Abstract
Rationale
Ropanicant (SUVN-911) (3-(6-Chloropyridine-3-yloxymethyl)-2-azabicyclo (3.1.0) hexane hydrochloride) is a novel α4β2 nicotinic acetylcholine receptor (nAChR) antagonist being developed for the treatment of depressive disorders.
Objectives
Pharmacological and neurochemical characterization of Ropanicant to support a potential molecule for the treatment of depressive disorders.
Methods
Ropanicant was assessed for antidepressant-like activity using the rat forced swimming test (FST) and differential reinforcement of low rate −72 s (DRL-72 s). Alleviation of anhedonia was assessed in chronic mild stress model using sucrose preference test. To understand the mechanism of action, serotonin levels, ionized calcium-binding adaptor molecule 1 (Iba1), and brain-derived neurotrophic factor (BDNF) were determined. The onset of antidepressant-like activity was determined using the reduction in submissive behavior assay. The effects on cognition and sexual functions were assessed using the object recognition task and sexual dysfunction assay respectively. Interaction of Ropanicant, TC-5214, and methyllycaconitine (MLA) with citalopram was investigated individually in mice FST.
Results
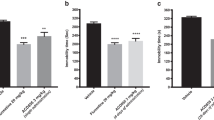
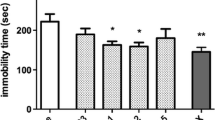
Ropanicant exhibited antidepressant like properties in the FST and DRL-72 s. A significant reduction in anhedonia was observed in the sucrose preference test. Oral administration of Ropanicant produced a significant increase in serotonin and BDNF levels, with a reduction in the Iba1 activity. The onset of antidepressant like effect with Ropanicant was within a week of treatment, and was devoid of cognitive dulling and sexual dysfunction. While Ropanicant potentiated the effect of citalopram in FST, such an effect was not observed with MLA or TC-5214.
Conclusions
Preclinical studies with Ropanicant support the likelihood of its therapeutic utility in the treatment of depressive disorders.







Similar content being viewed by others
References
Aboul-Fotouh S (2015) Behavioral effects of nicotinic antagonist mecamylamine in a rat model of depression: prefrontal cortex level of BDNF protein and monoaminergic neurotransmitters. Psychopharmacology 232(6):1095–1105
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Publishing, Arlington, VA
Andreasen JT, Redrobe JP (2009) Nicotine, but not mecamylamine, enhances antidepressant-like effects of citalopram and reboxetine in the mouse forced swim and tail suspension tests. Behav Brain Res 197:150–156
Awtry TL, Frank JG, Werling LL (2006) In vitro regulation of serotonin transporter activity by protein kinase A and nicotinic acetylcholine receptors in the prefrontal cortex of rats. Synapse 59(6):342–349
Bahr R, Lopez A, Rey JA (2019) Intranasal Esketamine (SpravatoTM) for use in treatment-resistant depression in conjunction with an oral antidepressant. Pharmacy and Therapeutics 44:340
Berger M, Riemann D, Höchli D, Spiegel R (1989) The cholinergic rapid eye movement sleep induction test with RS-86: State or trait marker of depression? Arch Gen Psychiatry 46:421–428
Björkholm C, Monteggia LM (2016) BDNF—a key transducer of antidepressant effects. Neuropharmacology 102:72–79
Blier P (2016) Neurobiology of depression and mechanism of action of depression treatments. J Clin Psychiatry 23:77(e319)
Caldarone BJ, Harrist A, Cleary MA, Beech RD, King SL, Picciotto MR (2004) High-affinity nicotinic acetylcholine receptors are required for antidepressant effects of amitriptyline on behavior and hippocampal cell proliferation. Biol Psychiat 56:657–664
Carlini VP, Poretti MB, Rask-Andersen M, Chavan RA, Ponzio MF, Sawant RS, de Barioglio SR, Schiöth HB, de Cuneo MF (2012) Differential effects of fluoxetine and venlafaxine on memory recognition: possible mechanisms of action. Prog Neuropsychopharmacol Biol Psychiatry 38:159–167
Chen C, Wang L, Rong X, Wang W, Wang X (2015) Effects of fluoxetine on protein expression of potassium ion channels in the brain of chronic mild stress rats. Acta Pharmaceutica Sinica B 5:55–61
