Abstract
The intramolecular proton transfer process and optical properties of a series of derivatives of 2-(2-hydroxyphenyl)-5-phenyl-1,3,4-oxadiazole (HOXD) have been studied. The effects of electron-donating and -withdrawing substituents on the intramolecular proton transfer and optical properties in the S0 and S1 states have been investigated to find out the relationships between them and the Hammett substituent constants (δ p). The statistically valid linear correlations are observed between the δ p and the ionization potential (IP), electron affinity (EA), the relative energies (ΔE) and the direct energy barriers (ΔE d) of the intramolecular proton transfer reactions, and the optical properties of HOXD and its substituted derivatives. The λ abs, λ fl, and λ ph of the electron-withdrawing substituted derivatives have bathochromic shifts, while the corresponding values of the electron-donating substituted derivatives show hypsochromic shifts compared with the parent compound HOXD. A successful tuning in the emission color was achieved: the emission wavelength was substituent δ p dependent, providing a powerful strategy for predicting the optical properties of novel electroluminophores.
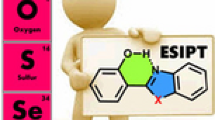
Graphical Abstract
The effects of substituents on the intramolecular proton transfer and optical properties for 2-(2-hydroxyphenyl)-5-phenyl-1,3,4-oxadiazole and its diverse derivatives have been theoretically investigated with the aim to get the relationships between the ESIPT reactions, optical properties, and the Hammett substituent constants (δ p).







Similar content being viewed by others
References
Chen C-L, Lin C-W, Hsieh C-C, Lai C-H, Lee G-H, Wang C-C, Chou P-T (2009) J Phys Chem A 113:205
Park S, Seo J, Kim SH, Park SY (2008) Adv Funct Mater 18:726
Nosenko Y, Wiosna-Sałyga G, Kunitski M, Petkova I, Singh A, Buma WJ, Thummel RP, Brutschy B, Waluk J (2008) Angew Chem Int Ed Engl 47:6037
Migani A, Bearpark MJ, Olivucci M, Robb MA (2007) J Am Chem Soc 129:3703
Shynkar VV, Klymchenko AS, Kunzelmann C, Duportail G, Muller CD, Demchenko AP, Freyssinet J-M, Mely Y (2007) J Am Chem Soc 129:2187
Wu Y, Peng X, Fan J, Gao S, Tian M, Zhao J, Sun S (2007) J Org Chem 72:62
Henary MM, Wu Y, Cody J, Sumalekshmy S, Li J, Mandal S, Fahrni CJ (2007) J Org Chem 72:4784
Qian Y, Li S, Zhang G, Wang Q, Wang S, Xu H, Li C, Li Y, Yang G (2007) J Phys Chem B 111:5861
Sakota K, Inoue N, Komoto Y, Sekiya H (2007) J Phys Chem A 111:4596
Strandjord AJG, Courtney SH, Friedrich DM, Barbara PF (1983) J Phys Chem 87:1125
McMorrow D, Kasha M (1984) J Phys Chem 88:2235
Schwartz BJ, Peteanu LA, Harris CB (1992) J Phys Chem 96:3591
Sengupta PK, Kasha M (1979) Chem Phys Lett 68:382
Das K, Sarkar N, Ghosh AK, Majumdar D, Nath DN, Bhattacharyya K (1994) J Phys Chem 98:9126
Douhal A, Lahmani F, Zewail AH (1996) Chem Phys 207:477
Douhal A, Lahmani F, Zehnacker-Rentien A (1993) Chem Phys 178:493
Sytnik A, Kasha M (1994) Proc Natl Acad Sci USA 91:8627
Liang F, Wang L, Ma D, Jing X, Wang F (2002) Appl Phys Lett 81:4
Tong H, Zhou G, Wang L, Jing X, Wang F, Zhang J (2003) Tetrahedron Lett 44:131
Brunner K, Dijken AV, Börner H, Bastiaansen JJAM, Kiggen NMM, Langeveld BMW (2004) J Am Chem Soc 126:6035
Yi Y, Zhu L, Shuai Z (2008) Macromol Theory Simul 17:12
Parthenopoulos DA, McMorrow D, Kasha M (1991) J Phys Chem 95:2668
Ma D, Liang F, Wang L, Lee ST, Hung L (2002) Chem Phys Lett 358:24
Keck J, Kramer HEA, Port H, Hirsch T, Fischer P, Rytz G (1996) J Phys Chem 100:14468
Douhal A, Sastre R (1994) Chem Phys Lett 219:91
Photochromism: memories and switches (2000) Special issue of Chem Rev 100
Doroshenko AO, Posokhov EA, Verezubova AA, Ptyagina LM (2000) J Phys Org Chem 13:253
