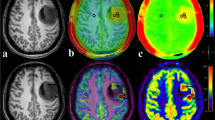
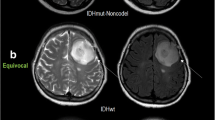
Abstract
Purpose
To noninvasively evaluate the value of three-dimensional pseudo-continuous arterial spin labeling (3D pCASL) and diffusion-weighted imaging (DWI) in diffuse gliomas grading as well as isocitrate dehydrogenase (IDH) 1 mutation status.
Methods
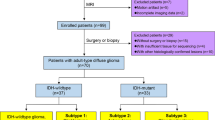
Fifty-six patients with pathologically confirmed diffuse gliomas with preoperative 3D pCASL and DWI were enrolled in this study. The Student’s t test and Mann-Whitney U test were used to evaluate differences in parameters of DWI and 3D pCASL between low and high grade as well as between mutant and wild-type IDH1 diffuse gliomas; receiver operator characteristic (ROC) analysis was used to assess the diagnostic performance. Subsequently, a multivariate stepwise logistic regression analysis was used to identify the independent parameters. Besides, Kruskal-Wallis H test was used to examine the differences among grades II, III, and IV diffuse gliomas.
Results
All parameters but CBFmean showed significant differences between low- and high-grade diffuse gliomas. In ROC analysis, the AUC of CBFmax, rCBFmean, rCBFmax, ADCmean, and ADCmin were 0.701, 0.730, 0.746, 0.810, and 0.856 respectively. Only the value of ADCmin was identified as the independent parameter in the differentiation of low- from high-grade diffuse gliomas. All parameters but CBFmean showed significant differences among the three grades. And the values of CBFmean, CBFmax, rCBFmean, and ADCmean showed significant differences between mutant and wild-type IDH1 in grade II–III astrocytoma.
Conclusion
Both 3D pCASL and DWI could be useful tools for distinguishing low- from high-grade diffuse gliomas and have the potential to differentiate different IDH1 mutation statuses of astrocytoma.





Similar content being viewed by others
References
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131(6):803–820. https://doi.org/10.1007/s00401-016-1545-1
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114(2):97–109. https://doi.org/10.1007/s00401-007-0243-4
Nabors LB, Portnow J, Ammirati M, Baehring J, Brem H, Brown P, Butowski N, Chamberlain MC, Fenstermaker RA, Friedman A, Gilbert MR, Hattangadi-Gluth J, Holdhoff M, Junck L, Kaley T, Lawson R, Loeffler JS, Lovely MP, Moots PL, Mrugala MM, Newton HB, Parney I, Raizer JJ, Recht L, Shonka N, Shrieve DC, Sills AK Jr, Swinnen LJ, Tran D, Tran N, Vrionis FD, Weiss S, Wen PY, McMillian N, Engh AM (2015) Central nervous system cancers, version 1.2015. J Natl Compr Cancer Netw 13(10):1191–1202
Hervey-Jumper SL, Berger MS (2016) Maximizing safe resection of low- and high-grade glioma. J Neuro-Oncol 130(2):269–282. https://doi.org/10.1007/s11060-016-2110-4
Weller M, van den Bent M, Tonn JC, Stupp R, Preusser M, Cohen-Jonathan-Moyal E, Henriksson R, Le Rhun E, Balana C, Chinot O, Bendszus M, Reijneveld JC, Dhermain F, French P, Marosi C, Watts C, Oberg I, Pilkington G, Baumert BG, Taphoorn MJB, Hegi M, Westphal M, Reifenberger G, Soffietti R, Wick W, European Association for Neuro-Oncology Task Force on G (2017) European Association for Neuro-Oncology (EANO) guideline on the diagnosis and treatment of adult astrocytic and oligodendroglial gliomas. Lancet Oncol 18(6):e315–e329. https://doi.org/10.1016/S1470-2045(17)30194-8
