Abstract
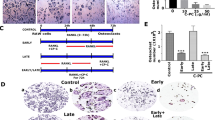
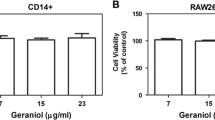
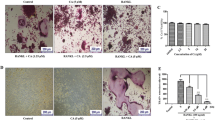
Dried plums and their polyphenols have been shown to suppress bone resorption by downregulating receptor activator NF-κB ligand (RANKL). Due to the anti-inflammatory and antioxidant properties of these compounds, this study was designed to investigate whether dried plum polyphenols exert additional, more direct effects on osteoclasts and their precursors. RAW 264.7 macrophages were used as a model to study osteoclast precursors and osteoclast differentiation and activity. Under inflammatory conditions induced by lipopolysaccharide (LPS), polyphenols extracted from dried plum (10, 20, and 30 μg/mL) downregulated osteoclast precursor cyclooxygenase expression and nitric oxide (NO) by inhibiting inducible NO synthase. NO and tumor necrosis factor (TNF)-α were also suppressed in the presence of RANKL during osteoclastogenesis by the polyphenols. Increased TNF-α production in response to oxidative stress, but not LPS, was decreased over time. As expected, LPS and H2O2 significantly increased the number of tartrate-resistant acid phosphatase-positive cells by 127% and 30%, respectively. Dried plum polyphenols decreased osteoclast differentiation under normal as well as inflammatory and oxidative stress conditions, coincident with the suppression of the transcription factor, nuclear factor for activated T cells (NFATc1). These inhibitory effects on osteoclastogenesis were confirmed in primary bone marrow cultures. Resorption pit formation was decreased to a similar extent as osteoclast differentiation, suggesting that dried plum polyphenols primarily affect osteoclast differentiation as opposed to activity. Our data demonstrate that dried plum polyphenols directly inhibit osteoclastogenesis, leading to a decrease in osteoclast activity, by downregulating NFATc1 and inflammatory mediators.







Similar content being viewed by others
References
New SA, Robins SP, Campbell MK, Martin JC, Garton MJ, Bolton-Smith C, Grubb DA, Lee SJ, Reid DM (2000) Dietary influences on bone mass and bone metabolism: further evidence of a positive link between fruit and vegetable consumption and bone health? Am J Clin Nutr 71:142–151
Muhlbauer RC, Lozano A, Reinli A, Wetli H (2003) Various selected vegetables, fruits, mushrooms and red wine residue inhibit bone resorption in rats. J Nutr 133:3592–3597
Park EK, Kim MS, Lee SH, Kim KH, Park JY, Kim TH, Lee IS, Woo JT, Jung JC, Shin HI, Choi JY, Kim SY (2004) Furosin, an ellagitannin, suppresses RANKL-induced osteoclast differentiation and function through inhibition of MAP kinase activation and actin ring formation. Biochem Biophys Res Commun 325:1472–1480
Yun JH, Pang EK, Kim CS, Yoo YJ, Cho KS, Chai JK, Kim CK, Choi SH (2004) Inhibitory effects of green tea polyphenol (-)-epigallocatechin gallate on the expression of matrix metalloproteinase-9 and on the formation of osteoclasts. J Periodontal Res 39:300–307
Wattel A, Kamel S, Mentaverri R, Lorget F, Prouillet C, Petit JP, Fardelonne P, Brazier M (2003) Potent inhibitory effect of naturally occurring flavonoids quercetin and kaempferol on in vitro osteoclastic bone resorption. Biochem Pharmacol 65:35–42
Nakatani N, Kayano S, Kikuzaki H, Sumino K, Katagiri K, Mitani T (2000) Identification, quantitative determination, and antioxidative activities of chlorogenic acid isomers in prune (Prunus domestica L.). J Agric Food Chem 48:5512–5516
