Abstract
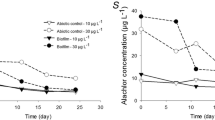
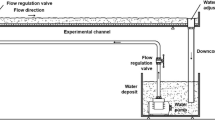
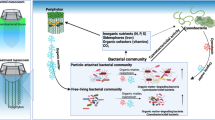
We conducted a field survey of periphyton cultivated on benthic mesh installations in freshwater aquatic systems, including two constructed wetlands and a pond, and also studied periphyton grown on a benthic mesh in laboratory mesocosms. The objectives of this study were to (1) determine if periphyton cultivated on benthic mesh denitrifies at higher rates than the underlying sediments and (2) determine if denitrification rates within periphyton vary with characteristics such as algal and bacterial community structure and biomass. We measured denitrification potential rates of field and laboratory periphyton by the acetylene inhibition method. We characterized algal community composition by algal identification and bacterial community composition by terminal restriction fragment length polymorphisms. Periphyton collected on benthic mesh from our field sites denitrified at significantly higher rates than the underlying sediments, regardless of sampling site or season. Results from both our field survey and laboratory studies indicated a significant, positive correlation between diatom presence and denitrification rate. In our laboratory studies, we found that periphyton with the highest diatom abundance showed the highest denitrification rates as well as a distinct bacterial community composition. These results suggest a synergistic relationship between diatoms and denitrifying bacteria that warrants further study.






Similar content being viewed by others
References
APHA (1998) Standard methods for the examination of water and wastewater. American Public Health Association, Washington, DC
Arnon S, Packman AI, Peterson CG, Gray KA (2007) Effects of overlying velocity on periphyton structure and denitrification. J Geophys Res 112:G01002
Biggs BJF, Goring DG, Nikora VI (1998) Subsidy and stress responses of stream periphyton to gradients in water velocity as a function of community growth form. J Phycol 34:598–607
Biggs BJF, Stevenson RJ, Lowe RL (1998) A habitat matrix conceptual model for stream periphyton. Arch Hydrobiol 143:21–56
Braid MD, Danields LM, Kitts CL (2003) Removal of PCR inhibitors from soil DNA by chemical flocculation. J Microb Methods 52(3):389–393
Bray JR, Curtis JT (1957) An ordination of the upland forest communities of Southern Wisconsin. Ecol Monogr 27(4):325–349
Burkholder JM, Wetzel RG, Klomparens KL (1990) Direct comparison of phosphate uptake by adnate and loosely attached microalgae within an intact biofilm matrix. Appl Environ Microbiol 56:2882–2890
Casterlin ME, Reynolds WW (1977) Seasonal algal succession and cultural eutrophication in a north temperate lake. Hydrobiologia 54(2):99–108
Clarke KR, Ainsworth M (1993) A method of linking multivariate community structure to environmental variables. Mar Ecol Prog Ser 92:205–219
Clavier J, Boucher G, Chauvaud L, Fichez R, Chifflet S (2005) Benthic response to ammonium pulses in a tropical lagoon: implications for coastal environmental processes. J Exp Mar Biol Ecol 316(2):231–241
Clement JC, Pinay G, Marmonier P (2002) Seasonal dynamics of denitrification along topohydrosequences in three different riparian wetlands. J Environ Qual 31:1025–1037
Crumpton WG (1987) A simple and reliable method for making permanent mounts of phytoplankton for light and fluorescence microscopy. Limnol Oceanogr 32:1154–1159
de Beer D, Schramm A (1999) Micro-environments and mass transfer phenomena in biofilms studied with microsensors. Water Sci Technol 39(7):173–178
Drenovsky RE, Vo D, Graham KJ, Scow KM (2004) Soil water content and organic carbon availability are major determinants of soil microbial community composition. Microb Ecol 48:424–430
Eriksson PG, Weisner SEB (1997) Nitrogen removal in a wastewater reservoir: the importance of denitrification by epiphytic biofilms on submersed vegetation. J Environ Qual 26(3):905–910
Espeland EM, Wetzel RG (2001) Effects of photosynthesis on bacterial phosphatase production in biofilms. Microb Ecol 42(3):328–337
Fisher MM, Wilcox LW, Graham LE (1998) Molecular characterization of epiphytic bacterial communities on charophycean green algae. Appl Environ Microbiol 64(11):4384–4389
Fleming-Singer MS, Horne AJ (2002) Enhanced nitrate removal efficiency in wetland microcosms using an episediment layer for denitrification. Environ Sci Technol 36:1231–1237
Gough HL, Stahl DA (2003) Optimization of direct cell counting in sediments. J Microb Methods 52:39–46
