Abstract
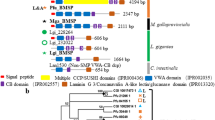
Shell matrix proteins (SMPs) that are embedded within calcified layers of mollusc shells are believed to play an essential role in controlling the biomineral synthesis and in increasing its mechanical properties. Among the wide diversity of mollusc shell textures, nacro-prismatic shells represent a tremendous opportunity for the investigation of the SMP evolution. Indeed, nacro-prismatic texture appears early in Cambrian molluscs and is still present in the shell of some bivalves, gastropods, cephalopods and very likely also, of some monoplacophorans. One key question is to know whether these shells are constructed from similar matrix protein assemblages, i.e. whether they share a common origin. Most of the molecular data published so far are restricted to two genera, the bivalve Pinctada and the gastropod Haliotis. The shell protein content of these two genera are clearly different, suggesting independent origins or considerable genetic drift from a common ancestor. In order to describe putatively conserved mollusc shell proteins, here we have investigated the SMP set of a new bivalve model belonging to another genera, the edible mussel Mytilus, using an up-to-date proteomic approach based on the interrogation of more than 70,000 EST sequences, recently available from NCBI public databases. We describe nine novel SMPs, among which three are completely novel, four are homologues of Pinctada SMPs and two are very likely homologues of Haliotis SMPs. This latter result constitutes the first report of conserved SMPs between bivalves and gastropods. More generally, our data suggest that mollusc SMP set may follow a mosaic pattern within the different mollusc models (Mytilus, Pinctada, Haliotis). We discuss the function of such proteins in calcifying matrices, the molecular evolution of SMP genes and the origin of mollusc nacro-prismatic SMPs.







Similar content being viewed by others
References
Adamkewicz SL, Harasewych MG, Blake J, Saudek D, Bult CJ (1997) A molecular phylogeny of the bivalve molluscs. Mol Biol Evol 14:619–629
Addadi L, Joester D, Nudelman F, Weiner S (2006) Mollusk shell formation: a source of new concepts for understanding biomineralization processes. Chem Rev 12:980–987
Atlan G, Balmain N, Berland S, Vidal B, Lopez E (1997) Reconstruction of human maxillary defects with nacre powder: histological evidence for bone regeneration. C R Acad Sci Paris 320:253–258
Auzoux-Bordenave S, Badou A, Gaume B, Berland S, Helléouet MN, Milet C, Huchette S (2010) Ultrastructure, chemistry and mineralogy of the growing shell of the European abalone Haliotis tuberculata. J Struct Biol 171:277–290
Bédouet L, Schuller J, Marin F, Milet C, Lopez E, Giraud M (2001) Soluble proteins of the nacre of the giant oyster Pinctada maxima and of the abalone Haliotis tuberculata: extraction and partial analysis of nacre proteins. Comp Biochem Physiol B 128:389–400
Berthelat F (2010) Nacre from mollusk shell: a model for high-performance structural materials. Bioinspir Biomin 20:035001
Bielefeld U, Zierold K, Körje KH, Becker W (1992) Calcium localization in the shell-forming tissue of the freshwater snail, Biomphalaria glabrata: a comparative study of various methods for localizing calcium. Histochem J 24:927–938
Bøggild O (1930) The shell structure of the mollusks. K Dan Vidensk Selsk Skr Naturvidensk Math Afd 9:233–326
Carter JG (1990) Skeletal biomineralization: patterns, processes and evolutionary trends. Van Nostrand Reinhold, New York
Chateigner D, Hedegaard C, Wenk HR (2000) Mollusc shell microstructures and crystallographic textures. J Struct Geol 22:1723–1735
