Abstract
Introduction
Rapidly increasing aging of the world’s population is causing a heightened prevalence of Alzheimer’s disease (AD) and mild cognitive impairment (MCI). The global burden, caused by this, is tremendous. In order to slow down the progression of the disease and preserve quality of life as much as possible, early identification of subjects at risk is indispensable within this framework.
Methods
In the present study, we combined independent component analysis and statistical parametric analysis to identify and compare the default-mode network (DMN) in healthy elderly and patients with MCI, with a special interest for hippocampal and lateral temporal involvement.
Results
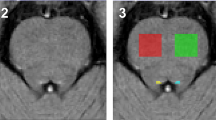
Functional results indicated reduced cortical activation in the DMN for MCI patients, compared with age- and education-matched healthy elderly controls, mainly in the retrosplenial region/posterior cingulate cortex, left hippocampus, and bilateral inferior and middle frontal areas. Increased activation for patients was observed in the medial prefrontal and bilateral middle temporal/angular cortex. Lateral temporal involvement in the DMN was in both the elderly control samples, and the patient group detected and suggested a slightly increased activation, more right than left, in middle temporal areas in the MCI patients, compared with healthy elderly.
Conclusion
Results are discussed with reference to the existing literature on early pathological changes in MCI and AD and subsequent compensation mechanisms in resting state and memory circuits.


Similar content being viewed by others
References
Gauthier S, Reisberg B, Zaudig M, Petersen RC, Ritchie K, Broich K, Belleville S, Brodaty H, Bennett D, Chertkow H, Cummings JL, de Leon M, Feldman H, Ganguli M, Hampel H, Scheltens P, Tierney MC, Whitehouse P, Winblad B, IPAEC (2006) Mild cognitive impairment. Lancet 367:1262–1270
Gustavson A, Scensson M et al (2011) Cost of disorders of the brain in Europe 2010. Eur Neuropsychopharmacol 21:718–779
Brookmeyer R, Johnson E, Ziegler-Graham K, Arrighi HM (2007) Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement 3:186–191
Petersen RC, Smith GE, Ivnik RJ, Tangalos EG, Schaid DJ, Thibodeau SN, Kokmen E, Waring SC, Kurland LT (1995) Apolipoprotein-E status as a predictor of the development of Alzheimer’s disease in memory-impaired individuals. JAMA 273:1274–1278
Petersen RC, Doody R, Kurz A, Mohs RC, Morris JC, Rabins PV, Ritchie K, Rossor M, Thal L, Winblad B (2001) Current concepts in mild cognitive impairment. Arch Neurol 58:1985–1992
Bondi MW, Wierenga CE (2007) Use of functional magnetic resonance imaging in the early identification of Alzheimer’s disease. Neuropsychol Rev 17:127–143
Calhoun VD, Adali T, Pearlson GD, Pekar JJ (2001) A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp 14:140–151
Braak H, Braak E (1991) Neuropathological staging of Alzheimer-related changes. Acta Neuropathol 82:239–259
Buckner RL, Andrews-Hanna JR, Schacter DL (2008) The brain’s default network—anatomy, function, and relevance to disease. J Cogn Neurosci 1124:1–38
Damoiseaux JS, Greicius MD (2009) Greater than the sum of its parts: a review of studies combining structural connectivity and resting-state functional connectivity. Brain Struct Funct 213:525–533
Raichle ME, Macleod AM, Snyder AZ, Powers WJ, Gusnard DA, Shulman GL (2001) A default mode of brain function. Proc Natl Acad Sci USA 98:676–682
Damoiseaux JS, Rombouts SARB, Barkhof F, Scheltens P, Stam CJ, Smith SM, Beckmann CF (2006) Consistent resting-state networks across healthy subjects. Proc Natl Acad Sci USA 103:13848–13853
Greicius MD, Srivastava G, Reiss AL, Menon V (2004) Default-mode network activity distinguishes Alzheimer’s disease from healthy aging: Evidence from functional MRI. Proc Natl Acad Sci USA 101:4637–4642