Kayano S, Kikuzaki H, Ikami T, Suzuki T, Mitani T, Nakatani N (2004) A new bipyrrole and some phenolic constituents in prunes (Prunus domestica L.) and their oxygen radical absorbance capacity (ORAC). Biosci Biotechnol Biochem 68:942–944
Franklin M, Bu SY, Lerner MR, Lancaster EA, Bellmer D, Marlow D, Lightfoot SA, Arjmandi BH, Brackett DJ, Lucas EA, Smith BJ (2006) Dried plum prevents bone loss in a male osteoporosis model via IGF-I and the RANK pathway. Bone 39:1331–1342
Arjmandi BH, Lucas EA, Juma S, Soliman A, Stoecker BJ, Khalil DA, Smith BJ, Wang C (2001) Dried plums prevent ovariectomy-induced bone loss in rats. J Am Nutraceut Assoc 4:50–56
Bu SY, Lucas EA, Franklin M, Marlow D, Brackett DJ, Boldrin EA, Devareddy L, Arjmandi BH, Smith BJ (2007) Comparison of dried plum supplementation and intermittent PTH in restoring bone in osteopenic orchidectomized rats. Osteoporos Int 18:931–942
Bu SY, Hunt TS, Smith BJ (2008) Dried plum polyphenols attenuate the detrimental effects of TNF-alpha on osteoblast function coincident with upregulation of Runx2, Osterix and IGF-I. J Nutr Biochem (in press)
Teitelbaum SL (2000) Bone resorption by osteoclasts. Science 289:1504–1508
Hsu H, Lacey DL, Dunstan CR, Solovyev I, Colombero A, Timms E, Tan HL, Elliott G, Kelley MJ, Sarosi I, Wang L, Xia XZ, Elliott R, Chiu L, Black T, Scully S, Capparelli C, Morony S, Shimamoto G, Bass MB, Boyle WJ (1999) Tumor necrosis factor receptor family member RANK mediates osteoclast differentiation and activation induced by osteoprotegerin ligand. Proc Natl Acad Sci USA 96:3540–3545
Jagger CJ, Lean JM, Davies JT, Chambers TJ (2005) Tumor necrosis factor-alpha mediates osteopenia caused by depletion of antioxidants. Endocrinology 146:113–118
Sakuma Y, Tanaka K, Suda M, Yasoda A, Natsui K, Tanaka I, Ushikubi F, Narumiya S, Segi E, Sugimoto Y, Ichikawa A, Nakao K (2000) Crucial involvement of the EP4 subtype of prostaglandin E receptor in osteoclast formation by proinflammatory cytokines and lipopolysaccharide. J Bone Miner Res 14:218–227
Jung JY, Lin AC, Ramos LM, Faddis BT, Chole RA (2003) Nitric oxide synthase I mediates osteoclast activity in vitro and in vivo. J Cell Biochem 89:613–621
Suda K, Udagawa N, Sato N, Takami M, Itoh K, Woo JT, Takahashi N, Nagai K (2004) Suppression of osteoprotegerin expression by prostaglandin E2 is crucially involved in lipopolysaccharide-induced osteoclast formation. J Immunol 172:2504–2510
Lean JM, Davies JT, Fuller K, Jagger CJ, Kirstein B, Partington GA, Urry ZL, Chambers TJ (2003) A crucial role for thiol antioxidants in estrogen-deficiency bone loss. J Clin Invest 112:915–923
Fretz JA, Shevde NK, Singh S, Darnay BG, Pike JW (2008) RANKL-induced nuclear factor of activated t cells (C1) autoregulates its own expression in osteoclasts and mediates the upregulation of tartrate-resistant acid phosphatase. Mol Endocrinol 22:737–750
Wattel A, Kamel S, Prouillet C, Petit JP, Lorget F, Offord E, Brazier M (2004) Flavonoid quercetin decreases osteoclastic differentiation induced by RANKL via a mechanism involving NFkappaB and AP-1. J Cell Biochem 92:285–295
Woo JT, Nakagawa H, Notoya M, Yonezawa T, Udagawa N, Lee IS, Ohnishi M, Hagiwara H, Nagai K (2004) Quercetin suppresses bone resorption by inhibiting the differentiation and activation of osteoclasts. Biol Pharm Bull 27:504–509
Nakagawa H, Wachi M, Woo JT, Kato M, Kasai S, Takahashi F, Lee IS, Nagai K (2002) Fenton reaction is primarily involved is a mechanism of (-)-epigallocatechim-3-gallate to induce osteoclastic cell death. Biochem Biophys Res Commun 292:94–101