Chateigner D, Ouhenia S, Krauss C, Hedegaard C, Gil O, Morales M, Lutterotti L, Rousseau M, Lopez E (2010) Voyaging around nacre with the X-ray shuttle: from bio-mineralisation to prosthetics via mollusc phylogeny. Mater Sci Eng A 528:37–51
Checa AG, Rodriguez-Navarro AB, Esteban-Delgado JF (2005) The nature and formation of calcitic columnar prismatic shell layers in pteriomorphian bivalves. Biomaterials 26:6404–6414
Checa AG, Cartwright JH, Willinger MG (2009a) The key role of the surface membrane in why gastropod nacre grows in towers. Proc Nat Acad Sci USA 106:38–43
Checa A, Ramirez-Rico J, Gonzalez-Segura A, Sanchez-Navas A (2009b) Nacre and false-nacre (foliated aragonite) in extant monoplacophorans (= Tryblidiida: Mollusca). Naturwissenschaften 96:111–122
Craft JA, Gilbert JA, Temperton B, Dempsey KE, Ashelford K, Tiwari B, Hutchinson TH, Chipman JK (2010) Pyrosequencing of Mytilus galloprovincialis cDNAs: tissue-specific expression patterns. PLoS One 5:e8875
Crenshaw MA (1972) The soluble matrix from Mercenaria mercenaria shell. Biomineralization 6:6–11
Dalbeck P, England J, Cusack M, Lee MR, Fallick AE (2008) Crystallographic and chemistry of the calcium carbonate polymorph switch in M. edulis shells. Eur J Mineral 18:601–609
Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, Chevenet F, Dufayard J-F, Guindon S, Lefort V, Lescot M, Claverie J-M, Gascuel O (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acid Res 36:465–469
Falini G, Albeck S, Weiner S, Addadi L (1996) Control of aragonite and calcite polymorphism by mollusk shell macromolecules. Science 271:67–69
Feng W, Sun W (2003) Phosphate replicated and replaced microstructure of molluscan shells from the earliest Cambrian in China. Acta Palaeontol Pol 48:21–30
Feng QL, Li HB, Pu G, Zhang M, Cui FZ, Li D (2000) Crystallographic alignment of calcite prisms in the oblique prismatic layer of the Mytilus edulis shell. J Mater Sci 35:3337–3340
Gazeau F, Gattuso J-P, Pronker E, Peene J, Heip CH, Middelburg JJ (2010) Effect of ocean acidification on the early stages of blue mussel Mytilus edulis. Biogeosciences 7:2051–2060
Gilbert PU, Metzler RA, Zhou D, Scholl A, Doran A, Young A, Kunz M, Tamura N, Coppersmith SN (2008) Gradual ordering in red abalone nacre. J Am Chem Soc 130:17519–17527
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Hattan SJ, Laue TM, Chasteen ND (2001) Purification and characterization of a novel calcium-binding protein from the extrapallial fluid of the mollusc, Mytilus edulis. J Biol Chem 276:4461–4468
Inoue N, Ishibashi R, Ishikawa T, Atsumi T, Aoki H, Komura A (2010) Gene expression patterns and pearl formation in the Japanese pearl oyster (Pinctada fucata): a comparison of gene expression patterns between the pearl sac and the mantles tissues. Aquaculture 308:S68–S74
Jackson AP, Vincent JV, Turner RM (1988) The mechanical design of nacre. Proc R Soc Lond 234:415–440
Jackson DJ, McDougall C, Green KM, Simpson F, Wörheide G, Degnan BM (2006) A rapidly evolving secretome builds and patterns a sea shell. BMC Biol 4:40
Jackson DJ, Wörheide G, Degnan BM (2007a) Dynamic expression of ancient and novel molluscan shell genes during ecological transitions. BMC Evol Biol 7:160
Jackson DJ, Macis L, Reitner J, Degnan BM, Wörheide G (2007b) Sponge paleogenomics reveals an ancient role for carbonic anhydrase in skeletogenesis. Science 316:1893–1895
Jackson DJ, McDougall C, Woodcroft B, Moase P, Rose RA, Kube M, Reinhardt R, Rokshar DS, Montagnani C, Joubert C, Piquemal D, Degnan BM (2010) Parallel evolution of nacre building gene sets in molluscs. Mol Biol Evol 27:591–608
