Abstract
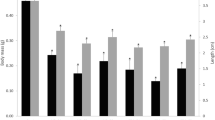
Environmental contaminants, such as polycyclic aromatic hydrocarbons (PAHs), can disrupt the endocrine system and affect reproductive function of humans and wildlife. In this study, we exposed Japanese medaka (Oryzias latipes) to water-borne or food-spiked phenanthrene, an ubiquitous PAH, and investigated the chronic effects of the chemical on medaka reproduction. The results showed that phenanthrene significantly prolonged the time to hatch for embryos. Other parameters such as fecundity and fertility, organ-somatic ratios, hepatic vitellogenin production, and the histology of testes and ovaries were not different between the control and phenanthrene-treated groups. This suggests that the time to hatch in embryos might be a more sensitive biomarker for environmental contaminants.




Similar content being viewed by others
References
Barron MG, Carls MG, Heintz R, Rice SD (2004a) Evaluation of fish early life-stage toxicity models of chronic embryonic exposures to complex polycyclic aromatic hydrocarbon mixtures. Toxicol Sci 8:60–67. doi:10.1093/toxsci/kfh051
Barron MG, Heintz R, Rice S (2004b) Relative potency of PAHs and heterocycles as aryl hydrocarbon receptor agonists in fish. Marine Environ Res 58:95–100. doi:10.1016/j.marenvres.2004.03.001
Bellas J, Thor P (2007) Effects of selected PAHs on reproduction and survival of the calanoid copepod Acartia tonsa. Ecotoxicology 16:465–474. doi:10.1007/s10646-007-0152-2
Caserta D, Maranghi L, Mantovani A, Marci R, Maranghi F, Moscarini M (2008) Impact of endocrine disruptor chemicals in gynaecology. Hum Reprod Update 14:59–72. doi:10.1093/humupd/dmm025
Chang CS, Liao SS (1987) Topographic recognition of cyclic hydrocarbons and related compounds by receptors for androgens, estrogens, and glucocorticoids. J Steroid Biochem 27:123–131. doi:10.1016/0022-4731(87)90303-7
Chen H-H (2003) Phase association of polycyclic aromatic hydrocarbons in sediments from Erhjin River. MS thesis, Chang-Jung Christian University, Tainan, Taiwan (in Chinese)
Dickerson SM, Gore AC (2007) Estrogenic environmental endocrine-disrupting chemical effects on reproductive neuroendocrine function and dysfunction across the life cycle. Rev Endocr Metab Disord 8:143–159. doi:10.1007/s11154-007-9048-y
Debruyn AM, Wernick BG, Stefura C, McDonald BG, Rudolph BL, Patterson L, Chapman PM (2007) In situ experimental assessment of lake whitefish development following a freshwater oil spill. Environ Sci Technol 15(41):5983–6989
Doong R-A, Lin Y-T (2004) Characterization and distribution of polycyclic aromatic hydrocarbon contaminations in surface sediment and water from Gao-ping River, Taiwan. Water Res 38:1733–1744. doi:10.1016/j.watres.2003.12.042
Edwards TM, Moore BC, Guillette LJ Jr (2006) Reproductive dysgenesis in wildlife: a comparative view. Int J Androl 29:109–121. doi:10.1111/j.1365-2605.2005.00631.x
Evans AD, Nipper M (2008) The influence of biomass on the toxicity of hydrophobic organic contaminants. Arch Environ Contam Toxicol 54:219–225. doi:10.1007/s00244-007-9019-z
Evanson M, Van Der Kraak GJ (2001) Stimulatory effects of selected PAHs on testosterone production in goldfish and rainbow trout and possible mechanisms of action. Comp Biochem Physiol 130:249–258
