Abstract
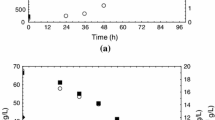
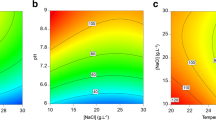
This study aimed at optimising the cultivation conditions for the production of carotenoids by Sporidiobolus salmonicolor (CBS 2636) in a bioreactor. The maximum content of total carotenoids in the full factorial design 2² was 3131.3 μg/L in synthetic medium with 80 g/L of glucose, 15 g/L of peptone, 5 g/L of malt extract, aeration of 1.5 vvm, agitation of 180 rpm, initial pH of 4.0 at 25 °C. In the kinetic study, we could observe that the bioproduction of carotenoids is associated with cell growth in the exponential phase, and the average specific growth (μ) in bioreactor is 0.046 h−1 with a maximum yield of 0.19 g cells/L h. The maximum yield of carotenoids (60.0 μg/Lh) is observed at 50-h bioproduction. The conversion factor for total organic carbon (TOC) in cells (YX/SCOT) was 2.97 g/g (0–50 h) and 0.254 g/g (50–100 h), the conversion factor glucose into cells (YX/Sglicose) was 0.168 g/g (0–100 h). The specific production of carotenoids (YP/X) was 390 μg of carotenoids per gram of cells, the conversion factor of carbon in the product (YP/SCOT) was 107.8 μg/g (0–50 h) and 34.4 μg/g (50–100 h), whereas the factor YP/Sglicose was 69.59 μg/g. The agitation and aeration provided better homogeneity in the culture medium, and hence greater availability of nutrients and oxygen, leading to higher production of carotenoids.



Similar content being viewed by others
References
Das A, Yoon S, Lee S, Kim J, Oh D, Kim S (2007) An update on microbial carotenoid production: application of recent metabolic engineering tools. Appl Microbiol Biotechnol 77:505–512
Botella-Pavía P, Rodríguez-Concepción M (2006) Carotenoid biotechnology in plants for nutritionally improved foods. Physiol Plant 126:369–381
El-Agamey A, Lowe GM, Mcgarvey DJ, Mortensen A, Phillip DM, Truscott G, Young AJ (2004) Carotenoid radical chemistry and antioxidant/pro-oxidant properties. Arch Biochem Biophys 430:37–48
Aksu Z, Eren AT (2007) Production of carotenoids by isolated yeast of Rhodotorula glutinis. Biochem Eng J 35:107–113
Krinsky NI (2001) Carotenoids as antioxidants. Nutrition 17:815–817
Gu Z, Deming C, Yongbin H, Zhigang C, Feirong G (2008) Optimization of carotenoids extraction from Rhodobacter sphaeroides. Food Sci Technol 14:1082–1088
Demming-Adams B, Gilmore AM, Adams WW (2006) In vivo functions of carotenoids in higher plants. Faseb J 10:403–412
Aguilar CP, González M, Cifuentes AS, Silva M (2004) Growth and accumulation of total carotenoids in two strains of Dunaliella salina Teod. (Chlorophyceae) from the northern and central coast of Perú. J Chilean Chem Soc 49:69–74
Fazeli MR, Tofighi H, Samadi N, Jamalifar H (2006) Effects of salinity on b-carotene production by Dunaliella tertiolecta DCCBC 26 isolated from Urmia salt lake, North of Iran. Bioresour Technol 97:2453–2456
Po-Fung I, Feng C (2005) Production of astaxanthin by the green microalga Chlorella zofingiensis in the dark. Process Biochem 40:733–738
García-González M, Moreno J, Manzano JC, Florencio FJ, Guerrero MG (2005) Production of Dunaliella salina biomass rich in 9-cis-β-carotene and lutein in a closed tubular photobioreactor. J Biotechnol 115:81–90
Dufossé L, Galaup P, Yaron A, Arad SM, Blanc P, Murthy KNC (2005) Microorganisms and microalgae as sources of pigments for food use: a scientific oddity or an industrial reality. Trends Food Sci Technol 16:389–406
Johnson EA, Schroeder WA (1995) Microbial carotenoids. Adv Biochem Eng Biotechnol 11:297–326
Goodwin TW (1980) The biochemistry of the carotenoids. Chapman & Hall, London
Hu ZC, Zheng YG, Wang Z, Shen YC (2006) pH control strategy in astaxanthin fermentation bioprocess by Xanthophyllomyces dendrorhous. Enzyme Microb Technol 39:586–590
Park PK, Kim EY, Chu KH (2007) Chemical disruption of yeast cells for the isolation of carotenoid pigments. Sep Purific Technol 53:148–152
Tinoi J, Rakariyatham N, Deming RL (2005) Simplex optimization of carotenoid production by Rhodotorula glutinis using hydrolyzed mung bean waste flour as substrate. Process Biochem 40:2551–2557
