Abstract
Purpose
The objective of this study was to appraise the safety profiles of HES preparations and to find out which HES preparation was the most acceptable in cardiovascular surgery through a comparison with control solutions.
Methods
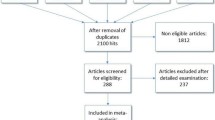
Pertinent randomized controlled trials were selected through a search of Pubmed, Embase, and Cochrane Controlled Trials Register. Quantitative and qualitative analysis was carried out to evaluate blood loss, blood transfusion, renal function, complications, reoperation, and mortality.
Results
A total of 3,234 patients from 52 randomized controlled trials were included. HES preparations versus control solutions in blood loss: HES 130 kD vs. albumin (SMD −0.61, 95% CI −0.82, −0.40), HES 200 kD vs. albumin (SMD −0.01, 95% CI −0.29, 0.28), HES 450 kD vs. albumin (SMD 0.47, 95% CI 0.26, 0.68). When comparing control solutions with HES preparations, 50% (HES 450 kD), 40.9% (HES 200 kD), and 18.2% (HES 130 kD) of the comparisons showed more blood/blood products infusion with HES than with control solutions. A numerically lower mortality rate seemed to be related to HES preparations (2.68 vs 4.23%). No difference was found in terms of complications, renal failure, or reoperation.
Conclusions
Perioperative administration of HES preparations is comparatively safe. The data appraising safety profiles of HES preparations are insufficient to make direct comparisons among themselves. As the third generation of HES preparations, HES 130 kD showed a trend toward lower blood loss and transfusion rates and is a suitable choice for cardiovascular surgery.






Similar content being viewed by others
References
Laxenaire MC, Charpentier C, Feldman L (1994) Anaphylactoid reactions to colloid plasma substitutes: incidence, risk factors, mechanisms. A French multicenter prospective study. Ann Fr Anesth Rèanim 13(3):301–310
Ford SA, Kam PC, Baldo BA, Fisher MM (2001) Anaphylactic or anaphylactoid reactions in patients undergoing cardiac surgery. J Cardiothorac Vasc Anesth 15(6):684–688
Nearman HS, Herman ML (1991) Toxic effects of colloids in the intensive care unit. Crit Care Clin 7(3):713–723
Treib J, Baron JF, Grauer MT, Strauss RG (1999) An international view of hydroxyethyl starches. Intensive Care Med 25(3):258–268
Schramko AA, Suojaranta-Ylinen RT, Kuitunen AH, Kukkonen SI, Niemi TT (2009) Rapidly degradable hydroxyethyl starch solutions impair blood coagulation after cardiac surgery: a prospective randomized trial. Anesth Analg 108(1):30–36
Schramko A, Suojaranta-Ylinen R, Kuitunen A, Raivio P, Kukkonen S, Niemi T (2001) Hydroxyethylstarch and gelatin solutions impair blood coagulation after cardiac surgery: a prospective randomized trial. Br J Anaesth 104(6):691–697
Shore-Lesserson L, Manspeizer HE, DePerio M, Francis S, Vela-Cantos F, Ergin MA (1999) Thromboelastography-guided transfusion algorithm reduces transfusions in complex cardiac surgery. Anesth Analg 88(2):312–319
Kozek-Langenecker SA, Jungheinrich C, Sauermann W, Van der Linden P (2008) The effects of hydroxyethyl starch 130/0.4 (6%) on blood loss and use of blood products in major surgery: a pooled analysis of randomized clinical trials. Anesth Analg 107(2):382–390
Wilkes MM, Navickis RJ, Sibbald WJ (2001) Albumin versus hydroxyethyl starch in cardiopulmonary bypass surgery: a meta-analysis of postoperative bleeding. Ann Thorac Surg 72(2):527–533
