibbett M (2009) Large-scale mine site restoration of Australian eucalypt forests after bauxite mining: soil management and ecosystem development. In: Batty L (ed) Ecology of industrial pollution: remediation, restoration and preservation. Cambridge University Press, Cambridge
Tokeshi M (1993) Species abundance patterns and community structure. Adv Ecol Res 24:112–186
US Environmental Protection Agency (USEPA) (1993) Methods for chemical analysis of water and wastes. Environ Monit Support Lab, Cincinnati
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
White JR, Reddy KR (2000) The effects of phosphorus loading on organic nitrogen mineralization of soils and detritus along a nutrient gradient in the northern Everglades, Florida. Soil Sci Soc of Am J 64:1525–1534
Acknowledgments
We thank Mr. John Gardner, Environmental Manager for Mining, ALCOA World Alumina Australia, for facilitating the funding to support this study. Support from the Department of Defense (DoD) grant # W911NF-10-1-0146 is also acknowledged. We also acknowledge the staff at Jamalco for logistics with sample collection, Maverick and Benjamin LeBlanc for their assistance in soil geochemical characterization, and the three anonymous reviewers for their very helpful critique.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dawn E. Lewis and Ashvini Chauhan contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. SI-1
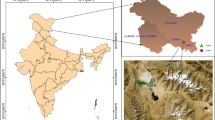
Map of Jamaica showing locations of bauxite rich soils. Also depicted is pictorial representation of un-mined and post-mined rehabilitated sites from where samples for this study were obtained. (DOC 3873 kb)
Fig. S1-1I
Taxonomic affiliation of bacterial phyla/taxa identified by PCR-DGGE analyses along un-mined and three rehabilitated chronosequence soils collected from the Mocho Mountains of Jamaica. Shown are A, wet season microbiota; B, dry season microbiota and C, total percentage associated with each identified phyla/taxa regardless of seasons and soils. MUnm refers to Mocha Unmined; R07 to site rehabilitated in 2007; R97 to site rehabilitated in 1997; and R87 to site rehabilitated in 1987. (DOC 62 kb)
Fig. SI-III
Taxonomic affiliation of bacterial phyla/taxa identified by PCR-clone library analyses along un-mined and three rehabilitated chronosequence soils collected from the Mocho Mountains of Jamaica. Shown are A, wet season microbiota; B, dry season microbiota and C, total percentage associated with each identified phyla/taxa regardless of seasons and soils. MUnm refers to Mocha Unmined; R07 to site rehabilitated in 2007; R97 to site rehabilitated in 1997; and R87 to site rehabilitated in 1987. (DOC 130 kb)
Fig. SIV
Differences in the bacterial phyla/taxa identified from PCR-DGGE and PCR-Cloning analyses evaluated by principal coordinate analysis (PCA). Percentages represented in the axis labels are variation explained by the first two principal coordinates. (DOC 35 kb)
Rights and permissions
About this article
Cite this article
Lewis, D.E., Chauhan, A., White, J.R. et al. Microbial and Geochemical Assessment of Bauxitic Un-mined and Post-mined Chronosequence Soils from Mocho Mountains, Jamaica. Microb Ecol 64, 738–749 (2012). https://doi.org/10.1007/s00248-012-0020-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-012-0020-3