Abstract
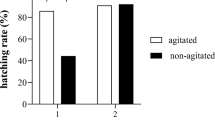
The Japanese medaka (Oryzias latipes) early-life-stage bioassay (18-day test) provides a suitable laboratory model for the evaluation of toxicant impact. The naturally high variability in the time to hatch for medaka embryos is, however, a major limitation in terms of the duration of the test. In this study, the 18-day test was modified to use agitation to synchronize hatch and reduce time to hatch for nonexposed embryos. Then, bioassays were conducted using two different complex mixtures (polycyclic aromatic hydrocarbons (PAHs) and naphthenic acids (NAs)) to compare the sensitivity of the measurement endpoints using the 18-day standard protocol (SP) and the modified protocol (MP). Agitation reduced time to hatch by one third or more and improved hatch success (100%). The MP proved to be a more sensitive test method for the sublethal evaluation of a stressor (PAH mixture) that caused reduced hatch length and induced signs of blue sac disease (BSD) including heart deformities, yolk sac–pericardial edema, and cranial–skeletal deformities. In contrast, there was some variability in the sensitivity of the test methods for developmental endpoints for the sublethal evaluation of a stressor (NA mixture) that had no effect on hatch length and was a weak inducer of signs of BSD. Even though the sensitivity of the measurement endpoints were slightly more or less sensitive for the MP vs. SP depending on the mixture tested, the MP using agitation is recommended as a cost-effective and rapid alternative for screening the sublethal impact of toxicants on the early-life stages of fish.





Similar content being viewed by others
References
APHA (American Public Health Association) (1989) Toxicity test methods for aquatic organisms. In: Standard methods for the examination of water and wastewater, seventeenth edition. APHA, Washington, D.C., pp 8–143
Belanger SE, Cherry DS, Cairns J Jr (1990) Functional and pathological impairment of Japanese medaka (Oryzias latipes) by long-term asbestos exposure. Aquat Toxicol 17:133–154
Benoit DA, Holcombe GW, Spehar RL (1991) Guidelines for conducting early life stage toxicity tests with Japanese medaka (Oryzias latipes). EPA/600/3-91/063. Environmental Research Laboratory-Duluth, Duluth, Minnesota
Bentivegna CS, Piatkowski T (1998) Effects of tributyltin on medaka (Oryzias latipes) embryos at different stages of development. Aquat Toxicol 44:117–128
Billiard SM, Hodson PV, Bols NC (2000) Does the potency of polycyclic aromatic hydrocarbons (PAHs) for inducing CYP1A1 in juvenile trout (Oncorhynchus mykiss) predict dioxin-like toxicity in early life stages? Mar Environ Res 50:308–309
Chen C-M, Cooper KR (1999) Developmental toxicity and EROD induction in the Japanese medaka (Oryzias latipes) treated with dioxin congeners. Bull Environ Contam Toxicol 63:423–429
Chikae M, Hatano Y, Ikeda R, Morita Y, Hsan Q, Tamiya E (2004) Effects of bis(2-ethylhexyl) phthalate and benzo[a]pyrene on the embryos of Japanese medaka (Oyzias latipes). Environ Toxicol Pharmacol 16:141–145
Colavecchia MV, Backus SM, Hodson PV, Parrott JL (2004) Toxicity of oil sands to early life stages of fathead minnows (Pimephales promelas). Environ Toxicol Chem 23:1709–1718
Downing G, Litvak MK (2002) Effects of light intensity, spectral composition and photoperiod on development and hatching of haddock (Melanogrammus aeglefinus) embryos. Aquaculture 213:265–278
Geffen AJ (2002) Length of herring larvae in relation to age and time of hatching. J Fish Biol 60:479–485
Gonzalez-Doncel M, Villalobos SA, Okihiro MS, Hinton DE (2004) Does shaking alter the time of hatching in incubated medaka (Oryzias latipes) embryos? Bull Environ Contam Toxicol 73:364–370
Gormley KL, Teather KL (2003) Developmental, behavioral, and reproductive effects experienced by Japanese medaka (Oryzias latipes) in response to short-term exposure to endosulfan. Ecotoxicol Environ Saf 54:330–338
Gray MA, Metcalfe CD (1999) Toxicity of 4-tert-octylphenol to early life stages of Japanese medaka (Oryzias latipes). Aquat Toxicol 46:149–154
Hamm JT, Hinton DE (2000) The role of development and duration of exposure to the embryotoxicity of diazinon. Aquat Toxicol 48:403–418
Hamor T, Garside ET (1976) Developmental rates of embryos of Atlantic salmon, Salmo salar L., in response to various levels of temperature, dissolved oxygen, and water exchange. Can J Zool 54:1912–1917
Helmstetter MF, Alden RW III (1995) Toxic responses of Japanese medaka (Oryzias latipes) eggs following topical and immersion exposures to pentachlorophenol. Aquat Toxicol 32:15–29
Huff DD, Grad G, Williamson CE (2004) Environmental constraints on spawning depth of yellow perch: the roles of low temperature and high solar ultraviolet radiation. Trans Am Fish Soc 133:718–726
Incardona JP, Collier TK, Scholz NL (2004) Defects in cardiac function precede morphological abnormalities in fish exposed to polycyclic aromatic hydrocarbons. Toxicol App Pharmacol 196:191–205
