Abstract
Objective
To describe in detail the intravenous, single oral and multiple oral dose enantioselective pharmacokinetics of tramadol in CYP2D6 extensive metabolizers (EMs) and poor metabolizers (PMs).
Methods
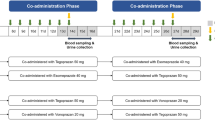
Eight EMs and eight PMs conducted three phases as an open-label cross-over trial with different formulations; 150 mg single oral tramadol hydrochloride, 50 mg single oral tramadol hydrochloride every 8 h for 48 h (steady state), 100 mg intravenous tramadol hydrochloride. Urine and plasma concentrations of (+/−)-tramadol and (+/−)-M1 were determined for 48 h after administration.
Results
In all three phases, there were significant differences between EMs and PMs in AUC and t1/2 of (+)-tramadol (P≤0.0015), (−)-tramadol (P≤0.0062), (+)-M1 (P≤0.0198) and (−)-M1 (P≤0.0370), and significant differences in Cmax of (+)-M1 (P<0.0001) and (−)-M1 (P≤0.0010). In Phase A and C, significant differences in tmax were seen for (+)-M1 (P≤0.0200). There were no statistical differences between the absolute bioavailability of tramadol in EMs and PMs. The urinary recoveries of (+)-tramadol, (−)-tramadol, (+)-M1 and (−)-M1 were statistically significantly different in EMs and PMs (P<0.05). Median antimodes of the urinary metabolic ratios of (+)-tramadol / (+)-M1 and (−)-M1 were 5.0 and 1.5, respectively, hereby clearly separating EMs and PMs in all three phases.
Conclusion
The impact of CYP2D6 phenotype on tramadol pharmacokinetics was similar after single oral, multiple oral and intravenous administration displaying significant pharmacokinetic differences between EMs and PMs of (+)-tramadol, (−)-tramadol, -(+)-M1 and (−)-M1. The O-demethylation of tramadol was catalysed stereospecific by CYP2D6 in the way that very little (+)-M1 was produced in PMs.


Similar content being viewed by others
References
Frankus E, Friderichs E, Kim SM, Osterloh G (1978) On separation of isomeres, structural elucidation and pharmacological characterization of 1-(m-methoxyphenyl)-2-(dimethylaminomethyl)-cyclohexan-1-ol. Arzneimittelforschung 28:114–121
Shipton EA (2000) Tramadol - present and future. Anaesth Intensive Care 28:363–374
Wu WN, Mckown LA, Liao S (2002) Metabolism of the analgesic drug ULTRAM (R) (tramadol hydrochloride) in humans: API-MS and MS/MS characterization of metabolites. Xenobiotica 32:411–425
Lintz W, Erlacin S, Frankus E, Uragg H (1981) Biotransformation of tramadol in man and animal. Arzneimittelforschung 31:1932–1943
Subrahmanyam V, Renwick AB, Walters DG, Young PJ, Price RJ, Tonelli AP et al (2001) Identification of cytochrome P-450 isoforms responsible for cis-tramadol metabolism in human liver microsomes. Drug Metab Dispos 29:1146–1155
Raffa RB, Friderichs E, Reimann W, Shank RP, Codd EE, Vaught JL (1992) Opioid and nonopioid components independently contribute to the mechanism of action of tramadol, an atypical opioid analgesic. J of Pharmacol Exp Ther 260:275–285
Grond S, Sablotzki A (2004) Clinical pharmacology of tramadol. Clin Pharmacokinetics 43:879–923
Gillen C, Haurand M, Kobelt DJ, Wnendt S (2000) Affinity, potency and efficacy of tramadol and its metabolites at the cloned human mu-opioid receptor. Naunyn-Schmiedebergs Arch Pharmacol 362:116–21. DOI 10.1007/s002100000266
Lai J, Ma SW, Porreca F, Raffa RB (1996) Tramadol, M1 metabolite and enantiomer affinities for cloned human opioid receptors expressed in transfected HN9.10 neuroblastoma cells. Eur J Pharmacol 316:369–732
