Abstract
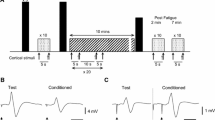
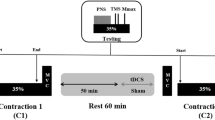
Previous studies have shown that the motor evoked potential (MEP) amplitude increases as force declines during a fatiguing muscle contraction, indicating that there is an increase in corticomotor excitability. In spite of this there is a progressive reduction in voluntary motor drive, as shown by an increase in the interpolated twitch force as fatigue develops. The aim of this study was to determine whether, by further increasing corticomotor excitability using a paired-pulse rTMS protocol designed to induce I-wave facilitation (iTMS), force loss during a sustained voluntary contraction could be reduced. We designed a cross-over study incorporating a 15-min period of iTMS (ISI 1.5 ms; 0.2 Hz; ∼AMT), following which MEP amplitude (first dorsal interosseous muscle) increased to 194 ± 38% of baseline (P < 0.05), compared to a control period of stimulation that did not increase MEP amplitude (single-pulse TMS; 0.2 Hz; ∼ 1.2 AMT). Eight right-handed healthy subjects received both iTMS and control stimulation, in a randomized order, a week apart. We measured percentage force loss at the end of a 10-s maximum right hand key-pinch task, and compared force loss before and after stimulation. There was an improvement in task performance following iTMS, with a reduction in force loss compared to pre-stimulation baseline (11.3 ± 2.0 vs. 17.6 ± 2.4%; post vs. pre; P < 0.05). There was no significant difference in force loss before and after control stimulation. The results indicate that by increasing corticomotor excitability using paired-pulse rTMS at trans-synaptic intervals, maximum voluntary force can be sustained at a higher level during a brief fatiguing maximal voluntary contraction.



Similar content being viewed by others
References
Benwell NM, Sacco P, Hammond GR, Byrnes ML, Mastaglia FL, Thickbroom GW (2006) Short-interval cortical inhibition and corticomotor excitability with fatiguing hand exercise: a central adaptation to fatigue? Exp Brain Res 170:191–198
Chen R, Gerloff C, Hallett M, Cohen LG (1997a) Involvement of the ipsilateral motor cortex in finger movements of different complexities. Ann Neurol 41:247–254
Chen R, Classen J, Gerloff C, Celnik P, Wassermann EM, Hallett M, Cohen LG (1997b) Depression of motor cortex excitability by low-frequency transcranial magnetic stimulation. Neurology 48:1398–1403
Chen R, Classen J, Gerloff C, Celnik P, Wassermann EM, Hallett M, Cohen LG (1997c) Depression of motor cortex excitability by low-frequency transcranial magnetic stimulation. Neurology 48:1398–1403
Cincotta M, Borgheresi A, Gambetti C, Balestrieri F, Rossi L, Zaccara G, Ulivelli M, Rossi S, Civardi C, Cantello R (2003) Suprathreshold 0.3 Hz repetitive TMS prolongs the cortical silent period: potential implications for therapeutic trials in epilepsy. Clin Neurophysiol 114:1827–1833
Classen J, Liepert J, Wise SP, Hallett M, Cohen LG (1998) Rapid plasticity of human cortical movement representation induced by practice. J Neurophysiol 79:1117–1123
Fujiwara T, Rothwell JC (2004) The after effects of motor cortex rTMS depend on the state of contraction when rTMS is applied. Clin Neurophysiol 115:1514–1518
Gandevia SC (2001) Spinal and supraspinal factors in human muscle fatigue. Physiol Rev 81:1725–1789
Gandevia SC, Allen GM, Butler JE, Taylor JL (1996) Supraspinal factors in human muscle fatigue: evidence for suboptimal output from the motor cortex. J Physiol (Lond) 490:529–536
Huang YZ, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC (2005) Theta burst stimulation of the human motor cortex. Neuron 45:201–206
Khedr EM, Farweez HM, Islam H (2003) Therapeutic effect of repetitive transcranial magnetic stimulation on motor function in Parkinson’s disease patients. Eur J Neurol 10:567–572
