Abstract
Understanding growth rates and other basic life-history information of imperilled species is essential to assessing the extent of threats to a population, but often difficult due to limited access to study subjects. Here we used mark-recapture data to estimate growth rates of juvenile Critically Endangered green sawfish (Pristis zijsron) in a globally important nursery in the eastern Indian Ocean (Western Australia). Our results suggest that growth of juvenile sawfish in this part of the central Western Australian coast is significantly slower compared to populations on the north-eastern coast of Australia. Additionally, growth rates differed between nearby areas within the nursery region, potentially due to differential productivity or anthropogenic effects. Morphological relationships between total length, rostral length, mouth gape, and clasper length are presented, which will allow for greater accuracy in estimating biological parameters in this species, while updated information on size at maturity (> 3200 mm) and size at birth (approximately 750–900 mm) will help to clarify life-history parameters for this data-poor species. Furthermore, there were distinct differences in the number of rostral teeth of green sawfish between this eastern Indian Ocean population and other populations throughout their current distribution, indicating substantial genetic differentiation in this species globally. These results will help to accurately assess growth trajectories and potential impacts of fisheries and other threats to green sawfish. Additionally, results highlight the importance of assessing population-specific growth rates in threatened species and of considering potential long-term life-history impacts of anthropogenic developments and activities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding basic biological and life-history information such as growth rates and generation length in aquatic species is crucial to accurately assessing population productivity and forecasting responses or resilience to environmental and anthropogenic pressures (Hobday et al. 2011; Goldman et al. 2012; Maunder and Punt 2013). This is particularly true for threatened species, where accurate assessments of potential population recovery rates or sustainable levels of fisheries interactions are essential to promoting proper management and recovery of declining populations (Dulvy et al. 2004; Cortés 2016). However, collecting basic biological information for threatened species is often difficult due to limited access to species that are naturally rare or have already undergone large population declines. Additionally, when working with threatened species there is limited potential for lethal sampling methods for assessing growth and reproductive parameters, which are often used in aquatic research. For example, growth curves for fishes are typically formed using length-at-age data calculated by measuring growth rings in calcified structures including otoliths or vertebrae (e.g. Campana 2001; Maceina et al. 2007), which often require lethal sampling of individuals.
Assessing growth rates through sequential recaptures of individuals offers a non-lethal method of measuring size development (e.g. Hamel et al. 2014; Meyer et al. 2014; Dureuil and Worm 2015). However, this technique requires access to individuals over extended time periods, which can be difficult when working with rare or migratory species, and requires animals to exhibit some level of site fidelity or residency to particular areas. Additionally, particularly in species which use different habitats during ontogeny, this can result in biased sampling of individuals, where, for example, juveniles using coastal nursery habitats are caught more often than adults using offshore habitats, or vice versa. Fortunately, recent advances in statistical modelling have shown that Bayesian methods for estimating growth, which can incorporate prior knowledge including lengths at birth and maximum lengths of a species, can still produce reasonably accurate growth models using age or size-biased data (Scherrer et al. 2021; Smart and Grammer 2021; Dureuil et al. 2022). Such techniques provide the opportunity to assess growth trajectories of a species or population from relatively sparse or biased recapture data, which is common in the case of threatened aquatic species.
The green sawfish (Pristis zijsron) is one such threatened species; currently considered Critically Endangered by the International Union for the Conservation of Nature (IUCN) and estimated to have undergone a more than 80% decline in global population size over the last 50 years (Harry et al. 2022). The species formerly ranged from South Africa across the western Indian Ocean to south-east Asia and Australia. While remnant populations exist throughout the Indo-Pacific, Australia is currently thought to host some of the last viable populations. Within Australia, green sawfish were historically found from the central Pacific east coast (New South Wales), around the northern continent to as far south-west as Shark Bay in the eastern Indian Ocean (Western Australia). However, populations in north-eastern Australia have declined substantially, with this species presumably extinct through much of its former east coast range (Wueringer 2017; Harry et al. 2022). In contrast, this species is still regularly found throughout most of its Western Australian range, with pupping reported along the expansive and sparsely populated coastline of northwest Australia (Stevens et al. 2008; Morgan et al. 2011, 2015). In particular, the Ashburton River region in the southern Pilbara, Western Australia, has been documented as a productive and consistent nursery habitat where pupping reliably occurs on an annual basis (Morgan et al. 2015), and where individuals remain throughout their juvenile phase until they reach a length of approximately 3 m (Morgan et al. 2017). A preliminary growth curve for this species has previously been estimated using length-at-age data from 18 specimens retrieved from fisheries interactions in north-eastern Australia (Peverell 2009), but there is genetic differentiation between green sawfish on the north-east and west coasts of Australia (Phillips et al. 2011), and growth rates have not been examined for this species in the south-western part of their Australian range. Growth rates for numerous taxa can vary widely between populations dependent on a number of factors, such as temperature and resource availability (Weatherley 1990; Hewett and Kraft 1993; Jobling 1997). As such, it is important to examine growth rates for what is potentially one of the last remaining, relatively robust populations of green sawfish globally, to ensure that accurate growth rates can be incorporated into population assessments.
Here, we use mark-recapture data collected over a four-year period to estimate growth rates of green sawfish in a globally significant population in north-western Australia. Bayesian methods were used to construct a von Bertalanffy growth curve from mark-recapture data, which was then compared with growth rates estimated for green sawfish in north-eastern Australia. Additionally, body condition, size at birth, maturity, and morphometric and meristic characteristics of this population are reported and compared with other populations globally.
Methods
Sawfish capture and measurement
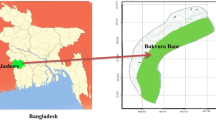
Green sawfish were captured in the Ashburton River and nearby lagoons and tidal mangrove creeks in the Pilbara region of Western Australia (Fig. 1). Surveys were conducted in mid-spring to early summer (Oct–Dec) and autumn (Apr–May) in 2011 and 2019–2022. Sawfish were captured using gillnets of 60 m length and 150 mm mesh size, set perpendicular to the bank. Nets were checked when activity was observed or at least once per hour. Upon capture, sawfish were removed from the net, sexed, and measured for total length (TL), total rostrum length (TRL), standard rostrum length (SRL), mouth gape (distance between corners of the mouth), inner and outer clasper length (in males; left clasper) and girth, which was measured immediately posterior to the pectoral fins and anterior to the first dorsal fin (Fig. 2). Additionally, the number of rostral teeth was counted, and sawfish were weighed to the nearest 100 g. Individuals were then externally tagged with either a small T-bar tag (4.5 cm length; model TBF, Hallprint Fish Tags, South Australia) or a larger dart tag (15 cm length; model PDAT, Hallprint Fish Tags, South Australia) depending on sawfish size. Alternatively, if the sawfish was a recapture, its tag number was noted. All sawfish were released at their site of capture.