Iwamatsu T (2004) Stages of normal development in the medaka Oryzias latipes. Mech Develop 121:605–618
Kaur R, Buckley B, Park SS, Kim YK, Cooper KR (1996) Toxicity test of Nanji Island Landfill (Seoul, Korea) leachate using Japanese medaka (Oryzias latipes) embryo larval assay. Bull Environ Contam Toxicol 57:84–90
Kestemont P, Jourdan S, Houbart M, Melard C, Paspatis M, Fontaine P, Cuvier A, Kentouri M, Baras E (2003) Size heterogeneity, cannibalism and competition in cultured predatory fish larvae: biotic and abiotic influences. Aquaculture 227:333–356
Kim Y, Cooper KR (1999) Toxicity of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) and polychlorinated biphenyls (PCBs) in the embryos and newly hatched larvae of the Japanese medaka (Oryzias latipes). Chemosphere 39:527–538
Kiparissis Y, Akhtar P, Hodson PV, Brown RS (2003) Partition-controlled delivery of toxicants: a novel in vivo approach for embryo toxicity testing. Environ Sci Technol 37:2262–2266
Kirchen RV, West WR (1976) The Japanese medaka: care and development. Carolina Biological Supply Company, North Carolina
Marty GD, Cech JJ Jr, Hinton DE (1990) Effect of incubation temperature on oxygen consumption and ammonia production by Japanese medaka, Oryzias latipes, eggs and newly hatched larvae. Environ Toxicol Chem 9:1397–1403
Metcalfe NB, Wright PJ, Thorpe JE (1992) Relationships between social status, otolith size at first feeding and subsequent growth in Atlantic salmon (Salmo salar). J Anim Ecol 61:585–589
Nero V, Farwell A, Lee LEJ, Van Meer T, MacKinnon MD, Dixon DG (2006) The effects of salinity on naphthenic acid toxicity to yellow perch: gill and liver histopathology. Ecotoxicol Environ Saf 65: 252–264
Newman JW, Denton DL, Morisseau C, Koger CS, Wheelock CE, Hinton DE, Hammock BD (2001) Evaluation of fish models of soluble epoxide hydrolase inhibition. Environ Health Perspect 109:61–66
Ojanguren AF, Reyes-Gavilan FG, Munoz RR (1999) Effects of temperature on growth and efficiency of yolk utilization in eggs and pre-feeding stages of Atlantic salmon. Aquacult Int 7:81–87
Oseid DM, Smith LL (1971) Survival and hatching of walleye eggs at various dissolved oxygen levels. Prog Fish Cult 33:81–85
Peters L (1999) The effects of oil sands aquatic reclamation on the early life stages of fish. MSc Thesis, University of Waterloo, Waterloo, Ontario
Pyle GG, Swanson SM, Lehmkuhl DM (2002) Toxicity of uranium mine receiving waters to early life stage fathead minnows (Pimephales promelas) in the laboratory. Environ Pollut 116:243–255
Rhodes SM, Farwell A, Hewitt LM, MacKinnon MD, Dixon DG (2005) The effects of dimethylated and alkylated polycyclic aromatic hydrocarbons on the embryonic development of the Japanese medaka. Ecotoxicol Environ Saf 60:247–258
Shi M, Faustman EM (1989) Development and characterization of a morphological scoring system for medaka (Oryzias latipes) embryo culture. Aquat Toxicol 15:127–140
Spencer HB, Hussein WR, Tchounwou PB (2002) Effects of tetrachloroethylene on the viability and development of embryos of the Japanese medaka, Oryzias latipes. Arch Environ Contam Toxicol 42:463–469
Teather KL, Boswell J, Gray MA (2000) Early life-history parameters of Japanese medaka (Oryzias latipes). Copeia 3:813–818
Teather K, Harris M, Boswell J, Gray M (2001) Effects of Acrobat MZ® and Tattoo C® on Japanese medaka (Oryzias latipes) development and adult male behavior. Aquat Toxicol 51:419–430
USEPA (United States Environmental Protection Agency) (1998) Method 3550B–Ultrasonic extraction—SW-846, test methods for evaluating solid waste, physical chemical methods, fifth revision. United States Environmental Protection Agency, Office of Solid Waste, Washington, DC
Villalobos SA, Hamm JT, Teh SJ, Hinton DE (2000) Thiobencarb-induced embryotoxicity in medaka (Oryzias latipes): stage-specific toxicity and the protective role of chorion. Aquat Toxicol 48:309–326
Wisk JD, Cooper KR (1990) The stage specific toxicity of 2,3,7,8- tetrachlorodibenzo-p-dioxin in embryos of the Japanese medaka (Oryzias latipes). Environ Toxicol Chem 9:1159–1169
Yang Z, Yang JX (2004) Effect of photoperiod on the embryonic development of obscure puffer (Takifugu obscurus). J Freshwat Ecol 19:53–58
Acknowledgments
Funding was provided by the Canadian Water Network (CWN) and by an NSERC Discovery Grant to DGD. Special thanks to Spencer Rhodes, for the preparation of the PAH mixture, and Mark Lampi, for the determination of light intensities. We also thank the many students who assisted with the maintenance of the medaka culture.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farwell, A., Nero, V., Croft, M. et al. Modified Japanese Medaka Embryo-Larval Bioassay for Rapid Determination of Developmental Abnormalities. Arch Environ Contam Toxicol 51, 600–607 (2006). https://doi.org/10.1007/s00244-005-0319-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-005-0319-x