Tang QY, Kukita T, Ushijima Y, Kukita A, Nagata K, Sandra F, Watanabe T, Toh K, Okuma Y, Kawasaki S, Rasubala L, Teramachi J, Miyamoto I, Wu Z, Iijima T (2006) Regulation of osteoclastogenesis by Simon extracts composed of caffeic acid and related compounds: successful suppression of bone destruction accompanied with adjuvant-induced arthritis in rats. Histochem Cell Biol 125:215–225
Kroon PA, Clifford MN, Crozier A, Day AJ, Donovan JL, Manach C, Williamson G (2004) How should we assess the effects of exposure to dietary polyphenols in vitro? Am J Clin Nutr 80:15–21
Chen Y, Shen S, Lee W, Hou W, Yang L, Lee TJF (2001) Inhibition of nitric oxide synthase inhibitors and lipopolysaccharide induced inducible NOS and cyclooxygenase-2 gene expression by rutin, quercetin, and quercetin pentaacetate in RAW 264.7 macrophages. J Cell Biochem 82:537–548
Rassi CM, Lieberherr M, Chaumaz G, Pointillart A, Cournot G (2005) Modulation of osteoclastogenesis in porcine bone marrow cultures by quercetin and rutin. Cell Tissue Res 319:383–393
Yun JH, Kim CS, Cho KS, Chai JK, Kim CK, Choi SH (2007) (-)-Epigallocatechin gallate induces apoptosis, via caspase activation, in osteoclasts differentiated from RAW 264.7 cells. J Periodontal Res 42:212–218
Horcajada-Molteni MN, Crespy V, Coxam V, Davicco MJ, Remesy C, Barlet JP (2000) Rutin inhibits ovariectomy-induced osteopenia in rats. J Bone Miner Res 15:2251–2258
Kim DO, Chun OK, Kim YJ, Moon HY, Lee CY (2003) Quantification of polyphenolics and their antioxidant capacity in fresh plums. J Agric Food Chem 51:6509–6515
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-Ciocalteu reagent. Oxidants Antioxidants 299:152–178
Cui W, Cuartas E, Ke J, Zhang Q, Einarsson HB, Sedgwick JD, Li J, Vignery A (2007) CD200 and its receptor, CD200R, modulate bone mass via the differentiation of osteoclasts. Proc Natl Acad Sci USA 104:14436–14441
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 126:131–138
Takami M, Kim N, Rho J, Choi Y (2002) Stimulation by toll-like receptors inhibits osteoclast differentiation. J Immunol 169:1516–1523
Yang CS, Landau JM (2000) Effects of tea consumption on nutrition and health. J Nutr 130:2409–2412
McGartland CP, Robson PJ, Murray LJ, Cran GW, Savage MJ, Watkins DC, Rooney MM, Boreham CA (2004) Fruit and vegetable consumption and bone mineral density: the Northern Ireland Young Hearts Project. Am J Clin Nutr 80:1019–1023
Santangelo C, Vari R, Scazzocchio B, Di BR, Filesi C, Masella R (2007) Polyphenols, intracellular signalling and inflammation. Ann Ist Super Sanita 43:394–405
Ben DD, Reznick AZ, Srouji S, Livne E (2008) Exposure to pro-inflammatory cytokines upregulates MMP-9 synthesis by mesenchymal stem cell-derived osteoprogenitors. Histochem Cell Biol 129:589–597
Kayano S, Kikuzaki H, Fukutsuka N, Mitani T, Nakatani N (2002) Antioxidant activity of prune (Prunus domestica L.) constituents and a new synergist. J Agric Food Chem 50:3708–3712
Nakatani N, Kayano SI, Kikuzaki H, Sumino K, Katagiri K, Mitani T (2000) Identification, quantitative determination, and antioxidative activities of chlorogneic acid isomers in prune (Prunus domestica L.). J Agric Food Chem 48:5512–5516
Pacifici R (1996) Estrogen, cytokines, and pathogenesis of postmenopausal osteoporosis. J Bone Miner Res 11:1043–1051
Cuzzocrea S, Mazzon E, Dugo L, Genovese T, Di PR, Ruggeri Z, Vegeto E, Caputi AP, Van De Loo FA, Puzzolo D, Maggi A (2003) Inducible nitric oxide synthase mediates bone loss in ovariectomized mice. Endocrinology 144:1098–1107