Davoli P, Mierau V, Weber RWS (2004) Carotenoids and fatty acids in red yeasts Sporobolomyces roseus and Rhodotorula glutinis. Appl Biochem Microbiol 40:392–397
Razavi SH, March I (2006) Effect of temperature and pH on the growth kinetics and carotenoid production by Sporobolomyces ruberrimus H110 using technical glycerol as carbon source. Iranian J Chem Eng 23:59–64
Maldonade IR, Rodriguez-Amaya DB, Scamparini ARP (2008) Carotenoids of yeasts isolated from the Brazilian ecosystem. Food Chem 107:145–150
Valduga E, Valerio A, Treichel H, Di Luccio M, Furigo AJ (2008) Study of the bio-production of carotenoids by Sporidiobolus salmonicolor (CBS 2636) using pre-treated agro-industrial substrates. J Chem Technol Biotechnol 83:1267–1274
Lim GB, Lee SY, Lee EK, Haam SJ, Kim WS (2002) Separation of astaxanthin from red yeast Phaffia rhodozyma by supercritical carbon dioxide extraction. Biochem Eng J 11:181–187
Liu YS, Wu JY, Ho KP (2006) Characterization of oxygen transfer conditions and their effects on Phaffia rhodozyma growth and carotenoid production in shake-flask cultures. Biochem Eng J 27:331–335
Fang TJ, Wang JM (2002) Extractability of astaxanthin in a mixed culture of a carotenoid over-producing mutant of Xanthophyllomyces dendrourhous and Bacillus circulans in two-stage batch fermentation. Process Biochem 37:1235–1245
Ramírez J, Gutierrez H, Gschaedler A (2001) Optimization of astaxanthin production by Phaffia rhodozyma through factorial design and response surface methodology. J Biotechnol 88:259–268
Valduga E, Tatsch PO, Tiggemann L, Zeni J, Colet R, Cansian JM, Treichel H, Di Luccio M (2009) Evaluation of the conditions of carotenoids production in a synthetic medium by Sporidiobolus salmonicolor (CBS 2636) in a bioreactor. Int J Food Sci Technol 44:2445–2451
Valduga E, Valerio A, Tatsch PO, Treichel H, Furigo AJ, Di Luccio M (2009) Assessment of cell disruption and carotenoids extraction from Sporidiobolus salmonicolor (CBS 2636). Food Bioproc Technol 2:234–238
Hiss H (2001) Cinética dos Processos Fermentativos. Editora Edgard Blücher Ltda, São Paulo
Bailey JE, Ollis DF (1986) Biochemical engineering fundamentals. McGraw-Hill Book Company, New York
Davies BH (1976) Carotenoid. Chemistry and biochemistry of plant pigments. Academic Press, New York
Silva C, Cabral JMS, Keulen FV (2004) Isolation of a β-total carotenoids over-producing soil bacterium, Sphingomonas sp. Biotechnol Lett 26:257–262
Association of Official Analytical Chemists, AOAC (1995) Official methods of analysis of the association of the analytical chemists, Washington, 1995
Miller GL (1959) Use of dinitrosalicyclic acid reagent for determination of reducing sugar. Analytical Chem 31:426–428
Buzzini P, Innocenti M, Turchetti B, Libkind D, van Broock M, Mulinacci N (2007) Carotenoid profiles of yeasts belonging to the genera Rhodotorula, Rhodosporidium, Sporobolomyces, and Sporidiobolus. Can J Microbiol 53:1024–1031
Choudari SM, Singhal R (2008) Media optimization for the production of b-carotene by Blakeslea trispora: a statistical approach. Bioresour Technol 99:722–730
Valduga E, Valerio A, Treichel H, Furigo AJ, Di Luccio M (2009) Optimization of the bio-production of total carotenoids by Sporidiobolus salmonicolor (CBS 2636) using response surface technique. Food Bioproc Technol 2:415–421
Garbayo I, Vílchez C, Nava-Saucedo JE, Barbotin JN (2003) Nitrogen, carbon and light-mediated regulation studies of carotenoid biosynthesis in immobilized mycelia of Gibberella fujikuroi. Enzyme Microb Technol 33:629–634
Liu YS, Wu JY (2007) Optimization of cell growth and carotenoid production of Xanthophyllomyces dendrorhous through statistical experiment design. Biochem Eng J 36:182–189
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valduga, E., Schwartz, C.R.M., Tatsch, P.O. et al. Evaluation of aeration and substrate concentration on the production of carotenoids by Sporidiobolus salmonicolor (CBS 2636) in bioreactor. Eur Food Res Technol 232, 453–462 (2011). https://doi.org/10.1007/s00217-010-1410-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-010-1410-8