Joubert C, Piquemal D, Marie B, Manchon L, Pierrat F, Zanella-Cléon I, Cochennec-Laureau N, Gueguen Y, Montagnani C (2010) Transcriptome and proteome analysis of Pinctada margaritifera calcifying mantle and shell: focus on biomineralization. BMC Genomics 11:613
Keith J, Stockwell S, Ball D, Remillard K, Kaplan D, Thanhauser T, Sherwood R (1993) Comparative analysis of macromolecules in mollusc shells. Comp Biochem Physiol B 105:487–496
Kono M, Hayashi N, Samata T (2000) Molecular mechanism of the nacreous layer formation in Pinctada maxima. Biochem Biophys Res Comm 269:213–218
Levi-Kalisman Y, Falini G, Addadi L, Weiner S (2001) Structure of the nacreous organic matrix of a bivalve mollusk shell examined in the hydrated state using cryo-TEM. J Struct Biol 135:8–17
Lin A, Meyers MA (2005) Growth and structure in abalone shell. Mater Sci Eng 390:27–41
Liu HL, Liu SF, Ge YJ, Liu J, Wang XY, Xie LP, Zhang RQ, Wang Z (2007) Identification and characterization of a biomineralization related gene PFMG1 highly expressed in the mantle of Pinctada fucata. Biochemistry 46:844–851
Lowenstam HA, Weiner S (1989) On biomineralization. Oxford University Press, New York
Mamangkey NG, Southgate PC (2009) Regeneration of excised mantle tissue by the silver-lip pearl oyster, Pinctada maxima (Jameson). Fish Shellfish Immun 27:164–174
Mann S (1988) Molecular recognition in biomineralization. Nature 332:119–124
Mann S (2001) Biomineralization: principles and concepts in bioinorganic materials chemistry. Oxford University Press, New York
Mann K, Weiss IM, André S, Gabius HJ, Fritz M (2000) The amino acid sequence of the abalone (Haliotis laevigata) nacre protein perlucin. Eur J Biochem 267:5257–5264
Mann K, Poustka AJ, Mann M (2008a) The sea urchin (Strongylocentrotus purpuratus) test and spine proteomes. Proteome Sci 6:22
Mann K, Poustka AJ, Mann M (2008b) In-depth, high-accuracy proteomics of sea urchin tooth organic matrix. Proteome Sci 6:33
Marie B, Luquet G, Pais de Barros J-P, Guichard N, Médakovic D, Marin F (2007) The shell matrix of the freshwater mussel Unio pictorum (paleoheterodonta, unionoida): involvement of acidic polysaccharides from glycoproteins in nacre mineralization. FEBS J 274:2933–2945
Marie B, Luquet G, Bédouet L, Milet C, Guichard N, Medakovic D, Marin F (2008) Nacre calcification in the freshwater mussel Unio pictorum: carbonic anhydrase activity and purification of a 95 kDa calcium-binding glycoprotein. ChemBioChem 9:2515–2523
Marie B, Marin F, Marie A, Bédouet L, Dubost L, Alcaraz G, Milet C, Luquet G (2009a) Evolution of nacre: biochemistry and proteomics of the shell organic matrix of the cephalopod Nautilus macromphalus. ChemBioChem 10:1495–1506
Marie B, Le Roy N, Marie A, Dubost L, Milet C, Bédouet L, Becchi M, Zanella-Cleon I, Jackson DJ, Degnan BM, Luquet G, Marin F (2009b) Nacre evolution: a proteomic approach. Mater Res Soc Symp Proc 1187:KK01–KK03
Marie B, Zanella-Cléon I, Le Roy N, Becchi M, Luquet G, Marin F (2010a) Proteomic analysis of the acid-soluble nacre matrix of the bivalve Unio pictorum: detection of novel carbonic anhydrase and putative protease inhibitor proteins. ChemBioChem 11:2138–2147
Marie B, Marie A, Jackson DJ, Dubost L, Degnan BM, Milet C, Marin F (2010b) Proteomic analysis of the organic matrix of the abalone Haliotis asinina calcified shell. Proteome Sci 8:54
Marie B, Zanella-Cléon I, Guichard N, Becchi M, Marin F (2011) Novel proteins from the calcifying shel matrix of the pacific oyster Crassostrea gigas. Mar Biotechnol. doi:10.1007/s10126-011-9379-2
Marin F, Amons R, Guichard N, Stiger M, Hecker A, Luquet G, Layrolle P, Alcaraz G, Riondet C, Woestbroek P (2005) Caspartin and Calprismin, two proteins of the shell calcitic prisms of the mediterranean fan mussel Pinna nobilis. J Biol Chem 280:33895–33908
