Abstract
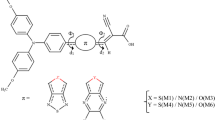
Thiophene and derivatives have been broadly used in metal-free organic dyes as π-bridge in the past 10 years. However, the relatively sharp and narrow visible absorption bands of these organic dyes not only severely attenuated the light capture capability but also restrict the efficiency. In this contribution, to design efficient sensitizers for dye-sensitized solar cells, a series of triphenylamine (TPA) dyes with 2-pyrone as the π-bridge are investigated using the density functional theory and time-dependent density functional theory approaches. The results show that the designed dyes have smaller gap between the highest occupied molecular orbital and the lowest unoccupied molecular orbital, the absorption bands are greatly bathochromic-shifted by at least 39 nm and the light-harvesting efficiencies are improved compared to the experimentally efficient sensitizer T with thiophene as the π-bridge. The calculated values of free energy change ΔG inject for all the designed dyes are very negative, which favors electron injection from the excited-state dye to the TiO2 conduction band edge. Our simulations show that the sensitizers studied here are strongly adsorbed to the TiO2 cluster. During light excitation, electrons are transferred from the TPA group through the π-spacer to the surface-bound cyanoacrylate, facilitating electron injection to the TiO2 nanoclusters. Our calculations indicate that the newly designed dyes will be promising candidates for the future solar cell applications.








Similar content being viewed by others
References
O’Regan B, Grätzel M (1991) Nature 353:737–740
Hagfeldt A, Grätzel M (1995) Chem Rev 95:49–68
Grätzel M (2001) Nature 414:338–344
Hamann TW, Jensen RA, Martinson ABF, Ryswyk HV, Hupp JT (2008) Energy Environ Sci 1:66–78
Preat J, Jacquemin D, Perpète EA (2010) Energy Environ Sci 3:891–904
Hagfeldt A, Boschloo G, Sun L, Kloo L, Pettersson H (2010) Chem Rev 110:6595–6663
Nazeeruddin MK, Pechy P, Renouard T, Zakeeruddin SM, Humphry-Baker R, Comte P, Liska P, Cevey L, Costa E, Shklover V, Spiccia L, Deacon GB, Bignozzi CA, Grätzel M (2001) J Am Chem Soc 123:1613–1624
Nazeeruddin MK, De Angelis F, Fantacci S, Selloni A, Viscardi G, Liska P, Ito S, Takeru B, Grätzel M (2005) J Am Chem Soc 127:16835–16847
Gao F, Wang Y, Zhang J, Shi D, Wang M, Humphry-Baker R, Wang P, Zakeeruddin SM, Grätzel M (2008) Chem Commun 44:2635–2637
Chen C-Y, Wang M, Li J-Y, Pootrakulchote N, Alibabaei L, Ngoc-le C, Decoppet J-D, Tsai J-H, Grätzel C, Wu C-G, Zakeeruddin SM, Grätzel M (2009) ACS Nano 3:3103–3109
Zhang GL, Bala H, Cheng YM, Shi D, Lv XJ, Yu QJ, Wang P (2009) Chem Commun 2198–2200
Yella A, Lee H-W, Tsao HN, Yi C, Chandiran AK, Nazeeruddin MK, Diau EW-G, Yeh C-Y, Zakeeruddin SM, Grätzel M (2011) Science 334:629–634
Wang ZS, Cui Y, Hara K, Dan-Oh Y, Kasada C, Shinpo A (2007) Adv Mater 19:1138–1141
Horiuchi T, Miura H, Sumioka K, Uchida S (2004) J Am Chem Soc 126:12218–12219
Kitamura T, Ikeda M, Shigaki K, Inoue T, Anderson NA, Ai X, Lian TQ, Yanagida S (2004) Chem Mater 16:1806–1812
Li C, Yum JH, Moon SJ, Herrmann A, Eickemeyer F, Pschirer NG, Erk P, Schoeboom J, Mullen K, Grätzel M, Nazeeruddin MK (2008) ChemSusChem 1:615–618
Hara K, Horiguchi T, Kinoshita T, Sayama K, Sugihara H, Arakawa H (2000) Sol Energy Mater Sol Cells 64:115–134
Hagberg DP, Yum JH, Lee HJ, De Angelis F, Marinado T, Karlsson KM, Humphry-Baker R, Sun L, Hagfeldt A, Grätzel M, Nazeeruddin MK (2008) J Am Chem Soc 130:6259–6266
Akhtaruzzaman M, Islam A, Yang F, Asao N, Kwon E, Singh SP, Han L, Yamamoto Y (2011) Chem Commun 47:12400–12402
Lin JT, Chen P-C, Yen Y-S, Hsu Y-C, Chou H-H, Yeh M-CP (2009) Org Lett 11:97–100
Xiao D, Martini LA, Snoeberger RC III, Crabtree RH, Batista VS (2011) J Am Chem Soc 133:9014–9022
Christie RM, Morgan KM, Islam MS (2008) Dyes Pigments 76:741–747
