Abstract
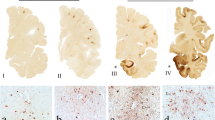
Negative outcomes of mild traumatic brain injury (mTBI) can be exacerbated by repeated insult. Animal models of repeated closed-head mTBI provide the opportunity to define acute pathological mechanisms as the number of mTBI increases. Furthermore, little is known about the effects of mTBI impact site, and how this may affect brain function. We use a closed head, weight drop model of mTBI that allows head movement following impact, in adult female rats to determine the role of the number and location of mTBI on brain pathology and behaviour. Biomechanical assessment of two anatomically well-defined mTBI impact sites were used, anterior (bregma) and posterior (lambda). Location of the impact had no significant effect on impact forces (450 N), and the weight impact locations were on average 5.4 mm from the desired impact site. No between location vertical linear head kinematic differences were observed immediately following impact, however, in the 300 ms post-impact, significantly higher mean vertical head displacement and velocity were observed in the mTBI lambda trials. Breaches of the blood brain barrier were observed with three mTBI over bregma, associated with immunohistochemical indicators of damage. However, an increased incidence of hairline fractures of the skull and macroscopic haemorrhaging made bregma an unsuitable impact location to model repeated mTBI. Repeated mTBI over lambda did not cause skull fractures and were examined more comprehensively, with outcomes following one, two or three mTBI or sham, delivered at 1 day intervals, assessed on days 1–4. We observe a mild behavioural phenotype, with subtle deficits in cognitive function, associated with no identifiable neuroanatomical or inflammatory changes. However, an increase in lipid peroxidation in a subset of cortical neurons following two mTBI indicates increasing oxidative damage with repeated injury in female rats, supported by increased amyloid precursor protein immunoreactivity with three mTBI. This study of acute events following closed head mTBI identifies lipid peroxidation in neurons at the same time as cognitive deficits. Our study adds to existing literature, providing biomechanics data and demonstrating mild cognitive disturbances associated with diffuse injury, predominantly to grey matter, acutely following repeated mTBI.






Similar content being viewed by others
References
Angoa-Pérez M, Kane MJ, Briggs DI, Herrera-Mundo N, Viano DC, Kuhn DM (2014) Animal models of sports-related head injury: bridging the gap between pre-clinical research and clinical reality. 129:916–931. doi:10.1111/jnc.12690
Ansari MA, Roberts KN, Scheff SW (2008a) Oxidative stress and modification of synaptic proteins in hippocampus after traumatic brain injury. Free Radical Biol Med 45:443–452. doi:10.1016/j.freeradbiomed.2008.04.038
Ansari MA, Roberts KN, Scheff SW (2008b) A time course of contusion-induced oxidative stress and synaptic proteins in cortex in a rat model of TBI. J Neurotrauma 25:513–526. doi:10.1089/neu.2007.0451
Bazarian JJ, Wong T, Harris M, Leahey N, Mookerjee S, Dombovy M (1999) Epidemiology and predictors of post-concussive syndrome after minor head injury in an emergency population. Brain Inj 13:173–189
Bennett RE, Mac Donald CL, Brody DL (2012) Diffusion tensor imaging detects axonal injury in a mouse model of repetitive closed-skull traumatic brain injury. Neurosci Lett 513:160–165. doi:10.1016/j.neulet.2012.02.024
Binder LM (1997) A review of mild head trauma. part II: clinical implications. J Clin Exp Neuropsychol 19:432–457. doi:10.1080/01688639708403871
Bolouri H, Saljo A, Viano DC, Hamberger A (2012) Animal model for sport-related concussion; ICP and cognitive function. Acta Neurol Scand 125:241–247. doi:10.1111/j.1600-0404.2011.01614.x
Bolton AN, Saatman KE (2014) Regional neurodegeneration and gliosis are amplified by mild traumatic brain injury repeated at 24-hour intervals. J Neuropathol Exp Neurol 73:933–947. doi:10.1097/NEN.0000000000000115
Broshek DK, Kaushik T, Freeman JR, Erlanger D, Webbe F, Barth JT (2005) Sex differences in outcome following sports-related concussion. J Neurosurg 102:856–863
