Abstract
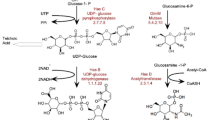
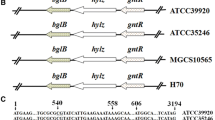
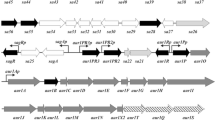
Hyaluronic acid (HA) is a ubiquitous linear polysaccharide in vertebrates and also is the capsule material of some pathogenic bacteria including group A and C streptococci. In bacteria, the HA synthase occurs in an operon (has) coding for enzymes involved in the production of HA precursors. We report two new members of the has operon family from Streptococcus equi subsp. zooepidemicus (S. zooepidemicus) and Streptococcus equi subsp. equi (S. equi). The has operon of S. zooepidemicus contains, in order, hasA, hasB, hasC, glum, and pgi, whereas these genes are separated on two operons in S. equi (hasA, hasB, hasC and hasC, glmU, pgi). The transcription start site and a σ70 promoter were experimentally identified 50 bp upstream of hasA in S. zooepidemicus. We performed a phylogenetic analysis of each of the has operon genes to determine the evolutionary origin(s) of the streptococcal has operon. In contrast to other capsular and exopolysaccharide operons, has operons have undergone no detectable interspecies lateral gene transfers in their construction, instead relying on intragenome gene duplication for their assembly. Specifically, hasC and glmU appear to have been duplicated into the S. zooepidemicus has operon from remotely located but near-identical paralogues most likely to improve HA productivity by gene dosage in this streptococcus. The intragene rearrangements appear to be ongoing events and the two has operons of the S. equi subspecies represent two alternatives of the same gene arrangement. A scenario for the evolution of streptococcal has operons is proposed.




Similar content being viewed by others
Abbreviations
- hasA :
-
Hyaluronate synthase
- hasB :
-
UDP-glucose dehydrogenase
- hasC :
-
glucose-1-phosphate uridyltransferase
- glmU :
-
N-acetyl-glucosamine-1-phosphate uridyltransferase
- pgi :
-
glucose-6-phosphate isomerase
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Anderson DG, McKay LL (1984) Genetic and physical characterization of recombinant plasmids associated with cell aggregation and high-frequency conjugal transfer in Streptococcus lactis ML3. J Bacteriol 158:954–962
Arrecubieta C, Lopez R, Garcia E (1994) Molecular characterization of cap3A, a gene from the operon required for the synthesis of the capsule of Streptococcus pneumoniae type 3: sequencing of mutations responsible for the unencapsulated phenotype and localization of the capsular cluster on the pneumococcal chromosome. J Bacteriol 176:6375–6383
Ashbaugh CD, Alberti S, Wessels MR (1998) Molecular analysis of the capsule gene region of group A streptococcus: the hasAB genes are sufficient for capsule expression. J Bacteriol 180:4955–4959
Bentley SD, Aanensen DM, Mavroidi A, Saunders D, Rabbinowitsch E, Collins M, Donohoe K, Harris D, Murphy L, Quail MA, Samuel G, Skovsted IC, Kaltoft MS, Barrell B, Reeves PR, Parkhill J, Spratt BG (2006) Genetic analysis of the capsular biosynthetic locus from all 90 pneumococcal serotypes. PLoS Genet 2:e31
Blank LM, McLaughlin RL, Nielsen LK (2005) Stable production of hyaluronic acid in Streptococcus zooepidemicus chemostats operated at high dilution rate. Biotechnol Bioeng 90:685–693
Bourgoin F, Pluvinet A, Gintz B, Decaris B, Guedon G (1999) Are horizontal transfers involved in the evolution of the Streptococcus thermophilus exopolysaccharide synthesis loci? Gene 233:151–161
Cartee RT, Forsee WT, Schutzbach JS, Yother J (2000) Mechanism of type 3 capsular polysaccharide synthesis in Streptococcus pneumoniae. J Biol Chem 275:3907–3914
Chaffin DO, McKinnon K, Rubens CE (2002) CpsK of Streptococcus agalactiae exhibits alpha2,3-sialyltransferase activity in Haemophilus ducreyi. Mol Microbiol 45:109–122
Chong BF, Blank LM, McLaughlin R, Nielsen LK (2005) Microbial hyaluronic acid production. Appl Microbiol Biotechnol 66:341–351
DeAngelis PL, Papaconstantinou J, Weigel PH (1993) Molecular cloning, identification, and sequence of the hyaluronan synthase gene from group A Streptococcus pyogenes. J Biol Chem 268:19181–19184