Fang M-D, Lee C-L, Yu C-S (2003) Distribution and source recognition of polycyclic aromatic hydrocarbons in the sediments of Hsin-ta Harbour and adjacent coastal areas, Taiwan. Marine Pollut Bull 46:941–953. doi:10.1016/S0025-326X(03)00099-7
Farwell A, Nero V, Croft M, Bal P, Dixon DG (2006) Modified Japanese medaka embryo-larval bioassay for rapid determination of developmental abnormalities. Arch Environ Contam Toxicol 51:600–607. doi:10.1007/s00244-005-0319-x
Frouin H, Pellerin J, Fournier M, Pelletier E, Richard P, Pichaud N, Rouleau C, Garnerot F (2007) Physiological effects of polycyclic aromatic hydrocarbons on soft-shell clam Mya arenaria. Aquat Toxicol 82:120–134. doi:10.1016/j.aquatox.2007.02.005
Hilscherova K, Kannan K, Holoubek I, Giesy JP (2002) Characterization of estrogenic activity of riverine sediments from the Czech Republic. Arch Environ Contam Toxicol 43:175–185. doi:10.1007/s00244-002-1128-0
Horng C-Y (1998) Influence of the marine polychaete, Capitella sp.I, on the fate of sediment-bound polycyclic aromatic hydrocarbons: the role of feeding activity. Ph.D. thesis, Rutgers University, New Brunswick, NJ
Hwang HM, Wade TL (2008) Aerial distribution, temperature-dependent seasonal variation, and sources of polycyclic aromatic hydrocarbons in pine needles from the Houston metropolitan area, Texas, USA. J Environ Sci Health A Tox Hazard Subst Environ Eng 43:1243–1251
Hylland K (2006) Polycyclic aromatic hydrocarbon (PAH) ecotoxicology in marine ecosystems. J Toxicol Environ Health A 69:109–123. doi:10.1080/15287390500259327
Incardona JP, Collier TK, Scholz NL (2004) Defects in cardiac function precede morphological abnormalities in fish embryos exposed to polycyclic aromatic hydrocarbons. Toxicol Appl Pharm 196:191–205. doi:10.1016/j.taap.2003.11.026
Incardona JP, Day HL, Collier TK, Scholz NL (2006) Developmental toxicity of 4-ring polycyclic aromatic hydrocarbons in zebrafish is differentially dependent on AH receptor isoforms and hepatic cytochrome P4501A metabolism. Toxicol Appl Pharmacol 217:308–321. doi:10.1016/j.taap.2006.09.018
Kang JH, Aasi D, Katayama Y (2007) Bisphenol A in the aquatic environment and its endocrine-disruptive effects on aquatic organisms. Crit Rev Toxicol 37:607–625. doi:10.1080/10408440701493103
Kohn MC, Melnick RL (2002) Biochemical origins of the non-monotonic receptor-mediated dose-response. J Mol Endocrinol 29:113–123. doi:10.1677/jme.0.0290113
Lin T-C, Chang F-H, Hsieh J-H, Chao H-R, Chao M-R (2002) Characteristics of polycyclic aromatic hydrocarbons and total suspended particulate in indoor and outdoor atmosphere of a Taiwanese temple. J Hazard Mater A95:1–12. doi:10.1016/S0304-3894(02)00146-2
Luthy RG, Aiken GR, Brusseau ML, Cunningham SD, Gschwend PM, Pignatello JJ, Reinhard M, Traina SJ, Webber WJ Jr, Westall JC (1997) Sequestration of hydrophobic organic contaminants by geosorbents. Environ Sci Technol 31:3341–3347. doi:10.1021/es970512m
Mathew R, McGrath JA, Di Toro DM (2008) Modeling polycyclic aromatic hydrocarbon bioaccumulation and metabolism in time-variable early life-stage exposures. Environ Toxicol Chem 27:1515–1525. doi:10.1897/07-355.1
Mattison DR, White NB, Nightingale MR (1980) The effect of benzo(a)pyrene on fertility, primordial oocyte number, and ovarian response to pregnant mare’s serum gonadotropin. Pediatr Pharmacol 1:143–151
