Abstract
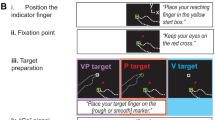
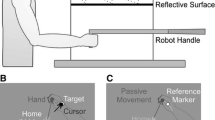
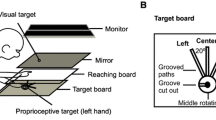
Reaching to targets with misaligned visual feedback of the hand leads to changes in proprioceptive estimates of hand position and reach aftereffects. In such tasks, subjects are able to make use of two error signals: the discrepancy between the desired and actual movement, known as the sensorimotor error signal, and the discrepancy between visual and proprioceptive estimates of hand position, which we refer to as the cross-sensory error signal. We have recently shown that mere exposure to a sensory discrepancy in the absence of goal-directed movement (i.e. no sensorimotor error signal) is sufficient to produce similar changes in felt hand position and reach aftereffects. Here, we sought to determine the extent that this cross-sensory error signal can contribute to proprioceptive recalibration and movement aftereffects by manipulating the magnitude of this signal in the absence of volitional aiming movements. Subjects pushed their hand out along a robot-generated linear path that was gradually rotated clockwise relative to the path of a cursor. On all trials, subjects viewed a cursor that headed directly towards a remembered target while their hand moved out synchronously. After exposure to a 30° rotated hand-cursor distortion, subjects recalibrated their sense of felt hand position and adapted their reaches. However, no additional increases in recalibration or aftereffects were observed following further increases in the cross-sensory error signal (e.g. up to 70°). This is in contrast to our previous study where subjects freely reached to targets with misaligned visual hand position feedback, hence experiencing both sensorimotor and cross-sensory errors, and the distortion magnitude systematically predicted increases in proprioceptive recalibration and reach aftereffects. Given these findings, we suggest that the cross-sensory error signal results in changes to felt hand position which drive partial reach aftereffects, while larger aftereffects that are produced after visuomotor adaptation (and that vary with the size of distortion) are related to the sensorimotor error signal.





Similar content being viewed by others
References
Abeele S, Bock O (2001) Sensorimotor adaptation to rotated visual input: different mechanisms for small versus large rotations. Exp Brain Res 140:407–410
Berniker M, Kording K (2012) Motor adaptation and proprioceptive estimates of limb state. Society for Neuroscience, New Orleans
Berniker M, Kording K (2008) Estimating the sources of motor errors for adaptation and generalization. Nat Neurosci 11:1454–1461
Block HJ, Bastian AJ (2012) Cerebellar involvement in motor but not sensory adaptation. Neuropsychologia 50(8):1766–1775
Cressman EK, Henriques DY (2009) Sensory recalibration of hand position following visuomotor adaptation. J Neurophysiol 102:3505–3518
Cressman EK, Henriques DY (2010) Reach adaptation and proprioceptive recalibration following exposure to misaligned sensory input. J Neurophysiol 103:1888–1895
Cressman EK, Salomonczyk D, Henriques DY (2010) Visuomotor adaptation and proprioceptive recalibration in older adults. Exp Brain Res 205:533–544
Harris CS (1963) Adaptation to displaced vision: visual, motor, or proprioceptive change? Science 140:812–813
Hay JC, Pick HL Jr (1966) Visual and proprioceptive adaptation to optical displacement of the visual stimulus. J Exp Psychol 71:150–158
Henriques DY, Cressman EK (2012) Visuomotor adaptation and proprioceptive recalibration. J Mot Behav 44:435–444
Henriques DY, Soechting JF (2003) Bias and sensitivity in the haptic perception of geometry. Exp Brain Res 150:95–108
Hinder MR, Riek S, Tresilian JR, de Rugy A, Carson RG (2010) Real-time error detection but not error correction drives automatic visuomotor adaptation. Exp Brain Res 201:191–207
Izawa J, Criscimagna-Hemminger SE, Shadmehr R (2012) Cerebellar contributions to reach adaptation and learning sensory consequences of action. J Neurosci 32:4230–4239