Puerto ED, Domingo C, Sanchez-Cortes S, García-Ramos JV, Aroca RF (2011) J Phys Chem C 115:16838–16843
Mizuguchi J (2004) J Phys Chem B 108:8926–8930
Thalacker C, Röger C, Würthner F (2006) J Org Chem 71:8098–8105
Ferreira ES, Hulme AN, McNab H, Quye A (2004) Chem Soc Rev 33:329–336
Seixas de Melo J, Rondão R, Burrows HD, Melo MJ, Navaratnam S, Edge R, Voss G (2006) J Phys Chem A 110:13653–13661
Hara K, Sato T, Katoh R, Furube A, Ohga Y, Shinpo A, Suga S, Sayama K, Sugihara H, Arakawa H (2003) J Phys Chem B 107:597–606
Wang Z-S, Cui Y, Dan-oh Y, Kasada C, Shinpo A, Hara K (2007) J Phys Chem C 111:7224–7230
Seo KD, Choi IT, Park YG, Kang S, Lee JY, Kim HK (2012) Dyes Pigments 94:469–474
Bochkov AY, Akchurin IO, Dyachenko OA, Traven VF (2013) Chem Commun 49:11653–11655
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JJA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision A.02. Gaussian Inc., Wallingford
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1998) Phys Rev B 37:785–789
Hariharan PC, Pople JA (1973) JA. Theor Chim Acta 28:213–222
Hay PJ, Wadt WR (1985) J Chem Phys 82:270–283
Hay PJ, Wadt WR (1985) J Chem Phys 82:284–298
Hay PJ, Wadt WR (1985) J Chem Phys 82:299–310
Preat J, Michaux C, Jacquemin D, Perpète EA (2009) J Phys Chem C 113:16821–16833
Pastore M, Mosconi E, De Angelis F, Gräzel M (2010) J Phys Chem C 114:7205–7212
Yanai T, Tew DP, Handy NC (2004) Chem Phys Lett 393:51–57
Chai J-D, Head-Gordon M (2008) Phys Chem Chem Phys 10:6615–6620
Vydrov OA, Heyd J, Krukau V, Scuseria GE (2006) J Chem Phys 125:074106
Francl MM, Pietro WJ, Hehre WJ, Binkley JS, Gordon MS, DeFrees DJ, Pople JA (1982) J Chem Phys 77:3654–3665
Barone V, Cossi M (1998) J Phys Chem A 102:1995–2001
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J. Chem. Phys. 72:650
Clark T, Chandrasekhar J, Schleyer PVR (1983) J Comp Chem 4:294–301
Ooyama Y, Harima Y (2012) ChemPhysChem 13:4032–4080
Liang M, Chen J (2013) Chem Soc Rev 42:3453–3488
Hirata N, Lagref J-J, Palomares EJ, Durrant JR, Nazeeruddin MK, Grätzel M, Censo DD (2004) Chem Eur J 10:595–602
Bonhote P, Moser JE, Humphry-Baker R, Vlachopoulos N, Zakeeruddin SM, Walder L, Grätzel M (1999) J Am Chem Soc 121:1324–1336
Clifford JN, Palomares E, Nazeeruddin MK, Thampi R, Grätzel M, Durrant JR (2004) J Am Chem Soc 126:5670–5671
Fan W, Tan D, Deng W-Q (2012) ChemPhysChem 13:2051–2060
Nalwa HS (2001) Handbook of advanced electronic and photonic materials and devices. Academic, San Diego, pp 1–3366
Jacquemin D, Preat J, Wathelet V, André JM, Perpète EA (2005) Chem Phys Lett 405:429–433
Asbury JB, Wang YQ, Hao E, Ghosh H, Lian T (2001) Res Chem Intermed 27:393–406
Barbara PF, Meyer TJ, Ratner MA (1996) J Phys Chem 100:13148–13168
Matthews D, Infelta P, Grätzel M (1996) Sol Energy Mater Sol Cells 44:119–155
Zimmermann C, Willig F, Ramakrishna S, Burfeindt B, Pettinger B, Eichberger R, Storck W (2001) J Phys Chem B 105:9245–9253
Anderson NA, Ai X, Lian T (2003) J Phys Chem B 107:14414–14421
Ramakrishna G, Singh AK, Palit DK, Ghosh HN (2004) J Phys Chem B 108:4775–4783
Katoh R, Furube A, Yoshihara T, Hara K, Fujihashi G, Takano S, Murata S, Arakawa H, Tachiya M (2004) J Phys Chem B 108:4818–4822
Vittadini A, Selloni A, Grätzel M (2000) J Phys Chem B 104:1300–1306
Guo ZY, Liang WZ (2008) J Phys Chem C 112:16655–16662
Dong H, Zhou X, Jiang C (2012) Theor Chem Acc 131:1102
Acknowledgments
Dr. Dong gratefully acknowledges the financial support from the National Science Foundation of China under Grant 21303079. Dr. Zhou appreciates the funding support by Technology Foundation for Selected Overseas Chinese Scholar from Ministry of Personnel of China.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Dong, H., Zan, Z. & Zhou, X. Can 2-pyrone derivative act as an effective π-linker for dye-sensitized solar cells: a theoretical study?. Theor Chem Acc 134, 1595 (2015). https://doi.org/10.1007/s00214-014-1595-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-014-1595-1