Abstract
Background
It is difficult to diagnosis early stage germ cell tumors originating in the basal ganglia, but early recognition is important for better outcome.
Objective
To evaluate serial MR images of basal ganglia germ cell tumors, with emphasis on the features of early stage tumors.
Materials and methods
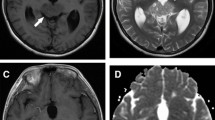
We retrospectively reviewed serial MR images of 15 tumors in 14 children and young adults. We categorized MR images of the tumors as follows: type I, ill-defined patchy lesions (<3 cm) without cyst; type II, small mass lesions (<3 cm) with cyst; and type III, large lesions (≥3 cm) with cyst. We also assessed temporal changes of the MR images.
Results
On the initial images, 8 of 11 (73%) type I tumors progressed to types II or III, and 3 of 4 (75%) type II tumors progressed to type III. The remaining 4 tumors did not change in type. All type II tumors (5/5, 100%) that changed from type I had a few tiny cysts. Intratumoral hemorrhage was observed even in the type I tumor. Ipsilateral hemiatrophy was observed in most of the tumors (13/15, 87%) on initial MR images. As tumors grew, cystic changes, intratumoral hemorrhage, and ipsilateral hemiatrophy became more apparent.
Conclusion
Early stage basal ganglia germ cell tumors appear as ill-defined small patchy hyperintense lesions without cysts on T2-weighted images, are frequently associated with ipsilateral hemiatrophy, and sometimes show microhemorrhage. Tumors develop tiny cysts at a relatively early stage.




Similar content being viewed by others
References
Packer RJ, Cohen BH, Cooney K (2000) Intracranial germ cell tumors. Oncologist 5:312–320
Echevarria ME, Fangusaro J, Goldman S (2008) Pediatric central nervous system germ cell tumors: a review. Oncologist 13:690–699
Raybaud C, Barkovich AJ et al (2012) Intracranial, orbital, and neck masses of childhood. In: Barkovich AJ, Raybaud C (eds) Pediatric neuroimaging. Lippincott Williams & Wilkins, Philadelphia
Crawford JR, Santi MR, Vezina G et al (2007) CNS germ cell tumor (CNSGCT) of childhood: presentation and delayed diagnosis. Neurology 68:1668–1673
Soejima T, Takeshita I, Yamamoto H et al (1987) Computed tomography of germinomas in basal ganglia and thalamus. Neuroradiology 29:366–370
Okamoto K, Ito J, Ishikawa K et al (2002) Atrophy of the basal ganglia as the initial diagnostic sign of germinoma in the basal ganglia. Neuroradiology 44:389–394
Higano S, Takahashi S, Ishii K et al (1994) Germinoma originating in the basal ganglia and thalamus: MR and CT evaluation. AJNR Am J Neuroradiol 15:1435–1441
Rasalkar DD, Chu WC, Cheng FW et al (2010) Atypical location of germinoma in basal ganglia in adolescents: radiological features and treatment outcomes. Br J Radiol 83:261–267
Kim DI, Yoon PH, Ryu YH et al (1998) MRI of germinomas arising from the basal ganglia and thalamus. Neuroradiology 40:507–511
Moon WK, Chang KH, Kim IO et al (1994) Germinomas of the basal ganglia and thalamus: MR findings and a comparison between MR and CT. AJR Am J Roentgenol 162:1413–1417
Lou X, Ma L, Wang FL et al (2009) Susceptibility-weighted imaging in the diagnosis of early basal ganglia germinoma. AJNR Am J Neuroradiol 30:1694–1699
Lou X, Tian C, Chen Z et al (2012) Differential diagnosis of infarct-like intracranial ectopic germinomas and subacute lacunar infarct on susceptibility-weighted imaging. J Magn Reson Imaging 36:92–98
Ozelame RV, Shroff M, Wood B et al (2006) Basal ganglia germinoma in children with associated ipsilateral cerebral and brain stem hemiatrophy. Pediatr Radiol 36:325–330
Phi JH, Cho BK, Kim SK et al (2010) Germinomas in the basal ganglia: magnetic resonance imaging classification and the prognosis. J Neurooncol 99:227–236
Wong ST, Yuen SC, Fong D (2009) Pathophysiological mechanism of ipsilateral cerebral and brainstem hemiatrophy in basal ganglia germ cell tumors: case report. Childs Nerv Syst 25:693–699
Fujii Y, Saito Y, Ogawa T et al (2008) Basal ganglia germinoma: diagnostic value of MR spectroscopy and (11)C-methionine positron emission tomography. J Neurol Sci 270:189–193
Lee J, Lee BL, Yoo KH et al (2009) Atypical basal ganglia germinoma presenting as cerebral hemiatrophy: diagnosis and follow-up with 11C-methionine positron emission tomography. Childs Nerv Syst 25:29–37
Sudo A, Shiga T, Okajima M et al (2003) High uptake on 11C-methionine positron emission tomographic scan of basal ganglia germinoma with cerebral hemiatrophy. AJNR Am J Neuroradiol 24:1909–1911
Jager PL, Vaalburg W, Pruim J et al (2001) Radiolabeled amino acids: basic aspects and clinical applications in oncology. J Nucl Med 42:432–445
Nakagawa M, Kuwabara Y, Sasaki M et al (2002) 11C-methionine uptake in cerebrovascular disease: a comparison with 18F-fDG PET and 99mTc-HMPAO SPECT. Ann Nucl Med 16:207–211
Coakley J, Kellie SJ, Nath C et al (2005) Interpretation of alpha-fetoprotein concentrations in cerebrospinal fluid of infants. Ann Clin Biochem 42:24–29
Fujimaki T, Mishima K, Asai A et al (2000) Levels of beta-human chorionic gonadotropin in cerebrospinal fluid of patients with malignant germ cell tumor can be used to detect early recurrence and monitor the response to treatment. Jpn J Clin Oncol 30:291–294
Tang J, Ma Z, Luo S et al (2008) The germinomas arising from the basal ganglia and thalamus. Childs Nerv Syst 24:303–306
Kwak R, Suzuki J (1970) Brain tumor with ipsilateral cerebral hemiatrophy. No To Shinkei 22:1439–1446
Kuhn MJ, Johnson KA, Davis KR (1988) Wallerian degeneration: evaluation with MR imaging. Radiology 168:199–202
Mutoh K, Okuno T, Ito M et al (1988) Ipsilateral atrophy in children with hemispheric cerebral tumors: CT findings. J Comput Assist Tomogr 12:740–743
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Lee, S.M., Kim, IO., Choi, Y.H. et al. Early imaging findings in germ cell tumors arising from the basal ganglia. Pediatr Radiol 46, 719–726 (2016). https://doi.org/10.1007/s00247-016-3542-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-016-3542-x