Abstract
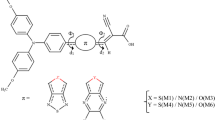
A series of metal-free donor–π-bridge–acceptor dyes based on phenoxazine as the electron donor and cyanoacrylic acid as the electron acceptor groups with different π-spacers were studied by density functional theory (DFT) and time-dependent DFT calculations. The aromatic rings such as benzene, furan, thiophene, pyrrole, thiazole, and silole were used as π-spacer to link the donor and acceptor moieties. All of the dyes with π-spacer exhibited a narrower band gap than the parent molecule without π-spacer due to the π-conjugation expansion. The results showed that the dye with a furan spacer (POXF) is the most efficient sensitizer for dye-sensitized solar cell due to the largest LHE, high V oc, and red-shifted absorption.







Similar content being viewed by others
References
Standridge SD, Schatz GC, Hupp JT (2009) J Am Chem Soc 131:8407–8409
Sánchez-de-Armas R, Lόpez JO, San-Miguel MA, Sanz JF (2010) J Chem Theory Comput 6:2856–2865
Feng J, Jiao Y, Ma W, Nazeeruddin MK, Grätzel M, Meng S (2013) J Phys Chem C 117:3772–3778
Agrawal S, Pastore M, Marotta G, Reddy MA, Chandrasekharam M, Angelis FD (2013) J Phys Chem C 117:9613–9622
Liang M, Xu W, Cai F, Chen P, Peng B, Chen J, Li Z (2007) J Phys Chem C 111:4465–4472
Singh SP, Roy MS, Justin Thomas KR, Balaiah S, Bhanuprakash K, Sharma GD (2012) J Phys Chem C 116:5941–5950
Justin Thomas KR, Hsu YC, Lin JT, Lee K, Ho KC, Lai CH, Cheng YM, Chou PT (2008) Chem Mater 20:1830–1840
Zhang G, Bala H, Cheng Y, Shi D, Lv X, Yu Q, Wang P (2009) Chem Commun 2198–2200
Ko SB, Cho AN, Kim MJ, Lee CR, Park NG (2012) Dyes Pigment 94:88–98
Miura H, Uchida S, Takata M, Sumioka K, Liska P (2008) Chem Commun 41:5194–5196
Koumura N, Wang ZS, Miyashita M, Uemura Y, Sekiguchi H, Cui Y, Mori A, Mori S, Hara K (2009) J Mater Chem 19:4829–4836
Hara K, Wang ZS, Sato T, Furube A, Katoh R, Sugihara H, Dan-oh Y, Kasada C, Shinpo A, Suga S (2005) J Phys Chem B 109:15476–15482
Kim S, Lee JK, Kang SO, Ko J, Yum JH, Fantacci S, Angelis FD, Censo DD, Nazeeruddin MK, Grätzel M (2006) J Am Chem Soc 128:16701–16707
Burke A, Schmidt-Medne L, Ito S, Grätzel M (2007) Chem Commun 3:234–236
Mori S, Nagata M, Nakahata Y, Yasuta K, Goto R, Kimura M, Taya M (2010) J Am Chem Soc 132:4054–4055
Li C, Yum JH, Moon SJ, Herrmann A, Eickemeyer F, Pschirer NG, Erk P, Schöneboom J, Müllen K, Grätzel M (2008) ChemSusChem 1:615–618
Kitamura T, Ikeda M, Shigaki K, Inoue T, Anderson NA, Ai X, Lian T, Yanagida S (2004) Chem Mater 16:1806–1812
Kim C, Choi H, Kim S, Baik C, Song K, Kang MS, Kang SO, Ko J (2008) J Org Chem 73:7072–7079
Liu WH, Wu IC, Lai CH, Lai CH, Chou PT, Li YT, Chen CL, Hsu YY, Chi Y (2008) Chem Commun 5152–5154
Yum JH, Hagberg DP, Moon SJ, Karlsson KM, Marinado T, Sun L, Hagfeldt A, Nazeeruddin MK, Grätzel M (2009) Angew Chem Int Ed 48:1576–1580
Zhang G, Bai Y, Li R, Shi D, Wenger S, Zakeeruddin SM, Grätzel M, Wang P (2009) Energy Environ Sci 2:92–95
Akhtaruzzaman Md, Islam MA, El-Shafei A, Asao N, Jin T, Han L, Alamry KA, Kosa SA, Asiri AM, Yamamoto Y (2013) Tetrahedron 69:3444–3450
Qin H, Wenger S, Xu M, Gao F, Jing X, Wang P, Zakeeruddin SM, Grätzel M (2008) J Am Chem Soc 130:9202–9203
Wang ZS, Cui Y, Hara K, Dan-oh Y, Kasada C, Shinpo A (2007) Adv Mater 19:1138–1141
Kim S, Choi H, Kim D, Song K, Kang SO, Ko J (2007) Tetrahedron 63:9206–9212
Kim S, Choi H, Baik C, Song K, Kang SO, Ko J (2007) Tetrahedron 63:11436–11443
Wan ZQ, Jia CY, Zhang JQ, Duan YD, Lin Y, Shi Y (2012) J Power Sources 199:426–431
Li SL, Jiang KJ, Shao KF, Yang LM (2006) Chem Commun 2792–2794