For compilation of morphometric and meristic data, incidental captures of green sawfish in other areas of Western Australia (Exmouth Gulf, Fortescue River estuary, and Kimberley) during related work (2018–2022) were included (see Fig. 1).
Data analysis
Mark-recapture data were analysed in R (v. 4.0.3; R Foundation for Statistical Computing, Vienna, Austria). A von Bertalanffy growth curve was constructed out of the mark-recapture data using the R package ‘GrowthEstimation’ (Dureuil et al. 2022), which implements the Fabens method (Fabens 1965) of constructing a von Bertalanffy curve using Bayesian techniques. This requires the input of an estimated length at birth (L0) and maximum length (L∞). We estimated L0 at 760 mm based on capture of a 760 mm individual in the Ashburton River that possessed a remnant rostral sheath and remnants of a yolk-sac, and the reported mean size at birth of 760 mm in the Gulf of Carpentaria (Peverell 2009). The mean L∞ was estimated at 5500 mm, with the maximum L∞ estimated at 6000 mm; while some literature suggests this species can attain lengths of over 7000 mm, verified records of individuals over 6000 mm are rare (Simpfendorfer 2013). Only recaptures with more than 90 days between captures were used to assess growth rates. Differences in annual growth rates (mm year−1) between males and females were investigated using an Analyses of Variance (ANOVA); these were then further examined by running separate sets of Bayesian analyses to form von Bertalanffy curves for recaptured males and females, and examining overlaps in 95% confidence intervals (CIs) of estimated parameters. Additionally, we tested whether there were differences in growth rates and body condition between individuals inhabiting different areas within the main study region (the Ashburton River mouth vs Hooley Lagoon vs Four Mile Creek; no sawfish inhabiting Hooley Creek were recaptured more than 90 days apart). An individual was assigned to a specific location if both its initial capture and recapture were at that location, and if unpublished (and published; Morgan et al. 2017) acoustic tracking data indicated that the individual inhabited predominantly that location between captures. If an individual was recaptured at a different site to its initial capture, or if acoustic tracking data indicated that the individual moved amongst several sites, the individual was assigned to a ‘mixed-location’ group. Some young-of-year individuals were not acoustically tracked, but were assumed to remain within their initial capture location if recaptured at that same location, due to limited movement of individuals of this size (Morgan et al. 2017). As with inter-sex differences, differences in growth rates between locations were examined first through an ANOVA and subsequently through overlaps in 95% CIs of von Bertalanffy parameters calculated for recaptures from different sites.
To determine whether location, sex, or time of year influenced sawfish body condition, we built a set of linear models in R estimating girth using total length (both parameters natural log transformed), with capture location, season (autumn or spring/summer), and sex included as additional predictor variables. The Akaike’s information criterion corrected for small sample size (AICc) was used to determine the model with the best fit combination of these predictor variables, with predictors that were included in the best-fit model deemed influential.
Additionally, morphometric data were tabulated and presented, including predictive relationships between total length and rostrum lengths (SRL and TRL), mass, girth, and mouth gape, as well as clasper length for males. To determine the best predictive equation for mass, again several potential linear models were built in R. This test set of models included those using either length, girth, or both length and girth as predictor variables, and every combination of untransformed or log-transformed predictors (length and girth) and response (mass). The best fit equation was determined using AICc. Similarly, to determine the best fit equation for describing the relationship between TL and various predictors (TRL, SRL, mouth gape, and clasper length), a series of linear models were built with log transformed and untransformed predictors (TRL, SRL, mouth gape, or clasper length) and response (TL), and with sex included either as a fixed predictor, as an interactive factor with the fixed predictor, or not included. AICc was used to determine the best fit model for each of these predictive relationships. Linear model assumptions were checked using residual plots for all models.
Results
A total of 143 unique green sawfish were captured within the Ashburton River region, with a further 9 individuals captured elsewhere in Western Australia included in morphometric analyses. The smallest individual caught was a female of 651 mm total length. However, this individual was substantially (> 100 mm) smaller than any other individual caught, had notably lower rostral tooth counts (20 teeth per side), and appeared underweight and unhealthy, suggesting potential developmental defects or premature birth. This individual was therefore excluded from all morphometric analyses. The remaining individuals ranged from 751 to 3228 mm total length.
The sex ratio of captures was slightly biased towards females across juvenile age classes, including when considering all individuals caught (1.2:1 F:M juvenile ratio) and exclusively young-of-year (YOY) individuals (1.3:1 F:M YOY ratio). All males captured were immature based on uncalcified clasper morphology, with the largest of these measured at 3228 mm TL. The largest female caught was 3195 mm TL; it is likely that all females caught in the study were also immature based on the immaturity of similar sized males. Therefore, all results are based on analyses of strictly juvenile green sawfish.
Young-of-year pups with visible (open or partially healed) yolk-sac scars were caught in October, November, and December in the Ashburton River region. Excluding the 651 mm individual previously described with potential morphological defects, these young-of-year pups ranged in size from 751 to 972 mm. Of these, pups with fully or partially open yolk-sac scars ranged from 751–894 mm TL. Additionally, three newborn pups (with remnant rostral sheaths and partially open yolk-sac scars) were caught in the south-western Exmouth Gulf in September 2021 (804–843 mm TL), and one pup with a visible yolk-sac scar was caught in the Fortescue River estuary in August 2022 (790 mm TL). Together, this information suggests an estimated pupping period of August–December in the southern Pilbara region of the eastern Indian Ocean, and an approximate size at birth of 750–900 mm TL.
Recaptures and growth rates
A total of 19 individuals were recaptured during the study over periods of up to 734 days, with three individuals recaptured twice (22 recaptures total). The mean (± SD) duration of the time between captures was 295 ± 170 days, and recaptured individuals ranged in size from 880 to 3228 mm.