Graham JM, Kent AD, Lauster GH, Yannarell AC, Graham LE, Triplett EW (2004) Seasonal dynamics of phytoplankton and planktonic protozoan communities in a northern temperate humic lake: Diversity in a dinoflagellate dominated system. Microb Ecol 48(4):528–540
Grossart H.-P, Levold F, Allgaler M, Simon M, Brinkhoff T (2005) Marine diatom species harbour distinct bacterial communities. Environ Microbiol 7(6):860–873
Haack TK, McFetters GA (1982) Nutritional relationships among microorganisms in an epilithic biofilm community. Microb Ecol 8(2):115–126
Hamels I, Mussche H, Sabbe K, Muylaert K, Vyverman W (2004) Evidence for constant and highly specific active food selection by benthic ciliates in mixed diatoms assemblages. Limnol Oceanogr 49(1):58–68
Hepinstall JA, Fuller RL (1994) Periphyton reactions to different light and nutrient levels and the response of bacteria to these manipulations. Arch Hydrobiol 131(2):161–173
Hauer FR, Lamberti GA (1996) Methods in stream ecology. Academic, San Diego, CA
Hey DL (1994) River or lake bottom apparatus for scavenger fish control. US Patent 576,244,9
Hillebrand H, Durselen CD, Kirschtel D, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J Phycol 35:403–424
Holtan-Hartwig L, Dorsch P, Bakken LR (2000) Comparison of denitrifying communities in organic soils: kinetics of NO and N2O reduction. Soil Biol Biochem 32:833–843
Howarth RW, Billen G, Swaney D, Townsend A, Jaworski N, Lajtha K, Downing JA, Elmgren R, Caraco N, Jordan T, Berendse F, Freney J, Kudeyarov V, Murdoch P, Zhu ZL (1996) Regional nitrogen budgets and riverine N & P fluxes for the drainages to the North Atlantic Ocean: Natural and human influences. Biogeochemistry 35:75–139
Janus LR, Angeloni NL, McCormack J, Rier ST, Tuchman NC, Kelly JJ (2005) Elevated atmospheric CO2 alters soil microbial communities associated with Trembling Aspen (Populus tremuloides) roots. Microb Ecol 50:102–109
Jeffrey SW, Humphrey GF (1975) New spectrophotometric equations for determining chlorophylls a, b, c1, and c2 in higher plants, algae, and natural phytoplankton. Biochem Physiol Pflanzen 167:191–194
Joye SB, Smith SV, Hollibaugh JT, Paerl HW (1996) Estimating denitrification rates in estuarine sediments: a comparison of stoichiometric and acetylene based methods. Biogeochemistry 33:197–215
Kostel JA, Wang A, St. Amand A, Gray KA (1999) 1. Use of a novel laboratory stream system to study the ecological impact of PCB exposure in a periphytic biolayer. Water Res 33(18):3735–3748
Kozub DD, Liehr SK (1999) Assessing denitrification rate limiting factors in a constructed wetland receiving landfill leachate. Water Sci Technol 40(3):75–82
Kurata A (1983) Nutrient removal by epiphytic microorganisms of Phragmites communis. In: Wetzel RG (ed) Periphyton of freshwater ecosystems. Junk, The Hague
Lamberti GA, Resh VH (1987) Comparability of introduced tiles and natural substrates for sampling lotic bacteria, algae and macroinvertebrates. Freshw Biol 15:21–30
Liu W, Marsh TL, Cheng H, Forney LJ (1997) Characterization of microbial diversity by determining terminal restriction fragment length polymorphisms of genes encoding 16S rRNA. Appl Environ Microbiol 63(11):4516–4522
McCormick PV, Stevenson RJ (1989) Effects of snail grazing on benthic algal community structure in different nutrient environments. J North Am Benthol Soc 8:162–172
McIntire CD (1968) Physiological-Ecological studies of benthic algae in laboratory streams. J Water Pollut Control Fed 40:1940–1952
Myklestad SM (1995) Release of extracellular products by phytoplankton with special emphasis on polysaccharides. Sci Total Environ 165(1–3):155–164
Murray RE, Cooksey KE, Priscu JC (1986) Stimulation of bacterial DNA synthesis by algal exudates in attached algal-bacterial consortia. Appl Environ Microbiol 52:1177–1182
Neely RK, Wetzel RG (1995) Simultaneous use of 14C and 3H to determine autotrophic production and bacterial protein production in periphyton. Microb Ecol 30:227–237
Olrik K, Blomqvist P, Brettum P, Cronberg G, Eloranta P (1998) Methods for quantitative assessment of phytoplankton in freshwaters, part I. Naturvårdsverket, Stockholm
Passy SI (2001) Spatial paradigms of lotic diatom distribution: a landscape ecology approach. J Phycol 37:370–378
Payne WJ (1991) A review of methods for field measurements of denitrification. For Ecol Manag 44:5–14
Peterson CG, Stevenson RJ (1990) Post-spate development of epilithic algal communities in different current environments. Can J Bot 68:2092–2102
Peterson CG, Valett HM, Dahm CN (2001) Shifts in habitat templates for lotic microalgae linked to interannual variation in snowmelt intensity. Limnol Oceanogr 46:858–870