Traylor RJ, Pearl RG (1996) Crystalloid versus colloid versus colloid: all colloids are not created equal. Anesth Analg 83(2):209–212
Jungheinrich C, Neff TA (2005) Pharmacokinetics of hydroxyethyl starch. Clin Pharmacokinet 44(7):681–699
Boldt J (2006) Do plasma substitutes have additional properties beyond correcting volume deficits? Shock 25(2):103–116
Strauss RGSC, Henriksen RA, Villhauer PJ (1988) Pentastarch may cause fewer effects on coagulation than hetastarch. Transfusion 28(3):257–260
Boldt J, Suttner S, Brosch C, Lehmann A, Rohm K, Mengistu A (2009) Cardiopulmonary bypass priming using a high dose of a balanced hydroxyethyl starch versus an albumin-based priming strategy. Anesth Analg 109(6):1752–1762
Pagliaro L, D'Amico G, Sorensen TI, Lebrec D, Burroughs AK, Morabito A, Tine F, Politi F, Traina M (1992) Prevention of first bleeding in cirrhosis. A meta-analysis of randomized trials of nonsurgical treatment. Ann Intern Med 117(1):59–70
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17(1):1–12
Cheng JW, Wei RL, Cai JP, Xi GL, Zhu H, Li Y, Ma XY (2007) Efficacy of different intraocular lens materials and optic edge designs in preventing posterior capsular opacification: a meta-analysis. Am J Ophthalmol 143(3):428–436
Kuitunen A, Suojaranta-Ylinen R, Kukkonen S, Niemi T (2007) A comparison of the haemodynamic effects of 4% succinylated gelatin, 6% hydroxyethyl starch (200/0.5) and 4% human albumin after cardiac surgery. Scand J Surg 96(1):72–78
Moiniche S, Mikkelsen S, Wetterslev J, Dahl JB (1998) A qualitative systematic review of incisional local anaesthesia for postoperative pain relief after abdominal operations. Br J Anaesth 81(3):377–383
Cook DJ, Mulrow CD, Haynes RB (1997) Systematic reviews: synthesis of best evidence for clinical decisions. Ann Intern Med 126(5):376–380
Niemi TT, Suojaranta-Ylinen RT, Kukkonen SI, Kuitunen AH (2006) Gelatin and hydroxyethyl starch, but not albumin, impair hemostasis after cardiac surgery. Anesth Analg 102(4):998–1006
Niemi T, Schramko A, Kuitunen A, Kukkonen S, Suojaranta-Ylinen R (2008) Haemodynamics and acid-base equilibrium after cardiac surgery: comparison of rapidly degradable hydroxyethyl starch solutions and albumin. Scand J Surg 97(3):259–265
Tigchelaar I, Gallandat Huet RC, Korsten J, Boonstra PW, van Oeveren W (1997) Hemostatic effects of three colloid plasma substitutes for priming solution in cardiopulmonary bypass. Eur J Cardiothorac Surg 11(4):626–632
Lumb PD (1987) A comparison between 25% albumin and 6% hydroxyethyl starch solutions on lung water accumulation during and immediately after cardiopulmonary bypass. Ann Surg 206(2):210–213
Dytkowska B, Karwacki Z, Suchorzewska J, Wujtewicz M (1998) Comparative assessment of 200/0.5 HAES 6% and Gelafundin in the treatment of hypovolaemia in post-coronary bypass patients. Med Sci Monit 4(6):CR1000–CR1003
Ragaller M, Muller M, Bleyl JU, Strecker A, Segiet TW, Ellinger K, Albrecht DM (2000) Hemodynamic effects of hypertonic hydroxyethyl starch 6% solution and isotonic hydroxyethyl starch 6% solution after declamping during abdominal aortic aneurysm repair. Shock 13(5):367–373
Boldt J, Zickmann B, Rapin J, Hammermann H, Dapper F, Hempelmann G (1994) Influence of volume replacement with different HES-solutions on microcirculatory blood flow in cardiac surgery. Acta Anaesthesiol Scand 38(5):432–438
Schroth M, Plank C, Meissner U, Eberle KP, Weyand M, Cesnjevar R, Dotsch J, Rascher W (2006) Hypertonic-hyperoncotic solutions improve cardiac function in children after open-heart surgery. Pediatrics 118(1):e76–e84