Bayesian analyses of all recaptures indicated von Bertalanffy growth parameters of k = 0.090 (95% CI: 0.074–0.11), and L∞ = 555 (95% CI: 517–597) (Fig. 3). Growth rates did not differ between males and females (again noting that all individuals recaptured were immature; ANOVA p = 0.4, 95% CIs fully overlap predicted values). However, growth rates were significantly different between locations within the main study area, with growth rates higher in the Ashburton River mouth and mixed location individuals compared to Hooley Lagoon or Four Mile Creek (ANOVA p < 0.05; 95% CIs of predicted values in Bayesian analyses did not overlap means) (Fig. 3). There was no difference in sawfish body condition between sexes, capture locations, or seasons, with total length included as the only predictor of girth in the final body condition model (Supplemental Table S1).
Growth rates of juvenile green sawfish Pristis zijsron recaptured in the Ashburton River (green), Hooley Lagoon (blue), Four Mile Creek (purple), or in mixed locations (yellow). A Estimated von Bertalanffy growth rate (black line) for combined Ashburton River, Hooley Lagoon and Four Mile Creek captures, based on Bayesian analysis of recapture data; grey shaded region denotes 95% confidence intervals. Connected points show growth of recaptures, and are coloured according to capture location; solid points denote individuals first caught as young-of year, where age could be estimated within months (assuming an October 15th birth date), and open points show individuals where age was estimated based on probable growth patterns of cohorts (all assuming an October 15th birth date). The red dashed line shows the growth rate for green sawfish in north-eastern Australia estimated by Peverell (2009). B Median growth rates of juveniles inhabiting different locations within the study (black horizontal bar), with boxes showing the first and third quartiles of growth, whiskers showing growth limits within 1.5 inter-quartile ranges of the median, and any outlying values shown with points
Estimation of mass
Mass, length, and girth were measured for 79 individuals, which provided the data to assess estimation of mass from TL and girth measurements (Fig. 4). Sex was not maintained as a predictor in the best fit model, indicating that estimation of mass using length and girth measurements did not differ between juvenile males and females (see Supplemental Table S1 for model selection results). The best fit equation (based on AICc) for estimation of mass (kg) using TL and girth (mm) was:
In most cases, this equation predicted mass with relative accuracy, resulting in a mean standard error of the estimate of 11.7%. As many studies do not measure girth, we also report here the best fit equation for predicting mass solely with length measurements:
It is notable, however, that this method for estimating mass resulted in a slightly higher mean standard error of the estimate of 13.0%. Therefore, incorporating girth measurements into estimation of mass likely yields more accurate results.
Morphometric and meristic measurements
Measurements of TRL, SRL, and TL were collected from 170 sawfish (including recaptures). The ratio of TRL and SRL to TL were both size (regression p < 0.01) and sex dependent (regression p < 0.05), with the rostrum making up slightly but significantly less of the TL in males compared to females, and as fish grew larger (Fig. 5). An interaction between sex and size was also significant for SRL (p = 0.012), indicating that the rate at which the ratio between SRL and TL changed with size was different between males and females. The best fit equations for predicting TL using TRL or SRL for both sexes are noted in Fig. 5 (see Supplemental Table S1 for model selection results).
Relationships between green sawfish Pristis zijsron total length (TL) and A standard rostral length (SRL); B total rostral length (TRL); C mouth gape; or D inner clasper length. Both sex and size had a slight but significant effect on the ratio of TRL and SRL to TL, but the relationship between mouth gape and total length did not differ between sexes. Best fit equations and R2 values are noted on each plot
The predictive relationships for mouth gape and clasper length (inner) using TL were both improved by applying natural log transformed predictor and response variables (Supplemental Table S1). Sex did not affect the relationship between mouth gape and total length. Both mouth gape and clasper length had a high correlation with total length (R2 > 0.95; Fig. 5), with the best fit predictive equations shown in Fig. 5. Note that all clasper lengths included in this analysis were from immature individuals.
A total of 148 unique individuals were included in tooth count analyses. Sawfish possessed between 23 and 30 rostral teeth per side, with the exception of the previously noted female individual excluded due to potential developmental defects (which had 20 teeth on both sides of the rostrum). Otherwise, female tooth counts ranged from 23–29 teeth per side, while males ranged from 23–30 teeth per side, noting that only a single male had 30 teeth on one side of the rostrum. Females had, on average, significantly fewer teeth than males (Wilcoxon rank sum p = 0.012) (Fig. 6).
Number of rostral teeth per side in green sawfish Pristis zijsron. Tooth counts for green sawfish populations globally A were in some cases substantially higher than tooth counts measured for either females B or males C in Western Australian individuals. Vertical dashed lines (plots B & C) show the respective mean tooth count values for each sex. Size of points in (A) relates to the percentage of sawfish showing that number of teeth within the dataset; only a range rather than distribution of rostral tooth counts is available for the Sudanese Red Sea (Elhassan 2018). Sources in (A) are as follows: aJabado et al. (2017); bElhassan (2018); cLeeney (2017); dFaria et al. (2013); eWhitty et al. (2014). Note that ‘Australia’ in (A) from Whitty et al. (2014) refers to all of northern Australia, including Western Australia, the Northern Territory, and Queensland, but does not necessarily include individuals from the current study area
Discussion
Western Australia currently supports the majority of the known robust populations of green sawfish remaining within their previous Indo-Pacific range (Harry et al. 2022), with the Ashburton River in the eastern Indian Ocean identified as an extremely important pupping and nursery area for this species (Morgan et al. 2015, 2017). While parts of the Pilbara region serve as important hubs for the Australian resource sector, this area also supports large sections of relatively pristine and undeveloped coastline. However, there is rapidly mounting pressure to further develop the region for salt, oil and gas, and iron ore mining, as well as several trawl and gillnet fisheries operating nearby. The growth rates and life-history information presented here will help to accurately assess the extent and mitigation of threats as well as the potential for population recovery and promotion of effective conservation measures. For example, the determination of a pupping period of approximately August – December for this species in the Pilbara region can inform scheduling for potential developments including times of year to limit construction or dredging around pupping habitats to avoid interference with female migrations and parturition behaviour.