Lev MH, Ozsunar Y, Henson JW, Rasheed AA, Barest GD, Harsh GR, Fitzek MM, Chiocca EA, Rabinov JD, Csavoy AN, Rosen BR, Hochberg FH, Schaefer PW, Gonzalez RG (2004) Glial tumor grading and outcome prediction using dynamic spin-echo MR susceptibility mapping compared with conventional contrast-enhanced MR: confounding effect of elevated rCBV of oligodendrogliomas [corrected]. AJNR Am J Neuroradiol 25(2):214–221
Rees J (2003) Advances in magnetic resonance imaging of brain tumours. Curr Opin Neurol 16(6):643–650. https://doi.org/10.1097/01.wco.0000102626.38669.b9
Hartmann C, Meyer J, Balss J, Capper D, Mueller W, Christians A, Felsberg J, Wolter M, Mawrin C, Wick W, Weller M, Herold-Mende C, Unterberg A, Jeuken JW, Wesseling P, Reifenberger G, von Deimling A (2009) Type and frequency of IDH1 and IDH2 mutations are related to astrocytic and oligodendroglial differentiation and age: a study of 1,010 diffuse gliomas. Acta Neuropathol 118(4):469–474. https://doi.org/10.1007/s00401-009-0561-9
Chen N, Yu T, Gong J, Nie L, Chen X, Zhang M, Xu M, Tan J, Su Z, Zhong J, Zhou Q (2016) IDH1/2 gene hotspot mutations in central nervous system tumours: analysis of 922 Chinese patients. Pathology 48(7):675–683. https://doi.org/10.1016/j.pathol.2016.07.010
Kloosterhof NK, Bralten LB, Dubbink HJ, French PJ, van den Bent MJ (2011) Isocitrate dehydrogenase-1 mutations: a fundamentally new understanding of diffuse glioma? Lancet Oncol 12(1):83–91. https://doi.org/10.1016/S1470-2045(10)70053-X
Cairncross JG, Wang M, Jenkins RB, Shaw EG, Giannini C, Brachman DG, Buckner JC, Fink KL, Souhami L, Laperriere NJ, Huse JT, Mehta MP, Curran WJ Jr (2014) Benefit from procarbazine, lomustine, and vincristine in oligodendroglial tumors is associated with mutation of IDH. J Clin Oncol Off J Am Soc Clin Oncol 32(8):783–790. https://doi.org/10.1200/JCO.2013.49.3726
Pollard PJ, Ratcliffe PJ (2009) Cancer. Puzzling patterns of predisposition. Science 324(5924):192–194. https://doi.org/10.1126/science.1173362
Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Siu IM, Gallia GL, Olivi A, McLendon R, Rasheed BA, Keir S, Nikolskaya T, Nikolsky Y, Busam DA, Tekleab H, Diaz LA Jr, Hartigan J, Smith DR, Strausberg RL, Marie SK, Shinjo SM, Yan H, Riggins GJ, Bigner DD, Karchin R, Papadopoulos N, Parmigiani G, Vogelstein B, Velculescu VE, Kinzler KW (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321(5897):1807–1812. https://doi.org/10.1126/science.1164382
Chaumeil MM, Larson PE, Yoshihara HA, Danforth OM, Vigneron DB, Nelson SJ, Pieper RO, Phillips JJ, Ronen SM (2013) Non-invasive in vivo assessment of IDH1 mutational status in glioma. Nat Commun 4:2429. https://doi.org/10.1038/ncomms3429
Nagashima H, Tanaka K, Sasayama T, Irino Y, Sato N, Takeuchi Y, Kyotani K, Mukasa A, Mizukawa K, Sakata J, Yamamoto Y, Hosoda K, Itoh T, Sasaki R, Kohmura E (2016) Diagnostic value of glutamate with 2-hydroxyglutarate in magnetic resonance spectroscopy for IDH1 mutant glioma. Neuro-Oncology 18(11):1559–1568. https://doi.org/10.1093/neuonc/now090
Warmuth C, Gunther M, Zimmer C (2003) Quantification of blood flow in brain tumors: comparison of arterial spin labeling and dynamic susceptibility-weighted contrast-enhanced MR imaging. Radiology 228(2):523–532. https://doi.org/10.1148/radiol.2282020409