Raffa RB, Friderichs E, Reimann W, Shank RP, Codd EE, Vaught JL et al (1993) Complementary and synergistic antinociceptive interaction between the enantiomers of tramadol. J of Pharmacol Exp Ther 267:331–340
Bamigbade TA, Davidson C, Langford RM, Stamford JA (1997) Actions of tramadol, its enantiomers and principal metabolite, O-desmethyltramadol, on serotonin (5-HT) efflux and uptake in the rat dorsal raphe nucleus. Br J Anaesth 79:352–356
Halfpenny DM, Callado LF, Hopwood SE, Bamigbade TA, Langford RM, Stamford JA (1999) Effects of tramadol stereoisomers on norepinephrine efflux and uptake in the rat locus coeruleus measured by real time voltammetry. Br J Anaesth 83:909–915
Bradford LD (2002) CYP2D6 allele frequency in European Caucasians, Asians, Africans and their descendants. Pharmacogenomics 3:229–243
Halling J, Petersen M, Damkier P, Nielsen F, Grandjean P, Weihe P et al (2005) Polymorphism of Cyp2D6, Cyp2C19, Cyp2C9 and Cyp2C8 in the Faroese population. Eur J Clin Pharmacol 61:491–497. DOI 10.1007/s00228-005-0938-1
Paar WD, Poche S, Gerloff J, Dengler HJ (1997) Polymorphic CYP2D6 mediates O-demethylation of the opioid analgesic tramadol. Eur J Clin Pharmacol 53:235–239
Poulsen L, Arendt-Nielsen L, Brosen K, Sindrup SH (1996) The hypoalgesic effect of tramadol in relation to CYP2D6. Clin Pharmacol Ther 60:636–644
Pedersen RS, Brosen K, Nielsen F (2003) Enantioselective HPLC method for quantitative determination of tramadol and O–desmethyltramadol in plasma and urine: application to clinical studies. Chromatographia 57:279–285
Pedersen RS, Damkier P, Brosen K (2005) Tramadol as a new probe for cytochrome P450 2D6 phenotyping: apopulation study. Clin Pharmacol Ther 77:458–67. DOI 10.1016/j.clpt.2005.01.014
Laugesen S, Enggaard TP, Pedersen RS, Sindrup SH, Brosen K (2005) Paroxetine, a cytochrome P450 2D6 inhibitor, diminishes the stereoselective O-demethylation and reduces the hypoalgesic effect of tramadol. Clin Pharmacol Ther 77:312–323
Fliegert F, Kurth B, Gohler K (2005) The effects of tramadol on static and dynamic pupillometry in healthy subjects-the relationship between pharmacodynamics, pharmacokinetics and CYP2D6 metaboliser status. Eur J Clin Pharmacol 61:257–266. DOI 10.1007/s00228–005–0920–y
Stamer UM, Lehnen K, Hothker F, Bayerer B, Wolf S, Hoeft A et al (2003) Impact of CYP2D6 genotype on postoperative tramadol analgesia. Pain 105:231–238. DOI 10.1016/S0304-3959(03)00212-4
Schellens JHM, Vanderwart HHF, Hoevers JW, Breimer DD (1988) Gas-chromatographic determination of sparteine and 2-Dehydrosparteine and 5-Dehydrosparteine in plasma and urine. J Chromatogr - Biomedical Applications 431:203–209
Brosen K, Otton SV, Gram LF (1985) Sparteine oxidation polymorphism in Denmark. Acta Pharmacol Toxicol 57:357–360
Kagimoto M, Heim M, Kagimoto K, Zeugin T, Meyer UA (1990) Multiple mutations of the human cytochrome-P450Iid6 gene (Cyp2D6) in poor metabolizers of debrisoquine - study of the functional-significance of individual mutations by expression of chimeric genes. J Biol Chem 265:17209–17214
Marez D, Legrand M, Sabbagh N, LoGuidice JM, Spire C, Lafitte JJ et al (1997) Polymorphism of the cytochrome P450 CYP2D6 gene in a European population: characterization of 48 mutations and 53 alleles, their frequencies and evolution. Pharmacogenetics 7:193–202
Hanioka N, Kimura S, Meyer UA, Gonzalez FJ (1990) The human Cyp2D locus associated with a common genetic-defect in drug oxidation: a G1934–A base change in intron-3 of a mutant Cyp2D6 allele results in an aberrant-3′ splice recognition site. Am J Hum Genet 47:994–1001