Size at birth and maturity
The size at birth of green sawfish observed here (approximately 750–900 mm TL) roughly agrees with most other accounts recorded for this species throughout its range, although there are a few reports of smaller neonates as well. For example, similarly to the current study, Peverell (2009) reports a size at birth of 760 mm in the Gulf of Carpentaria (south of the Arafura Sea, north-eastern Australia), and Elhassan (2018) reports full-term embryos from the Sudanese Red Sea measuring 600–800 mm TL and neonates measuring 810–890 mm TL. However, Faria (2007) reports specimens from Indonesia as small as 680 mm TL, although these were collected in the late 1800s, and therefore whether the individual was an embryo or neonate and the degree of shrinkage is unknown. Morgan et al. (2011) also reports the collection of a green sawfish rostrum from Western Australia (eastern Indian Ocean) measuring 160 mm, which would equate to a TL of approximately 606 mm based on the TRL-TL conversion established here; however, whether this rostrum was from a neonate or a premature birth cannot be determined. Additionally, a recent study within Australia’s Northern Territory observed individuals estimated as small as 570 mm TL (Davies et al. 2022), but these measurements were approximated from drone footage rather than directly measured. Overall, these observations indicate that size at birth is similar for green sawfish across their extant distribution, and likely averages approximately 750–900 mm, with occasional smaller individuals recorded.
In compiling the data presented here with previous published records, we can also narrow the probable size at maturity for this species. Our capture of an immature male of 3228 mm (with short and fully uncalcified claspers) sets a lower limit to the size at maturity for males. Previous minimum size records of mature individuals include mature females at 3500 and 3800 mm TL in the Sudanese Red Sea (Elhassan 2018) and north-eastern Australia (Peverell 2005), respectively, and a mature male at 3800 mm in the Sudanese Red Sea (Elhassan 2018). Together, this indicates a size at maturity for males between 3300 and 3800 mm TL, and likely a similar size range for females. While these ranges agree with the published lengths of mature individuals in primary literature, several reference volumes and articles list other sizes at maturity as rough estimates due to a lack of empirical data, including maturity for both sexes at 3000 mm (e.g. Last and Stevens 2009; Morgan et al. 2011) or 4300 mm (Last et al. 2016). Based on the sizes of immature and mature individuals in this study and recently published literature, these rougher estimates appear to be inaccurate.
Growth rates
To our knowledge, the only previous estimate of growth rates in green sawfish was constructed using vertebral aging data from 18 individuals of 870–4490 mm caught in the Gulf of Carpentaria in north-eastern Australia (Peverell 2009). The estimated growth rate calculated here was significantly slower than this previous estimate (95% CIs of growth parameter k did not overlap k reported by Peverell (2009)), to the extent that time to maturity (assuming a size of 3500 mm at maturity) increases from 7–8 years as estimated by Peverell (2009) to 9–10 years estimated in the current study. Both the previous and current estimation of growth rates are based on somewhat small sample sizes, and can be interpreted as preliminary growth curves for this species. Therefore, the error in both these studies, inherent of small sample sizes, could partially account for the observed differences in growth rates, as well as the error incurred by using two different methods to form growth curves. However, there are also several biological explanations for the differences in growth between these two populations. For example, there is genetic differentiation between green sawfish populations in north-eastern Australia compared to the eastern Indian Ocean in Western Australia (Phillips et al. 2011), and therefore the different growth rates could stem from a genetic basis.
There are also several environmental parameters that could affect growth. Life history parameters including growth rates have been known to vary between fish populations dependent on many factors including resource availability and temperature (e.g. Lahti et al. 2001; Carlson et al. 2006; Gillanders et al. 2015; Uthe et al. 2016; Marchand et al. 2018; Chittaro et al. 2022). Since elevated temperatures increase vital rates in ectotherms (Gillooly et al. 2001; 2002), growth rates generally increase with temperature (up to a point) as long as there is sufficient food availability to sustain heightened metabolic requirements. Water temperatures in the Gulf of Carpentaria are on average approximately 2 °C warmer than the coastal Pilbara region, depending on time of year (seatemperatures.net). Therefore, these warmer temperatures could account for the faster growth estimated for green sawfish in north-eastern Australia compared to the Pilbara region. Additionally, while it is difficult to directly compare productivity or prey availability between the two systems given a lack of data, it is likely that there is higher productivity in the Gulf of Carpentaria compared to the Pilbara region. The Gulf of Carpentaria is located in the wet and dryland tropics of Australia and has several major rivers that consistently discharge into nearshore environments, bringing terrestrial nutrient input into these areas (and resulting in higher productivity; Gillanders and Kingsford 2002; Broadley et al. 2022). In comparison, the southern Pilbara region in the present study comprises an arid desert with limited terrestrial run-off; the few rivers in this region flow infrequently for only a few days at a time following sporadic rainfall events. Furthermore, coastal waters in Western Australia have been found to be generally less productive than those elsewhere in Australia due to limited terrestrial run-off as well as interactions of major currents and limited upwelling in Western Australia (Molony et al. 2011). Therefore, differential productivity and resource availability may also account for the differences observed in the growth of green sawfish between north-eastern and Western Australia.
Differential productivity between sites is also a likely explanation for the heightened growth observed in the Ashburton River compared to the two smaller tidal mangrove creeks in the present study. These sites are geographically close (within ~ 12 km of each other), with larger individuals known to transit between them, as observed from recapture data here as well as acoustic tracking data (Morgan et al. 2017). Therefore, a genetic basis for differences in growth rates between sites does not seem likely. Additionally, water temperatures between the sites are similar year-round and therefore not likely to drive major differences in growth rates. However, these sites are characterised by distinct environmental circumstances: the Ashburton River mouth has sporadic freshwater flows and dense mangroves; Hooley Lagoon is a shallow mudflat lagoon which is connected to small mangrove creeks; and Four Mile Creek is a mangrove creek system. The Ashburton River mouth is the only major input of terrestrial nutrients for several hundred kilometres of coastline, and therefore probably offers heightened productivity compared to the Hooley Lagoon and Four Mile Creek. This is likely to be the most influential driver of differences in growth rates between sites, and further research into how productivity rates and prey availability vary seasonally and between these systems would help to confirm this. Additionally, Hooley Lagoon and Four Mile Creek are both near to developments (a gas processing plant and boat ramp, respectively), and any heightened noise, light, or human activity associated with these areas could have the potential to decrease growth rates if these factors increase refuging behaviour or affect foraging success (Becker et al. 2013; Davies et al. 2014; Voellmy et al. 2014). Comparatively, there is little human activity around the mouth of the Ashburton River due to lack of access (no paved roads or maintained boat launches; Authors, personal observation). In the case of Hooley Lagoon, a large offloading facility is situated adjacent to this site, with a rock wall extending ~ 250 m offshore that appears to limit movement of sawfish to the northeast (Lear and Morgan, unpublished data), and therefore could be artificially aggregating sawfish within the Hooley Lagoon area, with potential consequences for resource availability. However, it is difficult to determine whether any of these anthropogenic effects may be impacting growth, as no investigation of growth rates was conducted in this population before these boat ramps and developments were constructed. Whether anthropogenic noise, light, or artificial structures influence sawfish behaviour, feeding ecology, or growth is an important area for future research. Regardless of whether differences in resource availability, anthropogenic activities, or a combination thereof drive the differences in growth between nursery sites, the lack of significant difference in sawfish body condition between sites suggests that sawfish at all locations are obtaining sufficient energy to sustain condition and positive growth.