Rombouts SARB, Barkhof F, Goekoop R, Stam CJ, Scheltens P (2005) Altered resting state networks in mild cognitive impairment and mild Alzheimer’s disease: an fMRI study. Hum Brain Mapp 26:231–239
Qi ZG, Wu X, Wang ZQ, Zhang N, Dong HQ, Yao L, Li KC (2010) Impairment and compensation coexist in amnestic MCI default mode network. Neuroimage 50:48–55
Sorg C, Riedl V, Muhlau M, Calhoun VD, Eichele T, Laer L, Drzezga A, Forstl H, Kurz A, Zimmer C, Wohlschlager AM (2007) Selective changes of resting-state networks in individuals at risk for Alzheimer’s disease. Proc Natl Acad Sci USA 104:18760–18765
Ojemann GA, Schoenfield-McNeill J, Corina D (2009) The roles of human lateral temporal cortical neuronal activity in recent verbal memory encoding. Cereb Cortex 19:197–205
Sperling RA, Bates JF, Chua EF, Cocchiarella AJ, Rentz DM, Rosen BR, Schacter DL, Albert MS (2003) FMRI studies of associative encoding in young and elderly controls and mild Alzheimer’s disease. J Neurol Neurosurg Psychiatry 74:44–50
Machulda MM (2003) Comparison of memory fMRI response among normal, MCI, and Alzheimer’s patients. Neurology 61:1164–1164 (vol 61, pg 500, 2003)
Small SA, Perera GM, DeLaPaz R, Mayeux R, Stern Y (1999) Differential regional dysfunction of the hippocampal formation among elderly with memory decline and Alzheimer’s disease. Ann Neurol 45:466–472
Dickerson BC, Salat DH, Greve DN, Chua EF, Rand-Giovannetti E, Rentz DM, Bertram L, Mullin K, Tanzi RE, Blacker D, Albert MS, Sperling RA (2005) Increased hippocampal activation in mild cognitive impairment compared to normal aging and AD. Neurology 65:404–411
Johnson SC, Schmitz TW, Moritz CH, Meyerand ME, Rowley HA, Alexander AL, Hansen KW, Gleason CE, Carlsson CM, Ries ML, Asthana S, Chen K, Reiman EM, Alexander GE (2006) Activation of brain regions vulnerable to Alzheimer’s disease: the effect of mild cognitive impairment. Neurobiol Aging 27:1604–1612
Greicius MD, Supekar K, Menon V, Dougherty RF (2009) Resting-state functional connectivity reflects structural connectivity in the default mode network. Cereb Cortex 19:72–78
Hedden T, Van Dijk KRA, Becker JA, Mehta A, Sperling RA, Johnson KA, Buckner RL (2009) Disruption of functional connectivity in clinically normal older adults harboring amyloid burden. J Neurosci 29:12686–12694
Wang L, Zang YF, He Y, Liang M, Zhang XQ, Tian LX, Wu T, Jiang TZ, Li KC (2006) Changes in hippocampal connectivity in the early stages of Alzheimer’s disease: evidence from resting state fMRI. Neuroimage 31:496–504
Bai F, Zhang ZJ, Yu H, Shi YM, Yuan YG, Zhu WL, Zhang XR, Qian Y (2008) Default-mode network activity distinguishes amnestic type mild cognitive impairment from healthy aging: a combined structural and resting-state functional MRI study. Neurosci Lett 439:111–115
Wang K, Liang M, Wang L, Tian LX, Zhang XQ, Li KC, Jiang TZ (2007) Altered functional connectivity in early Alzheimer’s disease: a resting-state fMRI study. Hum Brain Mapp 28:967–978
Grady CL, McIntosh AR, Beig S, Keightley ML, Burian H, Black SE (2003) Evidence from functional neuroimaging of a compensatory prefrontal network in Alzheimer’s disease. J Neurosci 23:986–993
Oldfield RC (1971) Assessment and analysis of handedness—Edinburgh Inventory. Neuropsychologia 9:97
Petersen RC, Stevens JC, Ganguli M, Tangalos EG, Cummings JL, DeKosky ST (2001) Practice parameter: early detection of dementia: Mild cognitive impairment (an evidence-based review)—report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 56:1133–1142
Morris JC (1993) The clinical dementia rating (CDR)—current version and scoring rules. Neurology 43:2412–2414
Hughes CP, Berg L, Danziger WL, Coben LA, Martin RL (1982) A new clinical-scale for the staging of dementia. Br J Psychiatry 140:566–572