Buchele F, Morawska MM, Schreglmann SR, Penner M, Muser M, Baumann CR, Noain D (2016) Novel rat model of weight drop-induced closed diffuse traumatic brain injury compatible with electrophysiological recordings of vigilance states. J Neurotrauma 33:1171–1180. doi:10.1089/neu.2015.4001
Cassidy JD, Carroll LJ, Peloso PM, et al. (2004) Incidence, risk factors and prevention of mild traumatic brain injury: results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. J Rehabil Med 43(Suppl):28–60
Chen YUN, Constantini S, Trembovler V, Weinstock M, Shohami E (1996) An experimental model of closed head injury in mice: pathophysiology, histopathology, and cognitive deficits. J Neurotrauma 13:557–568. doi:10.1089/neu.1996.13.557
Cornelius C, Crupi R, Calabrese V et al (2013) Traumatic brain injury: oxidative stress and neuroprotection. Antioxid Redox Signal 19:836–853. doi:10.1089/ars.2012.4981
Dewitt DS, Perez-Polo R, Hulsebosch CE, Dash PK, Robertson CS (2013) Challenges in the development of rodent models of mild traumatic brain injury. J Neurotrauma 30:688–701. doi:10.1089/neu.2012.2349
Fehily B, Fitzgerald M (2016) Repeated mild traumatic brain injury: potential mechanisms of damage. Cell Transplant. doi:10.3727/096368916x692807
Fitzgerald M, Bartlett CA, Harvey AR, Dunlop SA (2010) Early events of secondary degeneration after partial optic nerve transection: an immunohistochemical study. J Neurotrauma 27:439–452. doi:10.1089/neu.2009.1112
Fuss B, Mallon B, Phan T, Ohlemeyer C, Kirchhoff F, Nishiyama A, Macklin WB (2000) Purification and analysis of in vivo-differentiated oligodendrocytes expressing the green fluorescent protein. Developmental Biology 218:259–274. doi:10.1006/dbio.1999.9574
Gao L, Macklin W, Gerson J, Miller RH (2006) Intrinsic and extrinsic inhibition of oligodendrocyte development by rat retina. Developmental Biology 290:277–286. doi:10.1016/j.ydbio.2005.11.007
Gregersen R, Christensen T, Lehrmann E, Diemer N, Finsen B (2001) Focal cerebral ischemia induces increased myelin basic protein and growth-associated protein-43 gene transcription in peri-infarct areas in the rat brain. Exp Brain Res 138:384–392. doi:10.1007/s002210100715
Guskiewicz KM, Marshall SW, Bailes J, McCrea M, Cantu RC, Randolph C, Jordan BD (2005) Association between recurrent concussion and late-life cognitive impairment in retired professional football players. Neurosurgery 57:719–726. doi:10.1227/01.neu.0000175725.75780.dd
Hall ED, Detloff MR, Johnson K, Kupina NC (2004) Peroxynitrite-mediated protein nitration and lipid peroxidation in a mouse model of traumatic brain injury. J Neurotrauma 21:9–20. doi:10.1089/089771504772695904
Hall ED, Vaishnav RA, Mustafa AG (2010) Antioxidant therapies for traumatic brain injury. Neurotherapeutics 7:51–61. doi:10.1016/j.nurt.2009.10.021
Higgins GC, Beart PM, Shin YS, Chen MJ, Cheung NS, Nagley P (2010) Oxidative stress: emerging mitochondrial and cellular themes and variations in neuronal injury. J Alzheimer’s Dis 20:453–473
Hohl A, da Silva Gullo J, Silva CCP et al (2012) Plasma levels of oxidative stress biomarkers and hospital mortality in severe head injury: a multivariate analysis. J Crit Care 27:523-e11
Ito D, Imai Y, Ohsawa K, Nakajima K, Fukuuchi Y, Kohsaka S (1998) Microglia-specific localisation of a novel calcium binding protein, Iba1. Mol Brain Res 57:1–9. doi:10.1016/S0169-328x(98)00040-0
Itoh T, Satou T, Nishida S, Tsubaki M, Imano M, Hashimoto S, Ito H (2010) Edaravone protects against apoptotic neuronal cell death and improves cerebral function after traumatic brain injury in rats. Neurochem Res 35:348–355
Kane MJ, Angoa-Pérez M, Briggs DI, Viano DC, Kreipke CW, Kuhn DM (2012) A mouse model of human repetitive mild traumatic brain injury. J Neurosci Methods 203:41–49. doi:10.1016/j.jneumeth.2011.09.003
Karr JE, Areshenkoff CN, Garcia-Barrera MA (2014) The neuropsychological outcomes of concussion: a systematic review of meta-analyses on the cognitive sequelae of mild traumatic brain injury. Neuropsychology 28:321–336. doi:10.1037/neu0000037
Kozlowski P, Raj D, Liu J, Lam C, Yung AC, Tetzlaff W (2008) Characterizing white matter damage in rat spinal cord with quantitative MRI and histology. J Neurotrauma 25:653–676