The similarities in growth rates between sexes found here is not unexpected, given that all animals examined were juveniles. Previous studies have found similar growth rates between sexes in a variety of fishes during early life stages, even when adult sizes differ (Simpfendorfer 2000; Curtis and Vincent 2006); sex-specific growth rates are more often found in larger animals reaching maturity (e.g. Simpfendorfer 1993; Simpfendorfer et al. 2000; Williams et al. 2012). However, it is notable that our sample sizes for growth in individual sexes were small (N = 8 or 14 recaptures) and could have obscured subtle differences in growth rates. Further investigation of sex specific growth rates and size at maturity in this species would help to clarify this point.
Morphometric relationships
The morphological relationships determined here will be useful in estimating morphometric parameters in future studies when specific measurements are missing. For example, the relationships between TRL/SRL and TL are especially important, as many “sightings” of sawfish globally are from collected rostra rather than live animals (e.g. Morgan et al. 2011; Whitty et al. 2014; Wueringer et al. 2023), and these relationships help to estimate the size of these animals from collected rostra alone. Similarly, as the mass of sawfish is not often measured due to impracticality in the field, the ability to estimate mass from measurements of girth and TL can lend important biological information about these animals, particularly regarding trophic interactions and energetics. Estimation of mouth gape from total length also provides biologically pertinent information as it enables an assessment of what prey size sawfish of different size classes are likely to target.
Although previous data describing morphological relationships in green sawfish are limited, the relationships determined here are relatively similar to the few previously reported. For example, in the current study (eastern Indian Ocean), TRL made up 23.1– 29.4% of TL, and TRL similarly made up 24.0–30.6% of TL in nine individuals sampled from elsewhere in the Indian Ocean (Faria 2007), though Faria et al. (2013) measured TRL as 22.9–33.6% of TL including individuals from both the Indian and Pacific Oceans. This indicates that there could be slight distinctions in morphological ratios between ocean basins or populations, though most individuals appear to be within the same general range.
There were, however, large differences in rostral tooth counts for sawfish captured in the Ashburton River region compared to those in other parts of the world. The lowest tooth counts, globally, were found in the present study, with the tooth count range determined here, for example, barely overlapping tooth count ranges from the Middle East (Fig. 6). There were also distinct differences in tooth counts between green sawfish within Australia, where up to 31 teeth per side have been found previously for assemblages in north-eastern Australia (Queensland; Whitty et al. 2014), or up to 32 teeth for sawfish in the ‘West Pacific’ (exact location unspecified but likely north-eastern Australia and Indonesia; Faria et al. 2013), while only a (rarely displayed) maximum of 30 teeth were found here for Western Australia. Geographical delineations in rostral tooth counts have also been identified in other sawfish species, including the narrow sawfish Anoxypristis cuspidata (22–29 teeth per side in Indian Ocean vs 17–30 in the West Pacific; Faria et al. 2013) and smalltooth sawfish Pristis pectinata (22–29 teeth per side in the United States and 20–30 teeth per side in West Atlantic vs 20–27 teeth per side in East Atlantic; Wiley et al. 2008; Faria et al. 2013); however, for both these species, there is major overlap in tooth count ranges between locations.
Differences in morphological characteristics between populations are often (though not always) indicative of genetic separation (Barlow 1961), as has been supported in the case of green sawfish by genetic studies finding population structure between the West Pacific and Indian Ocean (Faria et al. 2013) as well as between western and eastern Australia (Phillips et al. 2011). Distinct and non-overlapping ranges in morphological characteristics such as those observed in green sawfish tooth counts could suggest potential past population bottlenecks or founder effects, where resulting populations are formed from genetic material originating from a small pool of individuals (e.g. Spurgin et al. 2014). Considering the rapid and major population declines of sawfishes globally, the former may be more likely. Such a process is also supported by the low genetic diversity for this species found at least within parts of Australia (Phillips et al. 2011), and likely within other remnant populations throughout its range.
The sexual dimorphism in tooth counts for green sawfish found here, albeit minor, is mirrored in several other sawfishes. Similar to the present study, males of both largetooth sawfish Pristis pristis (Thorson 1976; Ishihara et al. 1991; Thorburn et al. 2007; Whitty et al. 2014) and smalltooth sawfish (Simpfendorfer et al. 2008; Wiley et al. 2008) exhibit higher numbers of rostral teeth than females. Conversely, male narrow sawfish potentially average less rostral teeth than females (Compagno and Last 1999), though other studies have found no difference between sexes for this species (Whitty et al. 2014). No sexual dimorphism has been noted for dwarf sawfish Pristis clavata (Thorburn et al. 2008; Whitty et al. 2014). There is no clear evolutionary advantage to having higher rostral tooth counts in males or females (Simpfendorfer et al. 2008). Thus, the consistent sexual dimorphism in rostral tooth counts found in several sawfish species is not necessarily adaptive, and instead could potentially arise from genetic linkages to other sexually selected traits in this group.
Conclusions and considerations for conservation
The life history information presented here for what is one of the least studied pristids will help to clarify several points about the basic biology and ecology of the highly imperilled green sawfish. Greater accuracy in the estimation of birth and maturation sizes of this species will help to inform our knowledge of population dynamics and occurrence of juveniles vs mature individuals across their current distribution. Furthermore, in combination with the updated growth rates provided here for juvenile green sawfish, this information will help to better inform our estimates of generation length for this species in Western Australia, one of the last global strongholds for this species. Such life-history information is highly relevant for fisheries management and assessing the vulnerability and recovery potential of this species.