Doroshenko AO, Posokhov EA, Verezubova AA, Ptyagina LM, Skripkina VT, Shershukov VM (2002) Photochem Photobiol Sci 1:92
Gaenko AV, Devarajan A, Tselinskii IV, Ryde U (2006) J Phys Chem A 110:7935
Tamoto N, Adachi C, Nagai K (1997) Chem Mater 9:1077
Uchida M, Adachi C, Koyama T, Taniguchi Y (1999) J Appl Phys 86:1680
Wang JF, Jabbour GE, Mash EA, Anderson J, Zhang Y, Lee PA, Armstrong NR, Peyghambarian N, Kippelen B (1999) Adv Mater 11:1266
Kido J, Hayase H, Hongawa K, Nagai K, Okuyama K (1994) Appl Phys Lett 65:2124
Antoniadis H, Inbasekaran M, Woo EP (1998) Appl Phys Lett 73:3055
Hu Y, Zhang Y, Liang F, Wang L, Ma D, Jing X (2003) Synth Met 137:1123
Yang Z, Yang S, Zhang J (2007) J Phys Chem A 111:6354
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JAJ, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, revision C.02. Gaussian Inc, Wallingford
Foresman JB, Head-Gordon M, Pople JA, Frisch MJ (1992) J Phys Chem 96:135
Dreuw A, Head-Gordon M (2005) Chem Rev 105:4009
Barone V, Polimeno A (2007) Chem Soc Rev 36:1724
Jacquemin D, Perpete EA, Ciofini I, Adamo C (2009) Acc Chem Res 42:326
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Sun M, Niu B, Zhang J (2008) J Mol Struct: Theochem 862:85
Sun M, Niu B, Zhang J (2008) Theor Chem Acc 119:489
Gahungu G, Zhang J (2005) J Mol Struct: Theochem 755:19
Li Z, Zhang J (2006) Chem Phys 331:159
Domingo LR, Picher MT, Zaragozá RJ (1998) J Org Chem 63:9183
Shukla MK, Leszczynski J (2005) Int J Quantum Chem 105:387
Halls MD, Schlegel HB (2001) Chem Mater 13:2632
Zhang J, Frenking G (2004) J Phys Chem A 108:10296
Zhang J, Frenking G (2004) Chem Phys Lett 394:120
Gahungu G, Zhang J (2005) J Phys Chem B 109:17762
Gahungu G, Zhang J (2005) Chem Phys Lett 410:302
Hu B, Gahungu G, Zhang J (2007) J Phys Chem A 111:4965
Ahlrichs R, Bär M, Häser M, Horn H, Kälmel C (1989) Chem Phys Lett 162:165
Santos L, Vargas A, Moreno M, Manzano BR, Lluch JM, Douhal A (2004) J Phys Chem A 108:9331
Hammett LP (1937) J Am Chem Soc 59:96
Hansch C, Leo A, Taft RW (1991) Chem Rev 91:165
Wheeler SE, Houk KN (2008) J Am Chem Soc 130:10854
Romero FA, Hwang I, Boger DL (2006) J Am Chem Soc 128:14004
Zdilla MJ, Dexheimer JL, Abu-Omar MM (2007) J Am Chem Soc 129:11505
Pohl R, Anzenbacher P Jr (2003) Org Lett 5:2769
Pohl R, Montes VA, Shinar J, Anzenbacher P Jr (2004) J Org Chem 69:1723
Pan Y, Fu Y, Liu S, Yu H, Gao Y, Guo Q, Yu S (2006) J Phys Chem A 110:7316
Zhang X, Wang C-J, Liu L-H, Jiang Y-B (2002) J Phys Chem B 106:12432
Fayet G, Joubert L, Rotureau P, Adamo C (2008) J Phys Chem A 112:4054
Cleij TJ, King JK, Jenneskens LW (2000) Macromolecules 33:89
Jacquemin D, Champagne B, André J-M (1996) Synth Met 80:205
Sugiyama K, Yoshimura D, Miyamae T, Miyazaki T, Ishii H, Ouchi Y, Seki K (1998) J Appl Phys 83:4928
Jena NR, Mishra PC (2005) J Phys Chem B 109:14205
Kasha M, Heldt J, Gormin D (1995) J Phys Chem 99:7281
Cossi M, Rega N, Scalmani G, Barone V (2003) J Comput Chem 24:669
Cossi M, Barone V (2001) J Chem Phys 115:4708
Aquino AJA, Lischka H, Hattig C (2005) J Phys Chem A 109:3201
Joachain CJ, Brandsen BH (1983) Physics of atoms and molecules. Longman, London
Köse ME, Mitchell WJ, Kopidakis N, Chang CH, Shaheen SE, Kim K, Rumbles G (2007) J Am Chem Soc 129:14257
Acknowledgments
Financial supports from the NSFC (Nos. 50873020, 20773022), the NCET-06-0321, and the NENU-STB-07-007 are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jin, R., Zhang, J. Substituent effects in the tuning of excited-state intramolecular proton transfer and optical properties of the derivatives of 2-(2-hydroxyphenyl)-5-phenyl-1,3,4-oxadiazole. Theor Chem Acc 124, 331–338 (2009). https://doi.org/10.1007/s00214-009-0613-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-009-0613-1