Khosropour R, Lackner F, Steinbereithner K, Watzek C, Piza F, Wagner O, Amesberger C (1980) Comparison of effects of hydroxyethylstarch (HES 200/0.5) administered pre- and postoperatively in vascular surgery with dextran 40 (60) (author's transl). Anaesthesist 29(11):616–622
Diehl JT, Lester JL 3rd, Cosgrove DM (1982) Clinical comparison of hetastarch and albumin in postoperative cardiac patients. Ann Thorac Surg 34(6):674–679
Moggio RA, Rha CC, Somberg ED, Praeger PI, Pooley RW, Reed GE (1983) Hemodynamic comparison of albumin and hydroxyethyl starch in postoperative cardiac surgery patients. Crit Care Med 11(12):943–945
Yen TS, Chan KC (2008) Intraoperative coagulation was more interfered by HES 200/0.5 than normal saline in off-pump coronary artery bypass surgery. J Cardiovasc Surg (Torino) 49(5):679–684
Mythen MG, Webb AR (1995) Perioperative plasma volume expansion reduces the incidence of gut mucosal hypoperfusion during cardiac surgery. Arch Surg 130(4):423–429
Verheij J, van Lingen A, Beishuizen A, Christiaans HM, de Jong JR, Girbes AR, Wisselink W, Rauwerda JA, Huybregts MA, Groeneveld AB (2006) Cardiac response is greater for colloid than saline fluid loading after cardiac or vascular surgery. Intensive Care Med 32(7):1030–1038
Boldt J, Mayer J, Brosch C, Lehmann A, Mengistu A (2010) Volume replacement with a balanced hydroxyethyl starch (HES) preparation in cardiac surgery patients. J Cardiothorac Vasc Anesth 24(3):399–407
Boldt J, Brosch C, Rohm K, Papsdorf M, Mengistu A (2008) Comparison of the effects of gelatin and a modern hydroxyethyl starch solution on renal function and inflammatory response in elderly cardiac surgery patients. Br J Anaesth 100(4):457–464
Mahmood A, Gosling P, Vohra RK (2007) Randomized clinical trial comparing the effects on renal function of hydroxyethyl starch or gelatine during aortic aneurysm surgery. Br J Surg 94(4):427–433
Godet G, Lehot JJ, Janvier G, Steib A, De Castro V, Coriat P (2008) Safety of HES 130/0.4 (Voluven(R)) in patients with preoperative renal dysfunction undergoing abdominal aortic surgery: a prospective, randomized, controlled, parallel-group multicentre trial. Eur J Anaesthesiol 25(12):986–994
London MJ, Ho JS, Triedman JK, Verrier ED, Levin J, Merrick SH, Hanley FL, Browner WS, Mangano DT (1989) A randomized clinical trial of 10% pentastarch (low molecular weight hydroxyethyl starch) versus 5% albumin for plasma volume expansion after cardiac operations. J Thorac Cardiovasc Surg 97(5):785–797
Marik PE, Iglesias J, Maini B (1997) Gastric intramucosal pH changes after volume replacement with hydroxyethyl starch or crystalloid in patients undergoing elective abdominal aortic aneurysm repair. J Crit Care 12(2):51–55
Gallandat Huet RC, Siemons AW, Baus D, van Rooyen-Butijn WT, Haagenaars JA, van Oeveren W, Bepperling F (2000) A novel hydroxyethyl starch (Voluven) for effective perioperative plasma volume substitution in cardiac surgery. Can J Anaesth 47(12):1207–1215
Perel P, Roberts I (2007) Colloids versus crystalloids for fluid resuscitation in critically ill patients. Cochrane Database Syst Rev (4):CD000567
Bunn F, Trivedi D, Ashraf S (2008) Colloid solutions for fluid resuscitation. Cochrane Database Syst Rev (1):CD001319
Hartog CS, Bauer M, Reinhart K (2011) The efficacy and safety of colloid resuscitation in the critically ill. Anesth Analg 112(1):156–164