The finding of varying growth rates based on fine-scale location differences within this study also raises several important considerations regarding conservation and threat assessments for this species. First, the higher growth rates of sawfish in the Ashburton River mouth (likely due, at least in part, to terrestrial nutrient input and resulting high productivity in this system) emphasizes the importance of preserving river mouths as highly productive habitats for estuarine and marine fishes. Further research determining relative productivity and prey availability in the Ashburton River mouth compared to nearby mangrove creeks would be highly beneficial for assessing habitat value for a range of species. Second, the potential for human activities and developments to affect growth and behaviour of green sawfish, whether through noise or light pollution, the formation of barriers to dispersal, or direct disruption of behaviour within the habitat (e.g., boating activity or fishing), emphasizes the importance of considering long-term, sublethal effects of such developments and activities. This is particularly relevant as the potential consequences of human developments or disturbance are most often assessed based on the occurrence of lethal injury to a species or destruction of core habitats; more subtle long-term effects, such as slowing of growth rates, are not often considered as it is challenging to assess such parameters on the fast-track scale of many developments. In the present case, however, it is difficult to determine with certainty whether anthropogenic activities are affecting growth rates of green sawfish in the Pilbara region, as there is no direct comparison of growth in this population before and after the noted developments. More work is needed to determine how light, noise, development structures, and other human interactions affect this species on a long-term basis, in order to ensure that this globally important green sawfish nursery remains robust and functional.
Data availability
The datasets generated during the current study are not publicly available due to funder guidelines but are available from the corresponding author on reasonable request.
References
Barlow GW (1961) Causes and significance of morphological variation in fishes. Syst Zool 10:105–117. https://doi.org/10.2307/2411595
Becker A, Whitfield AK, Cowley PD, Järnegren J, Næsje TF (2013) Potential effects of artificial light associated with anthropogenic infrastructure on the abundance and foraging behaviour of estuary-associated fishes. J Appl Ecol 50:43–50. https://doi.org/10.1111/1365-2664.12024
Broadley A, Stewart-Koster B, Burford MA, Brown CJ (2022) A global review of the critical link between river flows and productivity in marine fisheries. Rev Fish Biol Fish. https://doi.org/10.1007/s11160-022-09711-0
Campana SE (2001) Accuracy, precision and quality control in age determination, including a review of the use and abuse of age validation methods. J Fish Biol 59:197–242. https://doi.org/10.1006/jfbi.2001.1668
Carlson JK, Sulikowski JR, Baremore IE (2006) Do differences in life history exist for blacktip sharks, Carcharhinus limbatus, from the United States South Atlantic Bight and Eastern Gulf of Mexico? Environ Biol Fishes 77:279–292. https://doi.org/10.1007/s10641-006-9129-x
Chittaro P, Grandin C, Pacunski R, Zabel R (2022) Five decades of change in somatic growth of Pacific hake from Puget Sound and Strait of Georgia. PeerJ 10:13577. https://doi.org/10.7717/peerj.13577
Compagno LJV, Last PR (1999) Batoid fishes, chimeras and bony fishes part 1 (Elopidae to Linophrynidae). Food and Agricultural Organization of the United Nations, Rome, Italy
Cortés E (2016) Perspectives on the intrinsic rate of population growth. Methods Ecol Evol 7:1136–1145. https://doi.org/10.1111/2041-210X.12592
Curtis JMR, Vincent ACJ (2006) Life history of an unusual marine fish: survival, growth and movement patterns of Hippocampus guttulatus Cuvier 1829. J Fish Biol 68:707–733. https://doi.org/10.1111/j.0022-1112.2006.00952.x
Davies TW, Duffy JP, Bennie J, Gaston KJ (2014) The nature, extent, and ecological implications of marine light pollution. Front Ecol Environ 12:347–355. https://doi.org/10.1890/130281
Davies CL, Tothill T, Meeuwig JJ, Kyne PM (2022) Garig Gunak Barlu National Park Green Sawfish (Pristis zijsron) aggregation surveys. Charles Darwin University, Darwin, NT, AUS
Dulvy NK, Ellis JR, Goodwin NB, Grant A, Reynolds JD, Jennings S (2004) Methods of assessing extinction risk in marine fishes. Fish Fish 5:255–276. https://doi.org/10.1111/j.1467-2679.2004.00158.x
Dureuil M, Aeberhard WH, Dowd M, Pardo SA, Whoriskey FG, Worm B (2022) Reliable growth estimation from mark–recapture tagging data in elasmobranchs. Fish Res 256:106488. https://doi.org/10.1016/j.fishres.2022.106488
Dureuil M, Worm B (2015) Estimating growth from tagging data: an application to north-east Atlantic tope shark Galeorhinus galeus. J Fish Biol 87:1389–1410. https://doi.org/10.1111/jfb.12830
Elhassan IS (2018) Occurrence of the green sawfish Pristis zijsron in the Sudanese Red Sea with observations on reproduction. Endanger Species Res 36:41–47. https://doi.org/10.3354/esr00873
Fabens AJ (1965) Properties and fitting of the von Bertalanffy growth curve. Growth 29:265–289
Faria VV, McDavitt MT, Charvet P, Wiley TR, Simpfendorfer CA, Naylor GJ (2013) Species delineation and global population structure of Critically Endangered sawfishes (Pristidae). Zool J Linn Soc 167:136–164. https://doi.org/10.1111/j.1096-3642.2012.00872.x
Faria VV (2007) Taxonomic review, phylogeny, and geographical population structure of the sawfishes (Chondrichthyes, Pristiformes). Ecology and Evoluntionary Biology, Ames, Iowa, USA
Gillanders BM, Kingsford MJ (2002) Impact of changes in flow of freshwater on estuarine and open coastal habitats and the associated organisms. In: Gibson RN, Barnes M, Atkinson RJA (eds) Oceanography and Marine Biology an Annual Review. Taylor & Francis, London, p 233