Jones SA, Cressman EK, Henriques DY (2010) Proprioceptive localization of the left and right hands. Exp Brain Res 204:373–383
Jones SA, Byrne PA, Fiehler K, Henriques DY (2012) Reach endpoint errors do not vary with movement path of the proprioceptive target. J Neurophysiol 107:3316–3324
Kawato M (1999) Internal models for motor control and trajectory planning. Curr Opin Neurobiol 9:718–727
Kesten H (1958) Accelerated stochastic approximation. Ann Math Stat 29:41–59
Krakauer JW, Ghilardi MF, Ghez C (1999) Independent learning of internal models for kinematic and dynamic control of reaching. Nat Neurosci 2:1026–1031
Krakauer JW, Pine ZM, Ghilardi MF, Ghez C (2000) Learning of visuomotor transformations for vectorial planning of reaching trajectories. J Neurosci 20:8916–8924
Marko MK, Haith AM, Harran MD, Shadmehr R (2012) Sensitivity to prediction error in reach adaptation. J Neurophysiol 108(6):1752–1763
Martin TA, Keating JG, Goodkin HP, Bastian AJ, Thach WT (1996a) Throwing while looking through prisms. I. Focal olivocerebellar lesions impair adaptation. Brain 119(Pt 4):1183–1198
Martin TA, Keating JG, Goodkin HP, Bastian AJ, Thach WT (1996b) Throwing while looking through prisms. II. Specificity and storage of multiple gaze-throw calibrations. Brain 119(Pt 4):1199–1211
Miall RC, Wolpert DM (1996) Forward models for physiological motor control. Neural Netw 9:1265–1279
Redding GM, Wallace B (1996) Adaptive spatial alignment and strategic perceptual-motor control. J Exp Psychol Hum Percept Perform 22:379–394
Redding GM, Wallace B (2000) Prism exposure aftereffects and direct effects for different movement and feedback times. J Mot Behav 32:83–99
Redding GM, Wallace B (2004) First-trial adaptation to prism exposure: artifact of visual capture. J Mot Behav 36:291–304
Salomonczyk D, Cressman EK, Henriques DY (2011) Proprioceptive recalibration following prolonged training and increasing distortions in visuomotor adaptation. Neuropsychologia 49:3053–3062
Salomonczyk D, Henriques DY, Cressman EK (2012) Proprioceptive recalibration in the left and right hands following visuomotor adaptation. Exp Brain Res 217:187–196
Shadmehr R, Smith MA, Krakauer JW (2010) Error correction, sensory prediction, and adaptation in motor control. Annu Rev Neurosci 33:89–108
Simani MC, McGuire LM, Sabes PN (2007) Visual-shift adaptation is composed of separable sensory and task-dependent effects. J Neurophysiol 98:2827–2841
Synofzik M, Lindner A, Thier P (2008) The cerebellum updates predictions about the visual consequences of one’s behavior. Curr Biol 18:814–818
Treutwein B (1995) Adaptive psychophysical procedures. Vision Res 35:2503–2522
Tseng YW, Diedrichsen J, Krakauer JW, Shadmehr R, Bastian AJ (2007) Sensory prediction errors drive cerebellum-dependent adaptation of reaching. J Neurophysiol 98:54–62
van Beers RJ, Wolpert DM, Haggard P (2002) When feeling is more important than seeing in sensorimotor adaptation. Curr Biol 12:834–837
Wei K, Kording K (2009) Relevance of error: what drives motor adaptation? J Neurophysiol 101:655–664
Wolpert DM, Kawato M (1998) Multiple paired forward and inverse models for motor control. Neural Netw 11:1317–1329
Wolpert DM, Ghahramani Z, Jordan MI (1995) An internal model for sensorimotor integration. Science 269:1880–1882
Wong A, Shelhamer M (2011) Sensorimotor adaptation error signals are derived from realistic predictions of movement outcomes. J Neurophysiol 105:1130–1140
Acknowledgments
We wish to thank Ahmed Mostafa, Steven Jesin and Nilly Nourouzpour for their assistance with data collection. DS is supported by a Natural Sciences and Engineering Research Council (NSERC) doctoral scholarship, and this work was supported by a NSERC Discovery Grants awarded to DYPH and EKC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salomonczyk, D., Cressman, E.K. & Henriques, D.Y.P. The role of the cross-sensory error signal in visuomotor adaptation. Exp Brain Res 228, 313–325 (2013). https://doi.org/10.1007/s00221-013-3564-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-013-3564-7