Dart AB, Mutter TC, Ruth CA, Taback SP (2010) Hydroxyethyl starch (HES) versus other fluid therapies: effects on kidney function. Cochrane Database Syst Rev (1):CD007594
Himpe D (2003) Colloids versus crystalloids as priming solutions for cardiopulmonary bypass: a meta-analysis of prospective, randomised clinical trials. Acta Anaesthesiol Belg 54(3):207–215
Ali MA, Saleh M (2004) Selection of optimal quantity of hydroxyethyl starch in the cardiopulmonary bypass prime. Perfusion 19(1):41–45
Boks RH, Wijers MJ, Hofland J, Takkenberg JJ, Bogers AJ (2007) Low molecular starch versus gelatin plasma expander during CPB: does it make a difference? Perfusion 22(5):333–337
Boldt J, von Bormann B, Kling D, Borner U, Mulch J, Hempelmann G (1986) Volume replacement with a new hydroxyethyl starch preparation (3 percent HES 200/0.5) in heart surgery. Infusionsther Klin Ernähr 13(3):145–151
Boldt J, Kling D, Zickmann B, Jacobi M, von Bormann B, Dapper F, Hempelmann G (1990) Acute plasmapheresis during cardiac surgery: volume replacement by crystalloids versus colloids. J Cardiothorac Anesth 4(5):564–570
Boldt J, Zickmann B, Ballesteros BM, Stertmann F, Hempelmann G (1992) Influence of five different priming solutions on platelet function in patients undergoing cardiac surgery. Anesth Analg 74(2):219–225
Boldt J, Knothe C, Zickmann B, Andres P, Dapper F, Hempelmann G (1993) Influence of different intravascular volume therapies on platelet function in patients undergoing cardiopulmonary bypass. Anesth Analg 76(6):1185–1190
Boldt J, Knothe C, Schindler E, Hammermann H, Dapper F, Hempelmann G (1993) Volume replacement with hydroxyethyl starch solution in children. Br J Anaesth 70(6):661–665
Boldt J, Lehmann A, Rompert R, Haisch G, Isgro F (2000) Volume therapy with a new hydroxyethyl starch solution in cardiac surgical patients before cardiopulmonary bypass. J Cardiothorac Vasc Anesth 14(3):264–268
Boldt J, Brenner T, Lehmann A, Lang J, Kumle B, Werling C (2003) Influence of two different volume replacement regimens on renal function in elderly patients undergoing cardiac surgery: comparison of a new starch preparation with gelatin. Intensive Care Med 29(5):763–769
Boldt J, Brosch C, Ducke M, Papsdorf M, Lehmann A (2007) Influence of volume therapy with a modern hydroxyethylstarch preparation on kidney function in cardiac surgery patients with compromised renal function: a comparison with human albumin. Crit Care Med 35(12):2740–2746
Boldt J, Brosch C, Rohm K, Lehmann A, Mengistu A, Suttner S (2008) Is albumin administration in hypoalbuminemic elderly cardiac surgery patients of benefit with regard to inflammation, endothelial activation, and long-term kidney function? Anesth Analg 107(5):1496–1503
Bruegger D, Bauer A, Rehm M, Niklas M, Jacob M, Irlbeck M, Becker BF, Christ F (2005) Effect of hypertonic saline dextran on acid-base balance in patients undergoing surgery of abdominal aortic aneurysm. Crit Care Med 33(3):556–563
Brutocao D, Bratton SL, Thomas JR, Schrader PF, Coles PG, Lynn AM (1996) Comparison of hetastarch with albumin for postoperative volume expansion in children after cardiopulmonary bypass. J Cardiothorac Vasc Anesth 10(3):348–351
Choi YS, Shim JK, Hong SW, Kim JC, Kwak YL (2010) Comparing the effects of 5% albumin and 6% hydroxyethyl starch 130/0.4 on coagulation and inflammatory response when used as priming solutions for cardiopulmonary bypass. Minerva Anestesiol 76(8):584–591