Jarnum H, Steffensen EG, Knutsson L, Frund ET, Simonsen CW, Lundbye-Christensen S, Shankaranarayanan A, Alsop DC, Jensen FT, Larsson EM (2010) Perfusion MRI of brain tumours: a comparative study of pseudo-continuous arterial spin labelling and dynamic susceptibility contrast imaging. Neuroradiology 52(4):307–317. https://doi.org/10.1007/s00234-009-0616-6
Cebeci H, Aydin O, Ozturk-Isik E, Gumus C, Inecikli F, Bekar A, Kocaeli H, Hakyemez B (2014) Assessment of perfusion in glial tumors with arterial spin labeling; comparison with dynamic susceptibility contrast method. Eur J Radiol 83(10):1914–1919. https://doi.org/10.1016/j.ejrad.2014.07.002
Knopp EA, Cha S, Johnson G, Mazumdar A, Golfinos JG, Zagzag D, Miller DC, Kelly PJ, Kricheff II (1999) Glial neoplasms: dynamic contrast-enhanced T2*-weighted MR imaging. Radiology 211(3):791–798. https://doi.org/10.1148/radiology.211.3.r99jn46791
Alsop DC, Detre JA, Golay X, Gunther M, Hendrikse J, Hernandez-Garcia L, Lu H, MacIntosh BJ, Parkes LM, Smits M, van Osch MJ, Wang DJ, Wong EC, Zaharchuk G (2015) Recommended implementation of arterial spin-labeled perfusion MRI for clinical applications: a consensus of the ISMRM perfusion study group and the European consortium for ASL in dementia. Magn Reson Med 73(1):102–116. https://doi.org/10.1002/mrm.25197
Kang Y, Choi SH, Kim YJ, Kim KG, Sohn CH, Kim JH, Yun TJ, Chang KH (2011) Gliomas: histogram analysis of apparent diffusion coefficient maps with standard- or high-b-value diffusion-weighted MR imaging—correlation with tumor grade. Radiology 261(3):882–890. https://doi.org/10.1148/radiol.11110686
Zeng Q, Jiang B, Shi F, Ling C, Dong F, Zhang J (2017) 3D pseudocontinuous arterial spin-labeling MR imaging in the preoperative evaluation of gliomas. AJNR Am J Neuroradiol 38(10):1876–1883. https://doi.org/10.3174/ajnr.A5299
Lin L, Xue Y, Duan Q, Sun B, Lin H, Huang X, Chen X (2016) The role of cerebral blood flow gradient in peritumoral edema for differentiation of glioblastomas from solitary metastatic lesions. Oncotarget 7(42):69051–69059. https://doi.org/10.18632/oncotarget.12053
Shen N, Zhao L, Jiang J, Jiang R, Su C, Zhang S, Tang X, Zhu W (2016) Intravoxel incoherent motion diffusion-weighted imaging analysis of diffusion and microperfusion in grading gliomas and comparison with arterial spin labeling for evaluation of tumor perfusion. J Magn Reson Imaging 44(3):620–632. https://doi.org/10.1002/jmri.25191
Gunther M, Bock M, Schad LR (2001) Arterial spin labeling in combination with a look-locker sampling strategy: inflow turbo-sampling EPI-FAIR (ITS-FAIR). Magn Reson Med 46(5):974–984
Daumas-Duport C, Scheithauer B, O’Fallon J, Kelly P (1988) Grading of astrocytomas. A simple and reproducible method. Cancer 62(10):2152–2165
Plate KH, Breier G, Weich HA, Risau W (1992) Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature 359(6398):845–848. https://doi.org/10.1038/359845a0
Daumas-Duport C, Varlet P (2003) Dysembryoplastic neuroepithelial tumors. Rev Neurol 159(6–7 Pt 1):622–636
Godard S, Getz G, Delorenzi M, Farmer P, Kobayashi H, Desbaillets I, Nozaki M, Diserens AC, Hamou MF, Dietrich PY, Regli L, Janzer RC, Bucher P, Stupp R, de Tribolet N, Domany E, Hegi ME (2003) Classification of human astrocytic gliomas on the basis of gene expression: a correlated group of genes with angiogenic activity emerges as a strong predictor of subtypes. Cancer Res 63(20):6613–6625