Gillanders BM, Izzo C, Doubleday ZA, Ye Q (2015) Partial migration: growth varies between resident and migratory fish. Biol Lett 11:20140850. https://doi.org/10.1098/rsbl.2014.0850
Gillooly JF, Brown JH, West GB, Savage VM, Charnov EL (2001) Effects of size and temperature on metabolic rate. Science 293:2248–2251. https://doi.org/10.1126/science.1061967
Gillooly JF, Charnov EL, West GB, Savage VM, Brown JH (2002) Effects of size and temperature on developmental time. Nature 417:70–73. https://doi.org/10.1038/417070a
Goldman KJ, Cailliet GM, Andrews AH, Natanson LJ (2012) Assessing the age and growth of chondrichthyan fishes. In: Carrier JC, Musick JA, Heithaus MR (eds) Biology of Sharks and their Relatives. CRC Press, Boca Raton, pp 423–451
Hamel MJ, Koch JD, Steffensen KD, Pegg MA, Hammen JJ, Rugg ML (2014) Using mark–recapture information to validate and assess age and growth of long-lived fish species. Can J Fish Aquat Sci 71:559–566. https://doi.org/10.1139/cjfas-2013-039
Harry AV, Everett B, Faria V, Fordham S, Grant MI, Haque AB, Ho H, Jabado RW, Jones GCA, Lear KO, Morgan DL, Phillips NM, Spaet JLY, Tanna A, Wueringer BE (2022) Pristis zijsron Bleeker 1851. The IUCN Red List of Threatened Species. e.T39393A58304631. https://doi.org/10.2305/IUCN.UK.2022-2.RLTS.T39393A58304631.en
Hewett SW, Kraft CE (1993) The relationship between growth and consumption: comparisons across fish populations. Trans Am Fish Soc 122:814–821. https://doi.org/10.1577/1548-8659(1993)122%3c0814:TRBGAC%3e2.3.CO;2
Hobday A, Smith A, Stobutzki I, Bulman C, Daley R, Dambacher J, Deng R, Dowdney J, Fuller M, Furlani D (2011) Ecological risk assessment for the effects of fishing. Fish Res 108:372–384. https://doi.org/10.1016/j.fishres.2011.01.013
Ishihara H, Taniuchi T, Shimizu M (1991) Sexual dimorphism in number of rostral teeth in the sawfish, Pristis microdon collected from Australia and Papua New Guinea. Univ Mus Univ Tokyo Nat Cult 3:83–89
Jabado RW, Al Baharna RA, Al Ali SR, Al Suwaidi KO, Al Blooshi AY, Al Dhaheri SS (2017) Is this the last stand of the critically endangered green sawfish Pristis zijsron in the Arabian Gulf? Endan Spec Res 32:265–275. https://doi.org/10.3354/esr00805
Jobling M (1997) Temperature and growth: modulation of growth rate via temperature. Glob Warm 61:225–253. https://doi.org/10.1017/CBO9780511983375
Lahti K, Laurila A, Enberg K, Piironen J (2001) Variation in aggressive behaviour and growth rate between populations and migratory forms in the brown trout, Salmo trutta. Anim Behav 62:935–944. https://doi.org/10.1006/anbe.2001.1821
Last PR, Stevens JD (2009) Sharks and rays of Australia. CSIRO Publishing, Collingwood, VIC, Australia
Last P, Naylor G, Séret B, White W, de Carvalho M, Stehmann M (2016) Rays of the World. CSIRO publishing, Clayton South, VIC, Australia
Leeney RH (2017) Are sawfishes still present in Mozambique? A basal ecological study. Peerj 5:e2950. https://doi.org/10.7717/peerj.2950
Maceina MJ, Boxrucker J, Buckmeier DL, Gangl RS, Lucchesi DO, Isermann DA, Jackson JR, Martinez PJ (2007) Current status and review of freshwater fish aging procedures used by state and provincial fisheries agencies with recommendations for future directions. Fisheries 32:329–340. https://doi.org/10.1577/1548-8446(2007)32[329:CSAROF]2.0.CO;2
Marchand KA, Hughes GN, Litzgus JD (2018) Geographic variation in somatic growth rate of wood turtles (Glyptemys insculpta). Copeia 106:477–484. https://doi.org/10.1643/CH-18-022
Maunder MN, Punt AE (2013) A review of integrated analysis in fisheries stock assessment. Fish Res 142:61–74. https://doi.org/10.1016/j.fishres.2012.07.025
Meyer CG, O’Malley JM, Papastamatiou YP, Dale JJ, Hutchinson MR, Anderson JM, Royer MA, Holland KN (2014) Growth and maximum size of tiger sharks (Galeocerdo cuvier) in Hawaii. PLoS ONE 9:84799. https://doi.org/10.1371/journal.pone.0084799
Molony BW, Newman SJ, Joll L, Lenanton RCJ, Wise B (2011) Are Western Australian waters the least productive waters for finfish across two oceans? A review with a focus on finfish resources in the Kimberley region and North Coast Bioregion. J R Soc West Aust 94:323
Morgan DL, Whitty JM, Phillips NM, Thorburn DC, Chaplin JA, McAuley R (2011) North-western Australia as a hotspot for endangered elasmobranchs with particular reference to sawfishes and the Northern River Shark. J R Soc West Aust 94:345–358
Morgan DL, Allen MG, Ebner BC, Whitty JM, Beatty SJ (2015) Discovery of a pupping site and nursery for critically endangered green sawfish Pristis zijsron. J Fish Biol 86:1658–1663. https://doi.org/10.1111/jfb.12668
Morgan DL, Ebner BC, Allen MG, Gleiss AC, Beatty SJ, Whitty JM (2017) Habitat use and site fidelity of neonate and juvenile green sawfish Pristis zijsron in a nursery area in Western Australia. Endanr Spec Res 34:235–249. https://doi.org/10.3354/esr00847
Peverell SC (2005) Distribution of sawfishes (Pristidae) in the Queensland Gulf of Carpentaria, Australia, with notes on sawfish ecology. Environ Biol Fishes 73:391–402. https://doi.org/10.1007/s10641-005-1599-8
Peverell SC (2009) Sawfish (Pristidae) of the Gulf of Carpentaria. Queensland, Australia
Phillips NM, Chaplin JA, Morgan DL, Peverell SC (2011) Population genetic structure and genetic diversity of three critically endangered Pristis sawfishes in Australian waters. Mar Biol 158:903–915. https://doi.org/10.1007/s00227-010-1617-z
Scherrer SR, Kobayashi DR, Weng KC, Okamoto HY, Oishi FG, Franklin EC (2021) Estimation of growth parameters integrating tag-recapture, length-frequency, and direct aging data using likelihood and Bayesian methods for the tropical deepwater snapper Pristipomoides filamentosus in Hawaii. Fish Res 233:105753. https://doi.org/10.1016/j.fishres.2020.105753
Simpfendorfer CA (1993) Age and growth of the Australian sharpnose shark, Rhizoprionodon taylori, from north Queensland, Australia. Environ Biol Fishes 36:233–241. https://doi.org/10.1007/BF00001718