Chong Sung K, Kum Suk P, Mi Ja Y, Kyoung Ok K (2006) Effects of intravascular volume therapy using hydroxyethyl starch (130/0.4) on post-operative bleeding and transfusion requirements in children undergoing cardiac surgery: a randomized clinical trial. Acta Anaesthesiol Scand 50(1):108–111
Eising GP, Niemeyer M, Gunther T, Tassani P, Pfauder M, Schad H, Lange R (2001) Does a hyperoncotic cardiopulmonary bypass prime affect extravascular lung water and cardiopulmonary function in patients undergoing coronary artery bypass surgery? Eur J Cardiothorac Surg 20(2):282–289
Gallagher JD, Moore RA, Kerns D, Jose AB, Botros SB, Flicker S, Naidech H, Clark DL (1985) Effects of colloid or crystalloid administration on pulmonary extravascular water in the postoperative period after coronary artery bypass grafting. Anesth Analg 64(8):753–758
Gold MS, Russo J, Tissot M, Weinhouse G, Riles T (1990) Comparison of hetastarch to albumin for perioperative bleeding in patients undergoing abdominal aortic aneurysm surgery. A prospective, randomized study. Ann Surg 211(4):482–485
Haisch G, Boldt J, Krebs C, Suttner S, Lehmann A, Isgro F (2001) Influence of a new hydroxyethylstarch preparation (HES 130/0.4) on coagulation in cardiac surgical patients. J Cardiothorac Vasc Anesth 15(3):316–321
Hanart C, Khalife M, De Ville A, Otte F, De Hert S, Van der Linden P (2009) Perioperative volume replacement in children undergoing cardiac surgery: albumin versus hydroxyethyl starch 130/0.4. Crit Care Med 37(2):696–701
Hecht-Dolnik M, Barkan H, Taharka A, Loftus J (2009) Hetastarch increases the risk of bleeding complications in patients after off-pump coronary bypass surgery: a randomized clinical trial. J Thorac Cardiovasc Surg 138(3):703–711
Honkonen EL, Jarvela K, Huhtala H, Holm P, Lindgren L (2009) Hyper osmolality does not modulate natriuretic peptide concentration in patients after coronary artery surgery. Acta Anaesthesiol Scand 53(5):565–572
Iriz E, Kolbakir F, Akar H, Adam B, Keceligil HT (2005) Comparison of hydroxyethyl starch and ringer lactate as a prime solution regarding S-100beta protein levels and informative cognitive tests in cerebral injury. Ann Thorac Surg 79(2):666–671
Kasper SM, Meinert P, Kampe S, Gorg C, Geisen C, Mehlhorn U, Diefenbach C (2003) Large-dose hydroxyethyl starch 130/0.4 does not increase blood loss and transfusion requirements in coronary artery bypass surgery compared with hydroxyethyl starch 200/0.5 at recommended doses. Anesthesiology 99(1):42–47
Kirklin JK, Lell WA, Kouchoukos NT (1984) Hydroxyethyl starch versus albumin for colloid infusion following cardiopulmonary bypass in patients undergoing myocardial revascularization. Ann Thorac Surg 37(1):40–46
Kuitunen A, Hynynen M, Salmenpera M, Heinonen J, Vahtera E, Verkkala K, Myllyla G (1993) Hydroxyethyl starch as a prime for cardiopulmonary bypass: effects of two different solutions on haemostasis. Acta Anaesthesiol Scand 37(7):652–658
Kuitunen AH, Hynynen MJ, Vahtera E, Salmenpera MT (2004) Hydroxyethyl starch as a priming solution for cardiopulmonary bypass impairs hemostasis after cardiac surgery. Anesth Analg 98(2):291–297
Kvalheim VL, Farstad M, Steien E, Mongstad A, Borge BA, Kvitting PM, Husby P (2009) Infusion of hypertonic saline/starch during cardiopulmonary bypass reduces fluid overload and may impact cardiac function. Acta Anaesthesiol Scand 54(4):485–493
Ley SJ, Miller K, Skov P, Preisig P (1990) Crystalloid versus colloid fluid therapy after cardiac surgery. Heart Lung 19(1):31–40