Koenigsmann C, Brennan BJ, Negre CFA, Koepf M, Durrell AC, Milot RL, Batista VS, Brudvig GW, Crabtree RH, Schmuttenmaer CA, Ripolles TS, Torre JA, Bisquert J (2014) Phys Chem Chem Phys 16:16629–16641
Yu X, Ci Z, Liu T, Feng X, Wang C, Ma T, Bao M (2014) Dyes Pigment 102:126–132
Lee W, Yuk SB, Choi J, Kim HJ, Kim HW, Kim SH, Kim B, Ko MJ, Kim JP (2014) Dyes Pigment 102:13–21
Lee W, Choi J, Namgoong JW, Kim SH, Sun KC, Jeong SH, Yoo K, Ko MJ, Kim JP (2014) Dyes Pigment 104:185–193
Li M, Kou L, Diao L, Zhang Q, Li Z, Wu Q, Lu W, Pan D (2015) J Phys Chem A 119:3299–3309
Pastore M, Fantacci S, Angelis FD (2010) J Phys Chem C 114:22742–22750
Peng B, Yang S, Li L, Cheng F, Chen J (2010) J Chem Phys 132:034305–034309
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross HB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, revision A. 02; Gaussian. Inc, Wallingford, CT
Parr RG, Yang W (1989) Density-functional theory of atoms and molecules. Oxford University Press, New York
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Becke AD (1993) J Chem Phys 98:5648–5652
Hehre WJ, Ditchfield R, Pople JA (1972) J Chem Phys 56:2257–2261
Dunning Jr TH, Hay PJ (1977) Modern theoretical chemistry. In: Schaefer III HF (ed), vol 3. Plenum, New York
Hay PJ, Wadt WR (1985) J Chem Phys 82:270–283
Wadt WR, Hay PJ (1985) J Chem Phys 82:284–298
Zhang J, Li HB, Sun SL, Geng Y, Wu Y, Su ZM (2012) J Mater Chem 22:568–576
Wang CL, Wang J, Bai FQ, Chen J, Zhang HX (2014) Int J Quantum Chem 114:560–567
Zhang L, Cole JM (2015) ACS Appl Mater Interfaces 7:3427–3455
Hagfeldt A, Boschloo G, Sun L, Kloo L, Pettersson H (2010) Chem Rev 110:6595–6663
Karlsson KM, Jiang X, Eriksson SK, Gabrielsson E, Rensmo H, Hagfeldt A, Sun L (2011) Chem Eur J 17:6415–6424
Massin J, Ducasse L, Toupance T, Olivier C (2014) J Phys Chem C 118:10677–10685
Wang ZS, Hara K, Dan-oh Y, Kasada C, Shinpo A, Suga S, Arakawa H, Sugihara H (2005) J Phys Chem B 109:3907–3914
Cancès E, Mennucci B (1997) J Chem Phys 107:3032–3041
Kamiya M, Tsuneda T, Hirao K (2002) J Chem Phys 117:6010–6015
Heyd J, Scuseria G (2004) J Chem Phys 121:1187–1192
Chai JD, Head-Gordon M (2008) Phys Chem Chem Phys 10:6615–6620
Adamo C, Barone V (1999) J Chem Phys 110:6158–6170
Zhang CR, Liu ZJ, Chen YH, Chen HS, Wu YZ, Feng W, Wang DB (2010) Curr Appl Phys 10:77–83
Katoh R, Furube A, Yoshihara T, Hara K, Fujihashi G, Takano S, Murata S, Arakawa H, Tachiya M (2004) J Phys Chem B 108:4818–4822
Lu T, Chen F (2012) J Comp Chem 33:580–592
Mendizabal F, Lopéz A, Arratia-Pérez R, Zapata-Torres G (2015) Comput theor chem 1070:117–125
Pastore M, Fantacci S, Angelis FD (2013) J Phys Chem C 117:3685–3700
Yamaguchi S, Tamao K (1998) J Chem Soc, Dalton Trans 22:3693–3702
Martin RL (2003) J Chem Phys 118:4775–4777
Ferré N, Filatov M, Huix-Rotllant M (2016) Density-functional methods for excited states. Springer, Berlin
Guido CA, Cortona P, Mennucci B, Adamo C (2013) J Chem Theory Comput 9:3118–3126
Etienne T, Assfeld X, Monari A (2014) J Chem Theory Comput 10:3896–3905
Bahers TL, Adamo C, Ciofini I (2011) J Chem Theory Comput 7:2498–2506
Acknowledgements
The authors gratefully acknowledge financial support from the Isfahan University of Technology and computational support from the Sheikh Bahaei National High Performance Computing Center (SBNHPCC).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jafari Chermahini, Z., Najafi Chermahini, A. Theoretical study on the bridge comparison of TiO2 nanoparticle sensitizers based on phenoxazine in dye-sensitized solar cells. Theor Chem Acc 136, 34 (2017). https://doi.org/10.1007/s00214-017-2063-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-017-2063-5