McElroy AE, Bogler A, Weisbaum D, Norris M, Mendelman LV, Setlow R, Winn R (2006) Uptake, metabolism, mutant frequencies and mutational spectra in λ transgenic medaka embryos exposed to benzo[α]pyrene dosed sediments. Marine Environ Res 62:S273–S277. doi:10.1016/j.marenvres.2006.04.019
Meador JP, Stein JE, Reichert WL, Varanasi U (1995) Bioaccumulation of polycyclic aromatic hydrocarbons by marine organisms. Rev Environ Contam Toxicol 143:79–165
Monteiro PRR, Reis-Henriques MA, Coimbra J (2000a) Plasma steroid levels in female flounder (Platichthys flesus) after chronic dietary exposure to single polycyclic aromatic hydrocarbons. Marine Environ Res 49:453–467. doi:10.1016/S0141-1136(99)00085-9
Monteiro PRR, Reis-Henriques MA, Coimbra J (2000b) Polycyclic aromatic hydrocarbons inhibit in vitro ovarian steroidogenesis in the flounder (Platichthys flesus L). Aquat Toxicol 48:549–559. doi:10.1016/S0166-445X(99)00055-7
Oxendine SL, Cowden J, Hinton DE, Padilla S (2006) Vulnerable windows for developmental ethanol toxicity in the Japanese medaka fish (Oryzias latipes). Aquat Toxicol 80:396–404. doi:10.1016/j.aquatox.2006.10.007
PAN Pesticide Database (2008) Chemical toxicity studies on aquatic organisms. Available from http://www.pesticideinfo.org/List_AquireAll.jsp?Rec_Id=AQ614&offset=50 (accessed November 12, 2008)
Patel MR, Scheffler BE, Wang L, Willett KL (2006) Effects of benzo(a)pyrene exposure on killifish (Fundulus heteroclitus) aromatase activities and mRNA. Aquat Toxicol 77:267–278. doi:10.1016/j.aquatox.2005.12.009
Perera FP, Rauh V, Whyatt RM, Tsai WY, Tang D, Diaz D, Hoepner L, Barr D, Tu YH, Camann D, Kinney P (2006) Effect of prenatal exposure to airborne polycyclic aromatic hydrocarbons on neurodevelopment in the first 3 years of life among inner-city children. Environ Health Perspect 114:1287–1292
Ramakrishnan S, Wayne NL (2008) Impact of bisphenol-A on early embryonic development and reproductive maturation. Reprod Toxicol 25:177–183. doi:10.1016/j.reprotox.2007.11.002
Rasier G, Toppari J, Parent A-S, Bourguignon J-P (2006) Female sexual maturation and reproduction after prepubertal exposure to estrogens and endocrine disrupting chemicals: a review of rodent and human data. Mol Cell Endocrinol 254–255:187–201. doi:10.1016/j.mce.2006.04.002
Reynolds WJ, Feist SW, Jones GJ, Lyons BP, Sheahan DA, Stentiford GD (2003) Comparison of biomarker and pathological responses in flounder (Platichthys flesus L). induced by ingested polycyclic aromatic hydrocarbon (PAH) contamination. Chemosphere 52:1135–1145. doi:10.1016/S0045-6535(03)00332-1
Rhodes S, Farwell A, Hewitt LM, MacKinnon M, Dixon DG (2005) The effects of dimethylated and alkylated polycyclic aromatic hydrocarbons on the embryonic development of the Japanese medaka. Ecotoxicol Environ Safety 60:247–258. doi:10.1016/j.ecoenv.2004.08.002
Seruto C, Sapozhnikova Y, Schlenk D (2005) Evaluation of the relationships between biochemical endpoints of PAH exposure and physiological endpoints of reproduction in male California Halibut (Paralichthys californicus) exposed to sediments from a natural oil seep. Marine Environ Res 60:454–465. doi:10.1016/j.marenvres.2005.01.004