Gough AC, Miles JS, Spurr NK, Moss JE, Gaedigk A, Eichelbaum M et al (1990) Identification of the primary gene defect at the cytochrome-P450 Cyp2D locus. Nature 347:773–776
Evert B, Griese EU, Eichelbaum M (1994) Cloning and sequencing of a new nonfunctional Cyp2D6 allele - deletion of T-1795 in exon-3 generates a premature stop codon. Pharmacogenetics 4:271–274
Daly AK, Leathart JBS, London SJ, Idle JR (1995) An inactive cytochrome-P450 Cyp2D6 allele containing a deletion and a base substitution. Hum Genet 95:337–341
Saxena R, Shaw GL, Relling MV, Frame JN, Moir DT, Evans WE et al (1994) Identification of a new variant Cyp2D6 allele with a single-base deletion in exon-3 and its association with the poor metabolizer phenotype. Hum Mol Genet 3:923–926
Tyndale R, Aoyama T, Broly F, Matsunaga T, Inaba T, Kalow W et al (1991) Identification of a new variant Cyp2D6 allele lacking the codon encoding Lys-281-possible association with the poor metabolizer phenotypee. Pharmacogenetics 1:26–32
Broly F, Meyer UA (1993) Debrisoquine oxidation polymorphism - phenotypic consequences of a 3-base-pair deletion in exon 5 of the Cyp2D6 gene. Pharmacogenetics 3:123–130
Lintz W, Barth H, Becker R, Frankus E, Schmidt-Bothelt E (1998) Pharmacokinetics of tramadol and bioavailability of enteral tramadol formulations-2nd communication: drops with ethanol. Arzneimittelforschung 48:436–445
Lintz W, Barth H, Osterloh G, Schmidtbothelt E (1986) Bioavailability of enteral tramadol formulations. 1. Capsules. Arzneimittelforschung 36–2:1278–1283
Lintz W, Barth H, Osterloh G, Schmidt-Bothelt E (1998) Pharmacokinetics of tramadol and bioavailability of enteral tramadol formulations, 3rd communication: suppositories. Arzneimittelforschung 48:889–899
Enggaard TP, Poulsen L, Arendt-Nielsen L, Brosen K, Ossig J, Sindrup SH (20061) The analgesic effect of tramadol after intravenous injection in healthy volunteers in relation to CYP2D6. Anesth Analg 102:146–150. DOI 10.1213/01.ane.0000189613.613.61910.32
Klotz U (2003) Tramadol - the impact of its pharmacokinetic and pharmacodynamic properties on the clinical management of pain. Arzneimittelforschung 53):681–687
Bamigbade TA, Langford RM (1998) Tramadol hydrochloride: an overview of current use. Hosp Med 59:373–376
Lee CR, Mctavish D, Sorkin EM (1993) Tramadol: a preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in acute and chronic pain states. Drugs 46:313–340
Paar WD, Frankus P, Dengler HJ (1992) The metabolism of tramadol by human liver-microsomes. Clinical Investigator 70:708–710
Liu HC, Wang N, Yu Y, Hou YN (2003) Stereoselectivity in trans-tramadol metabolism and trans-O-demethyltramadol formation in rat liver microsomes. Acta Pharmacol Sin 24:85–90
Lintz W, Becker R, Gerloff J, Terlinden R (2000) Pharmacokinetics of tramadol and bioavailability of enteral tramadol formulations-4th Communication: Drops (without ethanol). Arzneimittelforschung 50:99–108
Acknowledgements
We gratefully thank The Alfred Benzon Foundation, The Lundbeck Foundation, The Danish Medical Research Council and The Danish Clinical Intervention Research Academy for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pedersen, R., Damkier, P. & Brøsen, K. Enantioselective pharmacokinetics of tramadol in CYP2D6 extensive and poor metabolizers. Eur J Clin Pharmacol 62, 513–521 (2006). https://doi.org/10.1007/s00228-006-0135-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-006-0135-x