Abstract
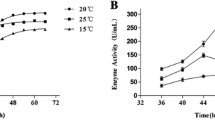
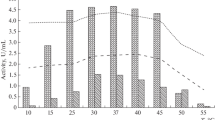
A psychrotrophic bacterial strain, Pseudoaltermonas sp. SM9913, was isolated from deep-sea sediment collected at 1,855 m depth. Two proteases produced by Pseudoaltermonas sp. SM9913 were purified, MPC-01 and MCP-02. MCP-01 is a serine protease with a molecular weight of 60.7 kDa. It is cold-adapted with an optimum temperature of 30–35°C. Its K m and E a for the hydrolysis of casein were 0.18% and 39.1 kJ mol−1, respectively. It had low thermostability, and its activity was reduced by 73% after incubation at 40°C for 10 min. MCP-02 is a mesophilic metalloprotease with a molecular weight of 36 kDa. Its optimum temperature for the hydrolysis of casein was 50–55°C. The K m and E a of MCP-02 for the hydrolysis of casein were 0.36% and 59.3 kJ mol−1, respectively. MCP-02 had high thermostability, and its activity was reduced by only 30.5% after incubation at 60°C for 10 min. At low temperatures, Pseudoaltermonas sp. SM9913 mainly produced the psychrophilic protease MCP-01.







Similar content being viewed by others
References
Chen XL, Zhang YZ, Wang YT, Gao PJ, Luan XW (2001) Psychrotrophilic protease from a deep sea psychrotrophilic strain Pseudoaltermonas sp. SM9913. Mar Sci (China) 25:4–8
Feller G, Gerday C (1997) Psychrophilic enzymes: molecular basis of cold adaptation. Cell Mol Life Sci 53:830–841
Gerday C, Aittaleb M, Bentahir M, Chessa JP, Claverie P, et al (2000) Cold-adapted enzymes: from fundamentals to biotechnology Tibtech March 18:103–107
Hamamoto T, Horikoshi K (1991) Characterisation of amylase from a psychrotrophic Vibrio isolated from a deep-sea mud sample. FEMS Microbiol Lett 84:79–84
Hamamoto T, Horikoshi K (1993) Deep-sea microbiology research within the Deepstar program. J Mar Biotechnol 1:119–124
Hoshino T, Ishizaki K, Sakamoto T, et al (1997) Isolation of a Pseudomonas species from fish intestine that produces a protease active at low temperature. Lett Appl Microbiol 25:70–72
Huston AL, Krieger-Brockett BB, Deming JW (2000) Remarkably low temperature optima for extracellular enzyme activity from Arctic bacteria and sea ice. Environ Microbiol 2:383–388
Kato C, Suzuki S, Hata S, Ito T, Horikoshi K (1995) The properties of a protease activated by high pressure from Sporosarcina sp. strain DSK25 isolated from deep-sea sediment. JAMSTEC J Deep Sea Res 32:7–13
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol regent. J Biol Chem 193:265–275
Margesin R, Schinner F (1991) Characterization of a metalloprotease from psychrophilic Xanthomonas maltophilia. FEMS Microbiol Lett. 79:257–262
Margesin R, Schinner F (1992) A comparison of extracellular proteases from three psychrotrophic strains of Pseudomonas fluorescens. J Gen Appl Microbiol 38:209–225
Margesin R, Palma N, Knauseder F, Schinner F (1991) Proteases of psychrotrophic bacteria isolated from glaciers. J Basic Microbiol 31:377–383
Morita RY (1975) Psychrophilic bacteria. Bacteriol Rev 39:144–167
Outzen H, Berglund GI, Smalas AO, Willassen NP (1994) Temperature and pH sensitivity of trypsins from Atlantic salmon (Salmo salar) in comparison with bovine and porcine trypsins. Comp Biochem Physiol B 115:33–45
Acknowledgements
The work was supported by the Hi-Tech Research and Development Program of China (No. 2002AA628100) and the National Natural Science Foundation of China (No. 40276047).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by O. Kinne, Oldendorf/Luhe
Rights and permissions
About this article
Cite this article
Chen, XL., Zhang, YZ., Gao, PJ. et al. Two different proteases produced by a deep-sea psychrotrophic bacterial strain, Pseudoaltermonas sp. SM9913. Marine Biology 143, 989–993 (2003). https://doi.org/10.1007/s00227-003-1128-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-003-1128-2