Sherry JP, Whyte JJ, Karrow NA, Gamble A, Boerman HJ, Bol NC, Dixon DG, Solomon KR (2006) The effect of creosote on vitellogenin production in rainbow trout (Oncorhynchus mykiss). Arch Environ Contam Toxicol 50:65–68. doi:10.1007/s00244-004-0255-1
Soin T, Smagghe G (2007) Endocrine disruption in aquatic insects: a review. Ecotoxicology 16:83–93. doi:10.1007/s10646-006-0118-9
Sverdrup LE, Jensen J, Kelley AE, Krogh PH, Stenersen J (2002) Efffects of eight polycyclic aromatic compounds on the survival and reproductionof Enchytraeus crypticus (Oligochaeta, Clitellata). Environ Toxicol Chem 21:109–114. doi:10.1897/1551-5028(2002)021<0109:EOEPAC>2.0.CO;2
Takeda K, Tsukue N, Yoshida S (2004) Endocrine-disrupting activity of chemicals in diesel exhaust and diesel exhaust particles. Environ Sci 11:33–45
Tang D, Li TY, Liu JJ, Zhou ZJ, Yuan T, Chen YH, Rauh VA, Xie J, Perera F (2008) Effects of prenatal exposure to coal-burning pollutants on children’s development in China. Environ Health Perspect 116:674–679. doi:10.1289/ehp.11763R
Tsai KS, Yang RS, Liu SH (2004) Benzo[a]pyrene regulates osteoblast proliferation through an estrogen receptor-related cyclooxygenase-1 pathway. Chem Res Toxicol 17:679–684. doi:10.1021/tx0499517
USEPA (United States Environmental Protection Agency) (2008) Test methods for evaluating solid waste, physical/chemical methods, SW-846. Available from http://epa.gov/epawaste/hazard/testmethods/index.html (accessed October 10, 2006)
Valerio F, Bottino P, Ugolini D, Cimberle MR, Tozzi GA, Frigerio A (1984) Chemical and photochemical degradation of polycyclic aromatic hydrocarbons in the atmosphere. Sci Total Environ 40:169–188. doi:10.1016/0048-9697(84)90350-4
Van de Wiele T, Vanhaecke L, Boeckaert C, Peru K, Headley J, Verstraete W, Siciliano S (2005) Human colon microbiota transform polycyclic aromatic hydrocarbons to estrogenic metabolites. Environ Health Perspect 113:6–10
Vinggaard AM, Hnida C, Larsen JC (2000) Environmental polycyclic aromatic hydrocarbons affect androgen receptor activation in vitro. Toxicology 145:173–183. doi:10.1016/S0300-483X(00)00143-8
von Westerhagen H (1988) Sublethal effects of pollutants on fish eggs and larvae. In: Hoar WS, Randall DJ (eds) Fish physiology: the physiology of developing fish, vol 11. Academic Press, San Diego, CA, pp 253–346
Walpita CN, Van der Geyten S, Rurangwa E, Darras VM (2007) The effect of 3, 5, 3’-triiodothyronine supplementation on zebrafish (Danio rerio) embryonic development and expression of iodothyronine deiodinases and thyroid hormone receptors. Gen Comp Endocrinol 152:206–214. doi:10.1016/j.ygcen.2007.02.020
Zha J, Wang Z (2006) Acute and early life stage toxicity of industrial effluent on Japanese medaka (Oryzias latipes). Sci Total Environ 357:112–119. doi:10.1016/j.scitotenv.2005.04.038
Acknowledgments
We thank Dr. Chien-min Chen for technical advice and support on the histological examination in this study. This work was supported by grants to W. L. from Changhua Christian Hospital, Chang Jung Christian University, and National Science Council (NSC95-2314-B-309-005) in Taiwan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horng, CY., Lin, HC. & Lee, W. A Reproductive Toxicology Study of Phenanthrene in Medaka (Oryzias latipes). Arch Environ Contam Toxicol 58, 131–139 (2010). https://doi.org/10.1007/s00244-009-9335-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-009-9335-6