Marin F, Pokroy B, Luquet G, Layrolle P, De Groot K (2007) Protein mapping of calcium carbonate biominerals by immunogold. Biomaterials 28:2368–2377
Marin F, Luquet G, Marie B, Medakovic D (2008) Molluscan shell proteins: primary structure, origin, and evolution. Curr Top Dev Biol 80:209–276
Medakovic D (2000) Carbonic anhydrase activity and biomineralization process in embryos, larvae and adult blue mussels Mytilus edulis. Hegol Mar Res 54:1–6
Miller AW, Reynolds AC, Sobrino C, Riedel GF (2009) Shellfish face uncertain future in high CO2 world: influence of acidification on oyster larvae calcification and growth in estuaries. PLoS One 4:e5661
Miyamoto H, Miyashita T, Okushima M, Nakano S, Morita T, Matsushiro A (1996) A carbonic anhydrase from the nacreous layer in oyster pearls. Proc Natl Acad Sci USA 93:9657–9660
Miyamoto H, Yano M, Miyashita T (2003) Similarities in the structure of nacrein, the shell-matrix protein, in a bivalve and a gastropod. J Moll Stud 69:87–89
Miyazaki Y, Usui T, Kajikawa A, Hishiyama H, Matsuzawa N, Nishida T, Machii A, Samata T (2008) Daily oscillation of gene expression associated with nacreous layer formation. Front Mater Sci China 2:162–166
Mutvei H (1980) The nacreous layer in molluscan shells. In: Omori M, Watabe N (eds) The mechanisms of mineralization in biological systems. Tokay University press, Tokyo, pp 49–56
Nakahara H (1991) Nacre formation in bivalve and gastropod molluscs. In: Suga S, Nakahara H (eds) Mechanisms and phylogeny of mineralization in biological systems. Springer-Verlag, New York, pp 343–350
Notredame C, Higgins DG, Heringa J (2000) T-Coffee: a novel method for fast and accurate multiple sequence alignment. J Mol Biol 302:205–217
Nudelman F, Shimoni E, Klein E, Rousseau M, Bourrat X, Lopez E, Addadi L, Weiner S (2008) Forming nacreous layer of the shells of the bivalves Atrina rigida and Pinctada margaritifera: an environmental- and cryo-scanning electron microscopy study. J Struct Biol 162:290–300
Ponder W, Lindberg DR (2008) Phylogeny and evolution of the mollusca. University of California Press, Berkeley
Rahman AM, Isa Y, Uehara T (2005) Proteins of calcified endoskeleton: II. Partial amino acid sequences of endoskeletal proteins and the characterization of proteinaceous organic matrix of spicules from the alcyonarian, Sinularia polydactyla. Proteomics 5:885–893
Rousseau M, Lopez E, Stempflé P, Brendlé M, Franke L, Guette A, Naslain R, Bourrat X (2005) Multiscale structure of sheet nacre. Biomaterials 26:6254–6262
Samata T, Hayashi N, Kono M, Hasegawa K, Horita C, Akera S (1999) A new matrix protein family related to the nacreous layer formation of Pinctada fucata. FEBS Lett 462:225–229
Smith-Keune C, Jerry DR (2009) High level of intra-specific variation in the NG repeat region of the Pinctada maxima N66 organic matrix protein. Aquat Res 40:1054–1063
Sudo S, Fujikawa T, Nagakura T, Ohkubo T, Sakaguchi K, Tanaka M, Nakashima K, Takahashi T (1997) Structure of mollusc shell framework proteins. Nature 387:563–564
Suzuki M, Saruwatari K, Kogure T, Yamamoto Y, Nishimura T, Kato T, Nagasawa H (2009) An acidic matrix protein, Pif, is a key macromolecules for nacre formation. Science 325:1388–1390
Tambutté S, Tambutté E, Zoccola D, Caminiti N, Lotto S, Moya A, Allemand D, Adkins J (2007) Characterization and role of carbonic anhydrase in the calcification process of the azooxanthellate coral Tubastrea aurea. Mar Biol 151:71–83