Khedr EM, Gilio F, Rothwell J (2004) Effects of low frequency and low intensity repetitive paired pulse stimulation of the primary motor cortex. Clin Neurophysiol 115:1259–1263
Kobayashi M, Hutchinson S, Theoret H, Schlaug G, Pascual-Leone A (2004) Repetitive TMS of the motor cortex improves ipsilateral sequential simple finger movements. Neurology 62:91–98
Muellbacher W, Ziemann U, Boroojerdi B, Hallett M (2000) Effects of low-frequency transcranial magnetic stimulation on motor excitability and basic motor behavior. Clin Neurophysiol 111:1002–1007
Muellbacher W, Ziemann U, Boroojerdi B, Cohen L, Hallett M (2001) Role of the human motor cortex in rapid motor learning. Exp Brain Res 136:431–438
Muellbacher W, Ziemann U, Wissel J, Dang N, Kofler M, Facchini S, Boroojerdi B, Poewe W, Hallett M (2002) Early consolidation in human primary motor cortex. Nature 415:640–644
Pascual-Leone A, Valls-Sole J, Wassermann EM, Hallett M (1994) Responses to rapid-rate transcranial magnetic stimulation of the human motor cortex. Brain 117(Pt 4):847–858
Quartarone A, Bagnato S, Rizzo V, Morgante F, Sant’angelo A, Battaglia F, Messina C, Siebner HR, Girlanda P (2005) Distinct changes in cortical and spinal excitability following high-frequency repetitive TMS to the human motor cortex. Exp Brain Res 161:114–124
Rossi S, Pasqualetti P, Rossini PM, Feige B, Ulivelli M, Glocker FX, Battistini N, Lucking CH, Kristeva-Feige R (2000) Effects of repetitive transcranial magnetic stimulation on movement-related cortical activity in humans. Cereb Cortex 10:802–808
Sacco P, Thickbroom GW, Thompson ML, Mastaglia FL (1997) Changes in corticomotor excitation and inhibition during prolonged sub-maximal muscle contractions. Muscle Nerve 20:1158–1166
Siebner HR, Mentschel C, Auer C, Conrad B (1999a) Repetitive transcranial magnetic stimulation has a beneficial effect on bradykinesia in Parkinson’s disease. Neuroreport 10:589–594
Siebner HR, Tormos JM, Ceballos-Baumann AO, Auer C, Catala MD, Conrad B, Pascual-Leone A (1999b) Low-frequency repetitive transcranial magnetic stimulation of the motor cortex in writer’s cramp. Neurology 52:529–537
Stefan K, Kunesch E, Cohen LG, Benecke R, Classen J (2000) Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 123(Pt 3):572–584
Strens LH, Fogelson N, Shanahan P, Rothwell JC, Brown P (2003) The ipsilateral human motor cortex can functionally compensate for acute contralateral motor cortex dysfunction. Curr Biol 13:1201–1205
Taylor JL, Butler JE, Allen GM, Gandevia SC (1996) Changes in motor cortical excitability during human muscle fatigue. J Physiol (Lond) 490:519–528
Taylor JL, Allen GM, Butler JE, Gandevia SC (2000) Supraspinal fatigue during intermittent maximal voluntary contractions of the human elbow flexors. J Appl Physiol 89:305–313
Thickbroom GW, Byrnes ML, Edwards DJ, Mastaglia FL (2006) Repetitive paired-pulse TMS at I-wave periodicity markedly increases corticospinal excitability: a new technique for modulating synaptic plasticity. Clin Neurophysiol 117:61–66
Touge T, Gerschlager W, Brown P, Rothwell JC (2001) Are the after-effects of low-frequency rTMS on motor cortex excitability due to changes in the efficacy of cortical synapses? Clin Neurophysiol 112:2138–2145
Acknowledgments
This study was supported by the Neuromuscular Foundation of Western Australia. NMB is a recipient of an Australian Postgraduate Award, Jean Rogerson Postgraduate Scholarship and 2004 Woodside Neurotrauma PhD Excellence Award.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Benwell, N.M., Mastaglia, F.L. & Thickbroom, G.W. Paired-pulse rTMS at trans-synaptic intervals increases corticomotor excitability and reduces the rate of force loss during a fatiguing exercise of the hand. Exp Brain Res 175, 626–632 (2006). https://doi.org/10.1007/s00221-006-0579-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-006-0579-3