Wolf RL, Wang J, Wang S, Melhem ER, O'Rourke DM, Judy KD, Detre JA (2005) Grading of CNS neoplasms using continuous arterial spin labeled perfusion MR imaging at 3 Tesla. J Magn Reson Imaging 22(4):475–482. https://doi.org/10.1002/jmri.20415
Togao O, Hiwatashi A, Yamashita K, Kikuchi K, Mizoguchi M, Yoshimoto K, Suzuki SO, Iwaki T, Obara M, Van Cauteren M, Honda H (2016) Differentiation of high-grade and low-grade diffuse gliomas by intravoxel incoherent motion MR imaging. Neuro-Oncology 18(1):132–141. https://doi.org/10.1093/neuonc/nov147
Chen SD, Hou PF, Lou L, Jin X, Wang TH, Xu JL (2014) The correlation between MR diffusion-weighted imaging and pathological grades on glioma. Eur Rev Med Pharmacol Sci 18(13):1904–1909
Han H, Han C, Wu X, Zhong S, Zhuang X, Tan G, Wu H (2017) Preoperative grading of supratentorial nonenhancing gliomas by high b-value diffusion-weighted 3 T magnetic resonance imaging. J Neuro-Oncol 133(1):147–154. https://doi.org/10.1007/s11060-017-2423-y
Labussiere M, Idbaih A, Wang XW, Marie Y, Boisselier B, Falet C, Paris S, Laffaire J, Carpentier C, Criniere E, Ducray F, El Hallani S, Mokhtari K, Hoang-Xuan K, Delattre JY, Sanson M (2010) All the 1p19q codeleted gliomas are mutated on IDH1 or IDH2. Neurology 74(23):1886–1890. https://doi.org/10.1212/WNL.0b013e3181e1cf3a
Mukasa A, Takayanagi S, Saito K, Shibahara J, Tabei Y, Furuya K, Ide T, Narita Y, Nishikawa R, Ueki K, Saito N (2012) Significance of IDH mutations varies with tumor histology, grade, and genetics in Japanese glioma patients. Cancer Sci 103(3):587–592. https://doi.org/10.1111/j.1349-7006.2011.02175.x
Reuss DE, Sahm F, Schrimpf D, Wiestler B, Capper D, Koelsche C, Schweizer L, Korshunov A, Jones DT, Hovestadt V, Mittelbronn M, Schittenhelm J, Herold-Mende C, Unterberg A, Platten M, Weller M, Wick W, Pfister SM, von Deimling A (2015) ATRX and IDH1-R132H immunohistochemistry with subsequent copy number analysis and IDH sequencing as a basis for an “integrated” diagnostic approach for adult astrocytoma, oligodendroglioma and glioblastoma. Acta Neuropathol 129(1):133–146. https://doi.org/10.1007/s00401-014-1370-3
Wesseling P, van den Bent M, Perry A (2015) Oligodendroglioma: pathology, molecular mechanisms and markers. Acta Neuropathol 129(6):809–827. https://doi.org/10.1007/s00401-015-1424-1
Tan WL, Huang WY, Yin B, Xiong J, Wu JS, Geng DY (2014) Can diffusion tensor imaging noninvasively detect IDH1 gene mutations in astrogliomas? A retrospective study of 112 cases. AJNR Am J Neuroradiol 35(5):920–927. https://doi.org/10.3174/ajnr.A3803
Tan W, Xiong J, Huang W, Wu J, Zhan S, Geng D (2017) Noninvasively detecting Isocitrate dehydrogenase 1 gene status in astrocytoma by dynamic susceptibility contrast MRI. J Magn Reson Imaging 45(2):492–499. https://doi.org/10.1002/jmri.25358
Lee S, Choi SH, Ryoo I, Yoon TJ, Kim TM, Lee SH, Park CK, Kim JH, Sohn CH, Park SH, Kim IH (2015) Evaluation of the microenvironmental heterogeneity in high-grade gliomas with IDH1/2 gene mutation using histogram analysis of diffusion-weighted imaging and dynamic-susceptibility contrast perfusion imaging. J Neuro-Oncol 121(1):141–150. https://doi.org/10.1007/s11060-014-1614-z