De Vuyst L, De Vin F, Vaningelgem F, Degeest B (2001) Recent developments in the biosynthesis and applications of heteropolysaccharides from lactic acid bacteria. Int Dairy J 11:687–707
Doolittle WF (1999) Phylogenetic classification and the universal tree. Science 284:2124–2129
Dougherty BA, van de Rijn I (1994) Molecular characterization of hasA from an operon required for hyaluronic acid synthesis in group A streptococci. J Biol Chem 269:169–175
Fitch WM (1970) Distinguishing homologous from analogous proteins. Syst Zool 19:99–113
Garcia E, Llull D, Munoz R, Mollerach M, Lopez R (2000) Current trends in capsular polysaccharide biosynthesis of Streptococcus pneumoniae. Res Microbiol 151:429–435
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17:754–755
Kamst E, Spaink PH (1999) Functional domains in the chitin oligosaccharide synthase NodC and related beta-polysaccharide synthases. Trends Glycosci Glycotechnol 11:187–199
Kumari K, Weigel PH (1997) Molecular cloning, expression, and characterization of the authentic hyaluronan synthase from group C Streptococcus equisimilis. J Biol Chem 272:32539–32546
Lander ES, Linton LM, Birren B et al (2001) Initial sequencing and analysis of the human genome. Nature 409:860–921
Lee JY, Spicer AP (2000) Hyaluronan: a multifunctional, megaDalton, stealth molecule. Curr Opin Cell Biol 12:581–586
Ludwig W, Strunk O, Westram R, Richter L, Meier H, Yadhukumar, Buchner A, Lai T, Steppi S, Jobb G, Forster W, Brettske I, Gerber S, Ginhart AW, Gross O, Grumann S, Hermann S, Jost R, Konig A, Liss T, Lussmann R, May M, Nonhoff B, Reichel B, Strehlow R, Stamatakis A, Stuckmann N, Vilbig A, Lenke M, Ludwig T, Bode A, Schleifer KH (2004) ARB: a software environment for sequence data. Nucleic Acids Res 32:1363–1371
Mavroidi A, Godoy D, Aanensen DM, Robinson DA, Hollingshead SK, Spratt BG (2004) Evolutionary genetics of the capsular locus of serogroup 6 pneumococci. J Bacteriol 186:8181–8192
Notredame C, Higgins DG, Heringa J (2000) T-Coffee: a novel method for fast and accurate multiple sequence alignment. J Mol Biol 302:205–217
Okinaka RT, Cloud K, Hampton O, Hoffmaster AR, Hill KK, Keim P, Koehler TM, Lamke G, Kumano S, Mahillon J, Manter D, Martinez Y, Ricke D, Svensson R, Jackson PJ (1999) Sequence and organization of pXO1, the large Bacillus anthracis plasmid harboring the anthrax toxin genes. J Bacteriol 181:6509–6515
Rogers MB, Watkins RF, Harper JT, Durnford DG, Gray MW, Keeling PJ (2007) A complex and punctate distribution of three eukaryotic genes derived by lateral gene transfer. BMC Evol Biol 7:89
Salzberg SL, White O, Peterson J, Eisen JA (2001) Microbial genes in the human genome: lateral transfer or gene loss? Science 292:1903–1906
Siebert PD, Chenchik A, Kellogg DE, Lukyanov KA, Lukyanov SA (1995) An improved PCR method for walking in uncloned genomic DNA. Nucleic Acids Res 23:1087–1088
Spicer AP, McDonald JA (1998) Characterization and molecular evolution of a vertebrate hyaluronan synthase gene family. J Biol Chem 273:1923–1932
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tlapak-Simmons VL, Baggenstoss BA, Clyne T, Weigel PH (1999) Purification and lipid dependence of the recombinant hyaluronan synthases from Streptococcus pyogenes and Streptococcus equisimilis. J Biol Chem 274:4239–4245
Ward PN, Field TR, Ditcham WG, Maguin E, Leigh JA (2001) Identification and disruption of two discrete loci encoding hyaluronic acid capsule biosynthesis genes hasA, hasB, and hasC in Streptococcus uberis. Infect Immun 69:392–399
Wibawan IW, Pasaribu FH, Utama IH, Abdulmawjood A, Lammler C (1999) The role of hyaluronic acid capsular material of Streptococcus equi subsp. zooepidemicus in mediating adherence to HeLa cells and in resisting phagocytosis. Res Vet Sci 67:131–135
Woese CR (1987) Bacterial evolution. Microbiol Rev 51:221–271
Acknowledgment
This work was supported by Grant A89937110 from the Australian Research Council.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Blank, L.M., Hugenholtz, P. & Nielsen, L.K. Evolution of the Hyaluronic Acid Synthesis (has) Operon in Streptococcus zooepidemicus and Other Pathogenic Streptococci. J Mol Evol 67, 13–22 (2008). https://doi.org/10.1007/s00239-008-9117-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-008-9117-1