Pinckney JL, Paerl HW (1997) Anoxygenic photosynthesis and nitrogen fixation by a microbial mat community in a Bahamian hypersaline lagoon. Appl Environ Microbiol 63(2):420–426
Polymenakou PN, Bertilsson S, Tselepides A, Stephanou EG (2005) Links between geographic location, environmental factors, and microbial community composition in sediments of the Eastern Mediterranean Sea. Microb Ecol 49(3):367–378
Riber HH, Wetzel RG (1987) Boundary-layer and internal diffusion effects on phosphorus fluxes in lake periphyton. Limnol Oceanogr 32:1181–1194
Rich JJ, Heichen RS, Bottomley PJ, Cromack Jr K, Myrold DD (2003) Community composition and functioning of denitrifying bacteria from adjacent meadow and forest soils. Appl Environ Microbiol 69:5974–5982
Romani AM, Sabater S (2000) Influence of algal biomass on extracellular enzyme activity in river biofilms. Microb Ecol 40(1):16–24
Saunders DL, Kalff J (2001) Nitrogen retention in wetlands, lakes and rivers. Hydrobiology 443:205–212
Schafer H, Abbas B, Witte H, Muyzer G (2002) Genetic diversity of ‘satellite’ bacteria present in cultures of marine diatoms. FEMS Microb Ecol 42(1):25–35
Sirivedhin T, Gray KA (2006) Factors affecting denitrification rates in experimental wetlands: field and laboratory studies. Ecol Eng 26:167–181
Sladeckova A, Marvan P, Vymazal J (1983) The utilization of periphyton in waterworks pretreatment for nutrient removal from enriched influents. In: Wetzel RG (ed) Periphyton of freshwater ecosystems. Junk, The Hague
Smith DJ, Underwood GJC (2000) The production of extracellular carbohydrates by estuarine benthic diatoms: the effects of growth phase and light and dark treatment. J Phycol 36:321–333
Sorensen J, Jorgensen T, Brandt S (1988) Denitrification in stream epilithon: seasonal variation in Gelbaek and Rabis Baek, Denmark. FEMS Microb Ecol 53:345–354
Steinman AD, Mulholland PJ, Kirschtel DB (1991) Interactive effects of nutrient reduction and herbivory on biomass, taxonomic structure, and P uptake in lotic periphyton communities. Can J Fish Aquat Sci 48:1951–1959
Stevenson RJ, Glover R (1993) Effects of algal density and current on ion transport through periphyton communities. Limnol Oceanogr 38:1276–1281
Teissier S, Torre M (2002) Simultaneous assessment of nitrification and denitrification on freshwater epilithic biofilms by acetylene block method. Water Res 36(15):3803–3811
Tiedje JM (1988) Ecology of denitrification and dissimilatory nitrate reduction to ammonium. In: Zehnder AJB (ed) Biology of anaerobic microorganisms. Wiley, New York, NY,
Toet S, Huibers LHFA, Van Logtestijn RSP, Verhoeven JTA (2003) Denitrification in the periphyton associated with plant shoots and in the sediment of a wetland system supplied with sewage treatment plant effluent. Hydrobiologia 501(1–3):29–44
Tuchman ML, Stevenson RJ (1980) Comparison of clay tile, sterilized rock, and natural substrate diatom communities in a small stream in southeastern Michigan, USA. Hydrobiologia 75:73–79
Van Raalte CD, Patriquin DG (1979) Use of the “cetylene blockage” technique for assaying denitrification in a salt marsh. Mar Biol 52:315–320
Wetzel RG (1983) Periphyton of freshwater ecosystems. Junk, The Hague
White JR, Reddy KR (1999) Influence of nitrate and phosphorus loading on denitrifying enzyme activity in Everglades Wetland soils. Soil Sci Soc Am J 63:1945–1954
Whitford LA (1960) The current effect and growth of fresh-water algae. Trans Am Microsc Soc 79:302–309
Zumft WG (1997) Cell biology and molecular basis of denitrification. Microbiol Mol Biol Rev 61:533–616
Acknowledgment
We would like to acknowledge the US Department Agriculture (grant # 2002–35102–12373) for supporting this project. We would also like to thank Dr. Donald Hey, Kathy Paap, Jerry Curan (Wetlands Research, Inc.), and Bob Kirshner (Chicago Botanic Gardens) for their help in maintaining the field sites. In addition, we would like to express our gratitude to Carla Ng, Laura Pigion, Clare Frederick, and Caitlin Kielhorn for their assistance with the fieldwork, and Dr. Deanna Hurum, Dr. Tanita Sirivedhin, Dr. Kristin Searcy, Dr. Mary Jo Kirisits, Dr. Gail Teitzel, Dr. Jill Kostel, and Dr. Susan Fishbain for their assistance in the laboratory. Finally, we would like to acknowledge Dr. Ann St. Amand for the assistance with algal identification.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ishida, C.K., Arnon, S., Peterson, C.G. et al. Influence of Algal Community Structure on Denitrification Rates in Periphyton Cultivated on Artificial Substrata. Microb Ecol 56, 140–152 (2008). https://doi.org/10.1007/s00248-007-9332-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-007-9332-0