Xiong J, Tan WL, Pan JW, Wang Y, Yin B, Zhang J, Geng DY (2016) Detecting isocitrate dehydrogenase gene mutations in oligodendroglial tumors using diffusion tensor imaging metrics and their correlations with proliferation and microvascular density. J Magn Reson Imaging 43(1):45–54. https://doi.org/10.1002/jmri.24958
Xiong J, Tan W, Wen J, Pan J, Wang Y, Zhang J, Geng D (2016) Combination of diffusion tensor imaging and conventional MRI correlates with isocitrate dehydrogenase 1/2 mutations but not 1p/19q genotyping in oligodendroglial tumours. Eur Radiol 26(6):1705–1715. https://doi.org/10.1007/s00330-015-4025-4
Kumar A, Bicer EM, Pfeffer P, Monopoli MP, Dawson KA, Eriksson J, Edwards K, Lynham S, Arno M, Behndig AF, Blomberg A, Somers G, Hassall D, Dailey LA, Forbes B, Mudway I (2017) Differences in the coronal proteome acquired by particles depositing in the lungs of asthmatic versus healthy humans. Nanomed: Nanotechnol Biol Med 13(8):2517–2521. https://doi.org/10.1016/j.nano.2017.06.008
Rakmanee S, Pakakasama S, Hongeng S, Sanguansin S, Thongmee A, Pongstaporn W (2017) Increased risk of Thai childhood acute lymphoblastic leukemia with the MiR196a2 T>C polymorphism. Asian Pac J Cancer Prev 18(4):1117–1120. https://doi.org/10.22034/APJCP.2017.18.4.1117
Zhao S, Lin Y, Xu W, Jiang W, Zha Z, Wang P, Yu W, Li Z, Gong L, Peng Y, Ding J, Lei Q, Guan KL, Xiong Y (2009) Glioma-derived mutations in IDH1 dominantly inhibit IDH1 catalytic activity and induce HIF-1alpha. Science 324(5924):261–265. https://doi.org/10.1126/science.1170944
Ichimura K, Pearson DM, Kocialkowski S, Backlund LM, Chan R, Jones DT, Collins VP (2009) IDH1 mutations are present in the majority of common adult gliomas but rare in primary glioblastomas. Neuro-Oncology 11(4):341–347. https://doi.org/10.1215/15228517-2009-025
Jensen MV, Haldeman JM, Zhang H, Lu D, Huising MO, Vale WW, Hohmeier HE, Rosenberg P, Newgard CB (2013) Control of voltage-gated potassium channel Kv2.2 expression by pyruvate-isocitrate cycling regulates glucose-stimulated insulin secretion. J Biol Chem 288(32):23128–23140. https://doi.org/10.1074/jbc.M113.491654
Baumann F, Leukel P, Doerfelt A, Beier CP, Dettmer K, Oefner PJ, Kastenberger M, Kreutz M, Nickl-Jockschat T, Bogdahn U, Bosserhoff AK, Hau P (2009) Lactate promotes glioma migration by TGF-beta2-dependent regulation of matrix metalloproteinase-2. Neuro-Oncology 11(4):368–380. https://doi.org/10.1215/15228517-2008-106
Sanson M, Marie Y, Paris S, Idbaih A, Laffaire J, Ducray F, El Hallani S, Boisselier B, Mokhtari K, Hoang-Xuan K, Delattre JY (2009) Isocitrate dehydrogenase 1 codon 132 mutation is an important prognostic biomarker in gliomas. J Clin Oncol Off J Am Soc Clin Oncol 27(25):4150–4154. https://doi.org/10.1200/JCO.2009.21.9832
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study was funded by the National Natural Science Foundation of China (No. 81372457).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed consent
For this type of retrospective study formal consent is not required.
Rights and permissions
About this article
Cite this article
Liu, T., Cheng, G., Kang, X. et al. Noninvasively evaluating the grading and IDH1 mutation status of diffuse gliomas by three-dimensional pseudo-continuous arterial spin labeling and diffusion-weighted imaging. Neuroradiology 60, 693–702 (2018). https://doi.org/10.1007/s00234-018-2021-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-018-2021-5