Miatton M, Wolters M, Lannoo E, Vingerhoets G (2004) Updated and extended Flemish normative data of commonly used neuropsychological tests. Psychol Belg 44:189–216
Ashburner J, Friston KJ (2000) Voxel-based morphometry—the methods. Neuroimage 11:805–821
Stevens MC, Kiehl KA, Pearlson G, Calhoun VD (2007) Functional neural circuits for mental timekeeping. Hum Brain Mapp 28:394–408
Petrella JR, Sheldon FC, Prince SE, Calhoun VD, Doraiswamy PM (2011) Default mode network connectivity in stable vs progressive mild cognitive impairment. Neurology 76:511–517
Jin M, Pelak VS, Cordes D (2012) Aberrant default mode network in subjects with amnestic mild cognitive impairment using resting-state functional MRI. Magn Reson Imaging 30:48–61
Grady CL, Furey ML, Pietrini P, Horwitz B, Rapoport SI (2001) Altered functional connectivity and impaired short-term memory in Alzheimer’s disease. Brain 124:739–756
Schwindt GC, Black SE (2009) Functional imaging studies of episodic memory in Alzheimer’s disease: a quantitative meta-analysis. Neuroimage 45:181–190
Collie A, Maruff P (2000) The neuropsychology of preclinical Alzheimer’s disease and mild cognitive impairment. Neurosci Biobehav Rev 24:365–374
Bai F, Liao W, Watson DR, Shi YM, Wang Y, Yue CX, Teng YH, Wu D, Yuan YG, Jia JP, Zhang ZJ (2011) Abnormal whole-brain functional connection in amnestic mild cognitive impairment patients. Behav Brain Res 216:666–672
Wang Z, Yan C, Zhao C, Qi Z, Zhou W, Lu J, He Y, Li K (2011) Spatial patterns of intrinsic brain activity in mild cognitive impairment and Alzheimer’s disease: a resting-state functional MRI study. Hum Brain Mapp 32:1720–1740
Smith CD, Umberger GH, Manning EL, Slevin JT, Wekstein DR, Schmitt FA, Markesbery WR, Zhang Z, Gerhardt GA, Kryscio RJ, Gash DM (1999) Critical decline in fine motor hand movements in human aging. Neurology 53:1458–1461
Agosta F, Rocca MA, Pagani E, Absinta M, Magnani G, Marcone A, Falautano M, Comi G, Gorno-Tempini ML, Filippi M (2010) Sesorimotor network rewiring in mild cognitive impairment and Alzheimer’s disease. Hum Brain Mapp 31:515–525
Karas GB, Burton EJ, Rombouts SARB, van Schijndel RA, O’Brien JT, Scheltens P, McKeith IG, Williams D, Ballard C, Barkhof FA (2003) Comprehensive study of gray matter loss in patients with Alzheimer’s disease using optimized voxel-based morphometry. Neuroimage 18:895–907
Karas GB, Scheltens P, Rombouts SARB, Visser PJ, van Schijndel RA, Fox NC, Barkhof F (2004) Global and local gray matter loss in mild cognitive impairment and Alzheimer’s disease. Neuroimage 23:708–716
Chetelat G, Landeau B, Eustache F, Mezenge F, Viader F, de la Sayette V, Desgranges B, Baron JC (2005) Using voxel-based morphometry to map the structural changes associated with rapid conversion in MCI: a longitudinal MRI study. Neuroimage 27:934–946
Whitwell JL, Shiung MM, Przybelski SA, Weigand SD, Knopman DS, Boeve BF, Petersen RC, Jack CR (2008) MRI patterns of atrophy associated with progression to AD in amnestic mild cognitive impairment. Neurology 70:512–520
Sluimer JD, van der Flier WM, Karas GB, van Schijndel R, Barnes J, Boyes RG, Cover KS, Olabarriaga SD, Fox NC, Scheltens P, Vrenken H, Barkhof F (2009) Accelerating regional atrophy rates in the progression from normal aging to Alzheimer’s disease. Eur Radiol 19:12826–12833
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th edn. American Psychiatric Association, Washington DC, pp 157–158, text rev
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Vogelaere, F., Santens, P., Achten, E. et al. Altered default-mode network activation in mild cognitive impairment compared with healthy aging. Neuroradiology 54, 1195–1206 (2012). https://doi.org/10.1007/s00234-012-1036-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-012-1036-6