Simpfendorfer CA (2000) Growth rates of juvenile dusky sharks, Carcharhinus obscurus (Lesueur, 1818), from southwestern Australia estimated from tag-recapture data. Fish Bull 98:811–811
Simpfendorfer CA, Chidlow J, McAuley R, Unsworth P (2000) Age and growth of the whiskery shark, Furgaleus macki, from southwestern Australia. Environ Biol Fishes 58:335–343. https://doi.org/10.1023/A:1007624828001
Simpfendorfer CA, Poulakis GR, O’Donnell PM, Wiley TR (2008) Growth rates of juvenile smalltooth sawfish Pristis pectinata Latham in the western Atlantic. J Fish Biol 72:711–723. https://doi.org/10.1111/j.1095-8649.2007.01764.x
Simpfendorfer C (2013) Pristis zijsron. The IUCN Red List of Threatened Species. e.T39393A18620401
Smart JJ, Grammer GL (2021) Modernising fish and shark growth curves with Bayesian length-at-age models. PLoS ONE 16:0246734. https://doi.org/10.1371/journal.pone.0246734
Spurgin LG, Illera JC, Jorgensen TH, Dawson DA, Richardson DS (2014) Genetic and phenotypic divergence in an island bird: isolation by distance, by colonization or by adaptation? Mol Ecol 23:1028–1039. https://doi.org/10.1111/mec.12672
Stevens JD, McAuley RB, Simpfendorfer CA, Pillans RD (2008) Spatial distribution and habitat utilisation of sawfish (Pristis spp) in relation to fishing in. A report to Department of the Environment Water, Heritage and the Arts, northern Australia
Thorburn DC, Morgan DL, Rowland AJ, Gill HS (2007) Freshwater sawfish Pristis microdon Latham, 1794 (Chondrichthyes: Pristidae) in the Kimberley region of Western Australia. Zootaxa 1471:27–41. https://doi.org/10.11646/zootaxa.1471.1.3
Thorburn DC, Morgan DL, Rowland AJ, Gill HS, Paling E (2008) Life history notes of the critically endangered dwarf sawfish, Pristis clavata, Garman 1906 from the Kimberley region of Western Australia. Environ Biol Fishes 83:139–145. https://doi.org/10.1007/s10641-007-9306-6
Thorson TB (1976) Observations on the reproduction of the sawfish, Pristis perotteti, in lake Nicaragua, with recommendations for its conservation. In: Thorson TB (ed) Investigations of the ichthyofauna of Nicaraguan lakes. University of Nebraska-Lincoln
Uthe P, Al-Chokhachy R, Zale AV, Shepard BB, McMahon TE, Stephens T (2016) Life history characteristics and vital rates of Yellowstone cutthroat trout in two headwater basins. N Am J Fish Manage 36:1240–1253. https://doi.org/10.1080/02755947.2016.1206643
Voellmy IK, Purser J, Flynn D, Kennedy P, Simpson SD, Radford AN (2014) Acoustic noise reduces foraging success in two sympatric fish species via different mechanisms. Anim Behav 89:191–198. https://doi.org/10.1016/j.anbehav.2013.12.029
Weatherley AH (1990) Approaches to understanding fish growth. Trans Am Fish Soc 119:662–672. https://doi.org/10.1577/1548-8659(1990)119%3c0662:ATUFG%3e2.3.CO;2
Whitty JM, Phillips NM, Thorburn DC, Simpfendorfer CA, Field IC, Peverell SC, Morgan DL (2014) Utility of rostra in the identification of Australian sawfishes (Chondrichthyes: Pristidae). Aquat Conserv: Mar Freshwat Ecosyst 24:791–804. https://doi.org/10.1002/aqc.2398
Wiley TR, Simpfendorfer CA, Faria VV, McDavitt MT (2008) Range, sexual dimorphism and bilateral asymmetry of rostral tooth counts in the smalltooth sawfish Pristis pectinata Latham (Chondrichthyes: Pristidae) of the southeastern United States. Zootaxa 1810:51–59
Williams AJ, Farley JH, Hoyle SD, Davies CR, Nicol SJ (2012) Spatial and sex-specific variation in growth of albacore tuna (Thunnus alalunga) across the South Pacific Ocean. PLoS ONE 7:e39318. https://doi.org/10.1371/journal.pone.0039318
Wueringer BE (2017) Sawfish captures in the Queensland shark control program, 1962 to 2016. Endanger Species Res 34:293–300. https://doi.org/10.3354/esr00853
Wueringer BE, Biskis VN, Pinkus GA (2023) Impacts of trophy collection and commercial fisheries on sawfishes in Queensland. Endanger Species Res 50:33–150. https://doi.org/10.3354/esr01222
Acknowledgements
This study was funded by Chevron Australia, and we would like to especially thank P. de Lestang and S. Moore for their major assistance in fieldwork management and logistical organisation. We would also like to thank the Thalanyji Sea Rangers for assistance with fieldwork on country, and M. Allen, B. Ebner, J. Whitty, S. Beatty, J. Keleher, M. Fraser, G. Herbert, C. Estrabeau, and A. Slater for assistance in the field. Data from incidental captures from elsewhere in Australia included in this study were collected with funding and assistance from Murdoch University, O2 Marine, Cape Conservation Group, and AECOM. We thank the reviewers for their constructive feedback and improvement of the manuscript.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This study was funded by Chevron Australia. Data from incidental captures from elsewhere in Australia included in this study were collected with funding and assistance from Murdoch University, O2 Marine, Cape Conservation Group, and AECOM.
Author information
Authors and Affiliations
Contributions
Karissa Lear and David Morgan conceived the study design and managed fieldwork and data collection. Data were collected by all authors, and analyses were performed by Karissa Lear. All authors contributed to the writing of the manuscript and have approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or non-financial competing interests to disclose.
Ethical approval
All work with animals was conducted under Murdoch University Animal Ethics permit #RW3191-19, and Western Australia Department of Fisheries permits #3378 and #250922121.
Additional information
Responsible Editor: J. Carlson .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lear, K.O., Fazeldean, T., Bateman, R.L. et al. Growth and morphology of Critically Endangered green sawfish Pristis zijsron in globally important nursery habitats. Mar Biol 170, 70 (2023). https://doi.org/10.1007/s00227-023-04220-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-023-04220-5