Kraus MF, Susmaras T, Caughlin BP, Walker CJ, Sweeney JA, Little DM (2007) White matter integrity and cognition in chronic traumatic brain injury: a diffusion tensor imaging study. Brain 130:2508–2519. doi:10.1093/brain/awm216
Loane DJ, Byrnes KR (2010) Role of Microglia in Neurotrauma. Neurotherapeutics 7:366–377. doi:10.1016/j.nurt.2010.07.002
Loane DJ, Kumar A, Stoica BA, Cabatbat R, Faden AI (2014) Progressive neurodegeneration after experimental brain trauma: association with chronic microglial activation. J Neuropathol Exp Neurol 73:14–29
Longhi L, Saatman KE, Fujimoto S et al (2005) Temporal window of vulnerability to repetitive experimental concussive brain injury. Neurosurgery 56:364–374 (discussion 364–374)
Luo J, Nguyen A, Villeda S et al (2014) Long-term cognitive impairments and pathological alterations in a mouse model of repetitive mild traumatic brain injury. Front Neurol 5:12. doi:10.3389/fneur.2014.00012
Lyeth BG, Jenkins LW, Hamm RJ et al (1990) Prolonged memory impairment in the absence of hippocampal cell death following traumatic brain injury in the rat. Brain Res 526:249–258. doi:10.1016/0006-8993(90)91229-A
Mannix R, Meehan WP, Mandeville J et al (2013) Clinical correlates in an experimental model of repetitive mild brain injury. Ann Neurol 74:65–75. doi:10.1002/ana.23858
Meehan WPI, Zhang J, Mannix R, Whalen MJ (2012) Increasing recovery time between injuries improves cognitive outcome after repetitive mild concussive brain injuries in mice. Neurosurgery 71:885–892. doi:10.1227/NEU.0b013e318265a439
Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods 11:47–60. doi:10.1016/0165-0270(84)90007-4
Mouzon B, Chaytow H, Crynen G et al (2012) Repetitive mild traumatic brain injury in a mouse model produces learning and memory deficits accompanied by histological changes. J Neurotrauma 29:2761–2773. doi:10.1089/neu.2012.2498
Mouzon BC, Bachmeier C, Ferro A et al (2014) Chronic neuropathological and neurobehavioral changes in a repetitive mild traumatic brain injury model. Ann Neurol 75:241–254. doi:10.1002/ana.24064
Mullen RJ, Buck CR, Smith AM (1992) NeuN, a neuronal specific nuclear protein in vertebrates. Development 116:201–211
Mychasiuk R, Hehar H, Candy S, Ma I, Esser MJ (2016) The direction of the acceleration and rotational forces associated with mild traumatic brain injury in rodents effect behavioural and molecular outcomes. J Neurosci Methods 257:168–178. doi:10.1016/j.jneumeth.2015.10.002
O’Hare Doig RL, Bartlett CA, Maghzal GJ, Lam M, Archer M, Stocker R, Fitzgerald M (2014) Reactive species and oxidative stress in optic nerve vulnerable to secondary degeneration. Exp Neurol 261C:136–146. doi:10.1016/j.expneurol.2014.06.007
Payne SC, Bartlett CA, Savigni DL, Harvey AR, Dunlop SA, Fitzgerald M (2013) Early proliferation does not prevent the loss of oligodendrocyte progenitor cells during the chronic phase of secondary degeneration in a cns white matter tract. PLoS One. doi:10.1371/journal.pone.0065710
Pellman EJ, Viano DC, Tucker AM, Casson IR (2003) Concussion in professional football: location and direction of helmet impacts—part 2. Neurosurgery 53:1328–1341
Pratico D, Reiss P, Tang LX, Sung S, Rokach J, McIntosh TK (2002) Local and systemic increase in lipid peroxidation after moderate experimental traumatic brain injury. J Neurochem 80:894–898
Psachoulia K, Jamen F, Young KM, Richardson WD (2009) Cell cycle dynamics of NG2 cells in the postnatal and ageing brain. Neuron Glia Biology 5:57–67. doi:10.1017/S1740925x09990354
Rabadi MH, Jordan BD (2001) The cumulative effect of repetitive concussion in sports. Clin J Sport Med 11:194–198
Ramos-Zuniga R, Gonzalez-de La Torre M, Jimenez-Maldonado M et al (2014) Postconcussion syndrome and mild head injury: the role of early diagnosis using neuropsychological tests and functional magnetic resonance/spectroscopy. World Neurosurg 82:828–835. doi:10.1016/j.wneu.2013.09.018
Roof RL, Hall ED (2000) Estrogen-related gender difference in survival rate and cortical blood flow after impact-acceleration head injury in rats. J Neurotrauma 17:1155–1169. doi:10.1089/neu.2000.17.1155
Saunders NR, Dziegielewska KM, Mollgard K, Habgood MD (2015) Markers for blood-brain barrier integrity: how appropriate is Evans blue in the twenty-first century and what are the alternatives? Front Neurosci 9:385. doi:10.3389/fnins.2015.00385
