Abstract
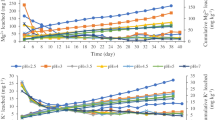
Acid rain is a serious environmental problem in the world and is of a particular concern in southern China where most of the soils are acidic. This study investigated the dynamics of cations, phosphorus (P), and soil organic matter (SOM) in the Latosol (acidic red soil) from south China under the influences of simulated acid rain (SAR). Laboratory experiments were performed by leaching the soil columns with SAR at pH levels ranging from 2.5 to 7.0 over a 21-day experimental period. Results show that about 34, 46, 20, and 77% of the original exchangeable soil Ca+2, Mg+2, K+, and Na+, respectively, were leached out by the SAR at pH 2.5 after 21 days. Two distinct patterns of the available phosphorus (AP) concentrations were observed: one at pH≤3.5 and the other at pH ≥ 4.0. At pH≤3.5, concentrations of the AP increased from the beginning of the experiments to day 5, then decreased from day 5 to 15, and finally increased from day 15 to the end of the experiments. At pH ≥ 4.0, concentrations of the AP increased consecutively from the beginning of the experiments to day 10 and decreased from day 10 to the end of the experiments. Such a finding is useful for agricultural practices since soil P is one of the most important macronutrients for plant growth. In general, SOM content decreased with time as the Latosol was leached by the SAR at all pH levels. A maximum concentration of soil fulvic acid was found after 15 days of the experiments due to the degradation of the SOM. A multiple regression analysis showed that a very strong relationship was obtained between the soil AP and the other three parameters (i.e., pH, SOM, and sorption P).






Similar content being viewed by others
References
ACSSSC (Agricultural Committee of Soil Science Society of China) (1983) Standard analysis method of soil agricultural chemicals. Science and Technology Publisher, Beijing, pp 179–190 (in Chinese)
Brady (1984) The nature and properties of soils. 9th edition. New York, London
Calace N, Fiorentini F, Petronio BM, Pietroletti M (2001) Effects of acid rain on soil humic compounds. Talanta 54:837–846
Duan L, Hao J, Xie SD, Zhou DP (2002) Determining weathering rates of soils in China. Geoderma 110:205–225
Hodson ME, Landan SJ (1999) A long-term soil leaching column experiment investigating the effect of variable sulphate loads on soil solution and soil drainage chemistry. Environ Pollut 104:11–19
Guo YL (2002) Acid rain problem and prevention in China. J Shanxi Finance and Economics University 24:106 (in Chinese)
Ivring PM (1983) Acidic precipitation effects on crops. A review and analysis of research. J Environ Qual 12:442–453
Jackson DR, Garrett BC, Bishop TA (1984) Comparison of batch and column methods for assessing leachability of hazardous waste. Envlron Sei Technol 18:668–673
Liao B, Seip HM, Larssen T (1997) Response of two Chinese forested soils to acidic inputs: leaching experiment. Geoderma 75:53–73
Liu KH, Mansell RS, Rhue RD (1990) Cation removal during application of acid solution into air dry soil columns. Soil Sci Soc Am J S4:1747–1753u
Menz FC, Seip HM (2004) Acid rain in Europe and the United States: an update. Environ Sci Policy 7:253–265
NSICSA (Nanjing Soil Institute of China Science Academy) (1978) Soil physical and chemical analysis. Science and Technology Publisher, Shanghai. pp 105–136 (in Chinese)
Sibanda HM, Yound SD (1988) Competitive adsorption of humus acids and P on goethite, gibbsite and two tropical soils. Soil Sci 37:197–204
US-EPA (2004) Acid rain program, 2003 progress report. Clean Air Markets Division, Office of Air and Radiation, U.S. Environmental Protection Agency, EPA 430-R-04-009
Yan CS (1988) Soil fertility study method (in Chinese). Agricultural Publisher, Beijing, pp 201–202
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ling, DJ., Zhang, JE., Ouyang, Y. et al. Role of Simulated Acid Rain on Cations, Phosphorus, and Organic Matter Dynamics in Latosol. Arch Environ Contam Toxicol 52, 16–21 (2007). https://doi.org/10.1007/s00244-006-0004-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-006-0004-8