Tanguy A, Bierne N, Saavedra C, Pina B, Bachère E, Kube M, Bazin E, Bonhomme F, Boudry P, Boulo V, Boutet I, Cancela L, Dossat C, Favrel P, Huvet A, Jarque S, Jollivet D, Klages S, Lapègue S, Leite R, Moal J, Moraga D, Reinhardt R, Samain J-F, Zouros E, Canario A (2008) Increasing genomic information in bivalves through new EST collections in four species: development of new genetic markers for environmental studies and genome evolution. Gene 408:27–36
Treccani L, Mann K, Heinemann F, Fritz M (2006) Perlwapin, an abalone nacre protein with three four-disulfide core (whey acidic protein) domains, inhibits the growth of calcium carbonate crystals. Biophys J 91:2601–2608
Vendrasco MJ, Porter SM, Kouchinsky A, Li G, Fernandez CZ (2010) New data on molluscs and their shell microstructures from the Middle Cambrian Gowers Formation, Australia. Palaeontology 53:97–135
Vernier P, De Pitta C, Bernante F, Varotto L, De Nardi B, Bovo G, Roch P, Novoa B, Figueras A, Pallavicini A, Lanfranchi G (2009) Mytibase: a knowledgebase of mussel (M. galloprovincialis) transcribed sequences. BMC Genomics 10:72
Wang A, Wang Y, Gu Z, Li S, Shi Y, Guo X (2010) Development of expressed sequence tags from the pearl oyster, Pinctada martensi Dunker. Mar Biotechnol. doi:10.1007/s10126-010-9296-9
Weiner S (1983) Mollusk shell formation: isolation of two organic matrix proteins associated with calcite deposition in the bivalve Mytilus californianus. Biochemistry 22:4139–4145
Weiner S, Traub W (1984) Macromolecules in mollusc shells and their functions in biomineralization. Philos Trans R Soc Lond 304:425–434
Weiner S, Lowenstam HA, Hood L (1977) Discrete molecular weight components of the organic matrices of mollusc shells. J Exp Mar Biol Ecol 30:45–51
Weiss I (2010) Jewels in Pearl. ChemBioChem 11:297–300
Weiss I, Schönitzer V (2006) The distribution of chitin in larval shells of the bivalve mollusk Mytilus galloprovincialis. J Struct Biol 153:264–277
Weiss IM, Göhring W, Fritz M, Mann K (2001) Perlustrin, a Haliotis laevigata (abalone) nacre protein, is homologous to the insulin-like growth factor binding protein N-terminal module of vertebrates. Biochem Biophys Res Commun 285:244–249
Westbroek P, Marin F (1998) A marriage of bone and nacre. Nature 392:861–862
Wheeler AP, George JW, Evans CA (1981) Control of CaCO3 nucleation and crystal growth by soluble matrix of oyster shell. Science 212:1397–1398
Wilbur KM, Jodrey LH (1955) Studies on shell formation. V. The inhibition of shell formation by carbonic anhydrase inhibitors. Biol Bull 108:359–365
Yin Y, Hang J, Paine ML, Reinhold VN, Chasteen D (2005) Structural characterisation of the major extrapallial fluid protein of the mollusc Mytilus edulis: implication for function. Biochemistry 44:10720–10731
Acknowledgments
The work of BM, NLR and FM is financially supported by an ANR (ACCRO-EARTH, ref. BLAN06-2_159971, coordinator Gilles Ramstein, LSCE) during the period 2007–2011. The “Conseil Régional de Bourgogne” (Dijon, France) provided additional supports for the acquisition of new equipment in the Biogeosciences research unit (UMR CNRS 5561). A complementary financial support was provided by INSU (Action INTERRVIE 2010). BM would like to thanks Davorin Medakovic for providing the fresh shells of Mytilus galloprovincialis and Jérome Thomas for handling shell pictures. The present protein sequences appear in the UniProtKB under the accession numbers P86853–P86861.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Marie, B., Le Roy, N., Zanella-Cléon, I. et al. Molecular Evolution of Mollusc Shell Proteins: Insights from Proteomic Analysis of the Edible Mussel Mytilus . J Mol Evol 72, 531–546 (2011). https://doi.org/10.1007/s00239-011-9451-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-011-9451-6