Schneider C, Porter NA, Brash AR (2008) Routes to 4-hydroxynonenal: fundamental issues in the mechanisms of lipid peroxidation. J Biol Chem 283:15539–15543. doi:10.1074/jbc.R800001200
Shitaka Y, Tran HT, Bennett RE, Sanchez L, Levy MA, Dikranian K, Brody DL (2011) Repetitive closed-skull traumatic brain injury in mice causes persistent multifocal axonal injury and microglial reactivity. J Neuropathol Exp Neurol 70:551–567. doi:10.1097/NEN.0b013e31821f891f
Signoretti S, Vagnozzi R, Tavazzi B, Lazzarino G (2010) Biochemical and neurochemical sequelae following mild traumatic brain injury: summary of experimental data and clinical implications. Neurosurg Focus 29:1–12. doi:10.3171/2010.9.FOCUS10183
Singh IN, Sullivan PG, Deng Y, Mbye LH, Hall ED (2006) Time course of post-traumatic mitochondrial oxidative damage and dysfunction in a mouse model of focal traumatic brain injury: implications for neuroprotective therapy. J Cereb Blood Flow Metab 26:1407–1418. doi:10.1038/sj.jcbfm.9600297
Smith SL, Andrus PK, Zhang JR, Hall ED (1994) Direct measurement of hydroxyl radicals, lipid peroxidation, and blood-brain barrier disruption following unilateral cortical impact head injury in the rat. J Neurotrauma 11:393–404. doi:10.1089/neu.1994.11.393
Stahel PF, Shohami E, Younis FM et al (2000) Experimental closed head injury[colon] analysis of neurological outcome, blood-brain barrier dysfunction, intracranial neutrophil infiltration, and neuronal cell death in mice deficient in genes for pro-inflammatory cytokines. J Cereb Blood Flow Metab 20:369–380
Szymanski CR, Chiha W, Morellini N et al (2013) Paranode abnormalities and oxidative stress in optic nerve vulnerable to secondary degeneration: modulation by 670 nm light treatment. PLoS One 8:e66448. doi:10.1371/journal.pone.0066448PONE-D-13-08628
Tavazzi B, Vagnozzi R, Signoretti S et al (2007) Temporal window of metabolic brain vulnerability to concussions: oxidative and nitrosative stresses– part II. Neurosurgery 61:390–396
Tyurin VA, Tyurina YY, Borisenko GG et al (2000) Oxidative stress following traumatic brain injury in rats. J Neurochem 75:2178–2189. doi:10.1046/j.1471-4159.2000.0752178.x
Vagnozzi R, Tavazzi B, Signoretti S et al (2007) Temporal window of metabolic brain vulnerability to concussions: mitochondrial-related impairment—part I. Neurosurgery 61:379–389. doi:10.1227/01.NEU.0000280002.41696.D8
Valavanidis A, Vlachogianni T, Fiotakis C (2009) 8-hydroxy-2′ -deoxyguanosine (8-OHdG): a critical biomarker of oxidative stress and carcinogenesis. J Environ Sci Health Part C 27:120–139. doi:10.1080/10590500902885684
Wells J, Kilburn MR, Shaw JA, Bartlett CA, Harvey AR, Dunlop SA, Fitzgerald M (2012) Early in vivo changes in calcium ions, oxidative stress markers, and ion channel immunoreactivity following partial injury to the optic nerve. J Neurosci Res 90:606–618. doi:10.1002/jnr.22784
Werner C, Engelhard K (2007) Pathophysiology of traumatic brain injury. Br J Anaesth 99:4–9. doi:10.1093/bja/aem131
Yamada KH, Kozlowski DA, Seidl SE et al (2012) Targeted gene inactivation of calpain-1 suppresses cortical degeneration due to traumatic brain injury and neuronal apoptosis induced by oxidative stress. J Biol Chem 287:13182–13193. doi:10.1074/jbc.M111.302612
Zhang QG, Laird MD, Han D et al (2012) Critical role of NADPH oxidase in neuronal oxidative damage and microglia activation following traumatic brain injury. PLoS One 7:e34504. doi:10.1371/journal.pone.0034504
Acknowledgements
We acknowledge financial support from the Department of Health Western Australia Merit Award. MF is supported by an NHMRC Career Development Fellowship (APP1087114). We thank Dr Caitlin Wyrwoll for kindly allowing us to use her ANY-Maze software and Dr Jeremy Smith for providing selected antibodies.
Disclosure statement
No competing financial interests exist.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yates, N.J., Lydiard, S., Fehily, B. et al. Repeated mild traumatic brain injury in female rats increases lipid peroxidation in neurons. Exp Brain Res 235, 2133–2149 (2017). https://doi.org/10.1007/s00221-017-4958-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-017-4958-8