Abstract
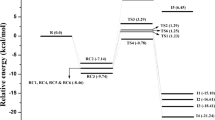
Density functional and MP2 calculations with extended basis sets were performed on the species participating in both the previously suggested and a newly proposed mechanisms of partial dechlorination of chloropicrin by simple sulfur species, both in gas phase and in a simulated water environment. Thermochemistry of both mechanisms in the gas phase was also studied using the chemical models G3 and G4. It is shown that the previously proposed reductive dehalogenation is not thermodynamically feasible at room temperature, as it should be according to the experimental evidence. Although inclusion of the solvent improves the results with respect to gas phase, the thermodynamics of the proposed mechanism by Zheng et al. is still unfavorable for obtaining the experimental products. An alternative mechanism is then proposed, involving the formation of HSCl, which is the intermediate that then undergoes redox reactions. Such a mechanism is exothermic and spontaneous, according to the computational results, and produces elementary sulfur in agreement with the experimental facts.

Similar content being viewed by others
References
Öberg G (2002) Appl Microbiol Biotechnol 58:565–581
Keppler F, Borchers R, Pracht J, Rheinsberger S, Schöler HF (2002) Environ Sci Technol 36:2479–2483
Smidt H, de Vos WM (2004) Annu Rev Microbiol 58:43–73
Holliger C, Wohlfarth G, Diekert G (1999) FEMS Microbiol Rev 22:383–398
Pera-Titus M, García-Molina V, Baños MA, Giménez J, Esplugas S (2004) Appl Catal B Environ 47:219–256
Osborne RL, Raner GM, Hager LP, Dawson JH (2006) J Am Chem Soc 128:1036–1037
Fetzner S (1998) Appl Microbiol Biotechnol 50:633–657
Carter WPL, Luo D, Malkina IL (1997) Atmos Environ 31:1425–1439
Plewa MJ, Wagner ED, Jazwierska P, Richardson SD, Chen PH, McKague AB (2004) Environ Sci Technol 38:62–68
Castro CA, Wade RS, Belser NO (1983) J Agric Food Chem 31:1184–1187
Sparks SE, Quistad GB, Casida JE (1997) Chem Res Toxicol 10:1001–1007
Zheng W, Yates SR, Papiernik SK, Guo M (2006) J Agric Food Chem 54:2280–2287
Becke AD (1993) J Chem Phys 98:1372–1377
Perdew JP, Ernzerhof M, Burke K (1996) J Chem Phys 105:9982–9985
Curtiss LA, Raghavachari K, Pople JA (1993) J Chem Phys 98:1293–1298
Curtiss LA, Raghavachari K, Redfern PC, Rassolov V, Pople JA (1998) J Chem Phys 109:7764–7776
Curtiss LA, Redfern PC, Raghavachari K (2007) J Chem Phys 126:084108
Tomasi J, Mennucci B, Cammi R (2005) Chem Rev 105:2999–3093
Gaussian 03, Revision B.03, Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr., Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian, Inc., Wallingford
Barss WM (1957) Chem Phys 27:1260–1266
Wade EA, Reak KE, Parsons BF, Clemes TP, Singmaster KA (2002) Chem Phys Lett 365:473–479
Haszeldine RN (1953) J Chem Soc 2525–2527
Agarwal UP, Barnes AJ, Orville-Thomas WJ (1985) Can J Chem 63:1705–1707
Ornellas FR (2000) Theor Chem Acc 103:469–476
Resende SM, Ornellas FR (2000) J Phys Chem A 104:11934–11939
Rayne S, Forest K (2010) Theochem 953:47–48
Jalkanen KJ, Degtyarenko IM, Nieminen RM, Cao X, Nafie LA, Zhu F, Barron LD (2008) Theor Chem Acc 119:191–210
Acknowledgments
This work was funded through project Comisión Sectorial de Investigación Científica (CSIC) 365/2006 (Universidad de la República, Uruguay). We also thank Programa de Desarrollo de las Ciencias Básicas (PEDECIBA) and Agencia Nacional de Investigación e Innovación (ANII) for partial financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Akira Imamura on the occasion of his 77th birthday and published as part of the Imamura Festschrift Issue.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ventura, O.N., Saenz-Méndez, P. & Bottinelli, F. Computational study on the partial dechlorination of the pesticide chloropicrin by sulfur species. Theor Chem Acc 130, 955–963 (2011). https://doi.org/10.1007/s00214-011-1057-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-011-1057-y