London MJ, Franks M, Verrier ED, Merrick SH, Levin J, Mangano DT (1992) The safety and efficacy of ten percent pentastarch as a cardiopulmonary bypass priming solution. A randomized clinical trial. J Thorac Cardiovasc Surg 104(2):284–296
Magder S, Potter BJ, Varennes BD, Doucette S, Fergusson D (2010) Fluids after cardiac surgery: a pilot study of the use of colloids versus crystalloids. Crit Care Med 38(11):2117–2124
Mastroianni L, Low HB, Rollman J, Wagle M, Bleske B, Chow MS (1994) A comparison of 10% pentastarch and 5% albumin in patients undergoing open-heart surgery. J Clin Pharmacol 34(1):34–40
Munsch CM, MacIntyre E, Machin SJ, Mackie IJ, Treasure T (1988) Hydroxyethyl starch: an alternative to plasma for postoperative volume expansion after cardiac surgery. Br J Surg 75(7):675–678
Ooi JS, Ramzisham AR, Zamrin MD (2009) Is 6% hydroxyethyl starch 130/0.4 safe in coronary artery bypass graft surgery? Asian Cardiovasc Thorac Ann 17(4):368–372
Sade RM, Stroud MR, Crawford FA Jr, Kratz JM, Dearing JP, Bartles DM (1985) A prospective randomized study of hydroxyethyl starch, albumin, and lactated Ringer's solution as priming fluid for cardiopulmonary bypass. J Thorac Cardiovasc Surg 89(5):713–722
Sirvinskas E, Sneider E, Svagzdiene M, Vaskelyte J, Raliene L, Marchertiene I, Adukauskiene D (2007) Hypertonic hydroxyethyl starch solution for hypovolaemia correction following heart surgery. Perfusion 22(2):121–127
Tigchelaar I, Gallandat Huet RC, Boonstra PW, van Oeveren W (1998) Comparison of three plasma expanders used as priming fluids in cardiopulmonary bypass patients. Perfusion 13(5):297–303
Tiryakioglu O, Yildiz G, Vural H, Goncu T, Ozyazicioglu A, Yavuz S (2008) Hydroxyethyl starch versus Ringer solution in cardiopulmonary bypass prime solutions (a randomized controlled trial). J Cardiothorac Surg 3:45
Van der Linden PJ, De Hert SG, Daper A, Trenchant A, Schmartz D, Defrance P, Kimbimbi P (2004) 3.5% urea-linked gelatin is as effective as 6% HES 200/0.5 for volume management in cardiac surgery patients. Can J Anaesth 51(3):236–241
Van der Linden PJ, De Hert SG, Deraedt D, Cromheecke S, De Decker K, De Paep R, Rodrigus I, Daper A, Trenchant A (2005) Hydroxyethyl starch 130/0.4 versus modified fluid gelatin for volume expansion in cardiac surgery patients: the effects on perioperative bleeding and transfusion needs. Anesth Analg 101(3):629–634
Vanhoonacker J, Ongenae M, Vanoverschelde H, Donadoni R (2009) Hydroxyethyl starch 130/0.4 versus modified fluid gelatin for cardiopulmonary bypass priming: the effects on postoperative bleeding and volume expansion needs after elective CABG. Acta Anaesthesiol Belg 60(2):91–97
Yap WW, Young D, Pathi V (2007) Effects of gelatine and medium molecular weight starch as priming fluid in cardiopulmonary bypass–a randomised controlled trial. Perfusion 22(1):57–61
Acknowledgments
This study was supported by the fund of National Nature Science Foundation of China (30772092). The authors indicate no financial conflict of interest. We thanks Tomi Niemi for providing numerical data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xue-Yin Shi and Zui Zou contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shi, XY., Zou, Z., He, XY. et al. Hydroxyethyl starch for cardiovascular surgery: a systematic review of randomized controlled trials. Eur J Clin Pharmacol 67, 767–782 (2011). https://doi.org/10.1007/s00228-011-1008-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-011-1008-5