Alayan J, Ivanovski S, Gemmell E, Ford P, Hamlet S, Farah CS (2006) Deficiency of iNOS contributes to Porphyromonas gingivalis-induced tissue damage. Oral Microbiol Immunol 21:360–365
van’t Hof RJ, Armour KJ, Smith LM, Armour KE, Wei XQ, Liew FY, Ralston SH (2000) Requirement of the inducible nitric oxide synthase pathway for IL-1-induced osteoclastic bone resorption. Proc Natl Acad Sci USA 97:7993–7998
Lee SK, Huang H, Lee SW, Kim KH, Kim KK, Kim HM, Lee ZH, Kim HH (2004) Involvement of iNOS-dependent NO production in the stimulation of osteoclast survival by TNF-alpha. Exp Cell Res 298:359–368
Kim SF, Huri DA, Snyder SH (2005) Inducible nitric oxide synthase binds, S-nitrosylates, and activates cyclooxygenase-2. Science 310:1966–1970
Salvemini D, Misko TP, Masferrer JL, Seibert K, Currie MG, Needleman P (1993) Nitric oxide activates cyclooxygenase enzymes. Proc Natl Acad Sci USA 90:7240–7244
Pacifici R (2007) Estrogen deficiency, T cells and bone loss. Cell Immunol. September 19 [Epub ahead of print]
O’Gradaigh D, Ireland D, Bord S, Compston JE (2004) Joint erosion in rheumatoid arthritis: interactions between tumour necrosis factor alpha, interleukin 1, and receptor activator of nuclear factor kappaB ligand (RANKL) regulate osteoclasts. Ann Rheum Dis 63:354–359
Ritchlin CT, Haas-Smith SA, Li P, Hicks DG, Schwarz EM (2003) Mechanisms of TNF-alpha- and RANKL-mediated osteoclastogenesis and bone resorption in psoriatic arthritis. J Clin Invest 111:821–831
Iqbal J, Sun L, Kumar TR, Blair HC, Zaidi M (2006) Follicle-stimulating hormone stimulates TNF production from immune cells to enhance osteoblast and osteoclast formation. Proc Natl Acad Sci USA 103:14925–14930
Rahman I, Marwick J, Kirkham P (2004) Redox modulation of chromatin remodeling: impact on histone acetylation and deacetylation, NF-kappaB and pro-inflammatory gene expression. Biochem Pharmacol 68:1255–1267
Staniforth V, Chiu LT, Yang NS (2006) Caffeic acid suppresses UVB radiation-induced expression of interleukin-10 and activation of mitogen-activated protein kinases in mouse. Carcinogenesis 27:1803–1811
Lean JM, Jagger CJ, Kirstein B, Fuller K, Chambers TJ (2005) Hydrogen peroxide is essential for estrogen-deficiency bone loss and osteoclast formation. Endocrinology 146:728–735
Nathens AB, Bitar R, Marshall JC, Watson RW, Dackiw AP, Fan J, Hiscott J, Rotstein OD (2001) Antioxidants increase lipopolysaccharide-stimulated TNF alpha release in murine macrophages: role for altered TNF alpha mRNA stability. Shock 16:361–367
Bulger EM, Garcia I, Maier RV (1998) Dithiocarbamates enhance tumor necrosis factor-alpha production by rabbit alveolar macrophages, despite inhibition of NF-kappaB. Shock 9:397–405
Cremin P, Kasim-Karakas S, Waterhouse AL (2001) LC/ES-MS detection of hydroxycinnamates in human plasma and urine. J Agric Food Chem 49:1747–1750
Olthof MR, Hollman PC, Katan MB (2001) Chlorogenic acid and caffeic acid are absorbed in humans. J Nutr 131:66–71
Acknowledgements
This material is based upon work supported by Cooperative State Research, Education and Extension Service, U.S. Department of Agriculture, under award 2006-35200-17383. Additionally, this study was supported by the California Dried Plum Board and the Oklahoma Center for the Advancement of Science and Technology (HR06-109).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bu, S.Y., Lerner, M., Stoecker, B.J. et al. Dried Plum Polyphenols Inhibit Osteoclastogenesis by Downregulating NFATc1 and Inflammatory Mediators. Calcif Tissue Int 82, 475–488 (2008). https://doi.org/10.1007/s00223-008-9139-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-008-9139-0