Clayton AH, Croft HA, Handiwala L (2014) Antidepressants and sexual dysfunction: mechanisms and clinical implications. Postgrad Med 126(2):91–99
Daly JW (2005) Nicotinic agonists, antagonists, and modulators from natural sources. Cell Mol Neurobiol 25:513–552
Dulawa SC, Janowsky DS (2019) Cholinergic regulation of mood: from basic and clinical studies to emerging therapeutics. Mol Psychiatry 24:694–709
Fryer JD, Lukas RJ (1999) Antidepressants noncompetitively inhibit nicotinic acetylcholine receptor function. J Neurochem 72:1117–1124
Goswami SK, Vishwanath M, Gangadarappa SK, Razdan R, Inamdar MN (2014) Efficacy of ellagic acid and sildenafil in diabetes-induced sexual dysfunction. Pharmacogn Mag 10(Suppl 3):S581
Janowsky D, Davis J, El-Yousef MK, Sekerke HJ (1972) A cholinergic-adrenergic hypothesis of mania and depression. The Lancet 300:632–635
Janowsky DS, El-Yousef MK, Davis JM (1974) Acetylcholine and depression. Psychosom Med 36:248–257
Jutkiewicz EM, Wood SK, Houshyar H, Hsin LW, Rice KC, Woods JH (2005) The effects of CRF antagonists, antalarmin, CP154, 526, LWH234, and R121919, in the forced swim test and on swim-induced increases in adrenocorticotropin in rats. Psychopharmacology 180(2):215–223
Higgins A, Nash M, Lynch AM (2010) Antidepressant-associated sexual dysfunction: impact, effects, and treatment. Drug, Healthcare and Patient Safety 2:141
Kenny PJ, File SE, Neal MJ (2000) Evidence for a complex influence of nicotinic acetylcholine receptors on hippocampal serotonin release. J Neurochem 75:2409–2414
Levin ED, Briggs SJ, Christopher NC, Rose JE (1993) Chronic nicotinic stimulation and blockade effects on working memory. Behav Pharmacol 4:179–182
Levin ED, Caldwell DP (2006) Low-dose mecamylamine improves learning of rats in the radial-arm maze repeated acquisition procedure. Neurobiol Learn Mem 86:117–122
Li X, Rainnie DG, McCarley RW, Greene RW (1998) Presynaptic nicotinic receptors facilitate monoaminergic transmission. J Neurosci 18:1904–1912
Lippello et al (2008) TC-5214 (S-(+)-Mecamylamine): A neuronal nicotinic receptor modulator with antidepressant activity. CNS Neurosci Ther 14:266–277
Lopez-Valdes HE, Garcia-Colunga J (2001) Antagonism of nicotinic acetylcholine receptors by inhibitors of monoamine uptake. Mol Psychiatry 6:511–519
Louis C, Llopis K, Danober L, Panayi F, Lestage P, Beracochea D (2019) New procognitive enhancers acting at the histamine H3 and AMPA receptors reverse natural forgetting in mice: comparisons with donepezil and memantine in the object recognition task. Behav Pharmacol 30:351–357
Lueptow LM (2017) Novel object recognition test for the investigation of learning and memory in mice. JoVE 126:e55718
Ma Z, Pearson E, Isgor C, Tao R (2006) Evidence of reuptake inhibition responsible for mecamylamine-evoked increases in extracellular serotonin. Brain Res 1073:321–324
Malatynska E, Goldenberg R, Shuck L et al (2002) Reduction of submissive behavior in rats: a test for antidepressant drug activity. Pharmacology 64:8–17
Malatynska E, Knapp RJ (2005) Dominant–submissive behavior as models of mania and depression. Neurosci Biobehav Rev 29:715–737
Malatynska E, Pinhasov A, Creighton CJ, Crooke JJ, Reitz AB, Brenneman DE, Lubomirski MS (2007) Assessing activity onset time and efficacy for clinically effective antidepressant and antimanic drugs in animal models based on dominant–submissive relationships. Neurosci Biobehav Rev 31:904–919
Marek GJ, Salek AA (2020) Extending the specificity of DRL 72-s behavior for screening antidepressant-like effects of glutamatergic clinically validated anxiolytic or antidepressant drugs in rats. J Pharmacol Exp Ther 374:200–210
Mineur YS, Obayemi A, Wigestrand MB (2013) Cholinergic signalling in the hippocampus regulates social stress resilience and anxiety-and depression-like behavior. Proc Natl Acad Sci 110:3573–3578
Mineur YS, Mose TN, Blakeman S, Picciotto MR (2018) Hippocampal α7 nicotinic ACh receptors contribute to modulation of depression‐like behaviour in C57BL/6J mice. Br J Pharmacol 175:1903–14
Moser PC, Bergis OE, Jegham S et al (2002) SL65.0155, A novel 5-hydroxytryptamine4 receptor partial agonist with potent cognition-enhancing properties. J Pharmacol Exp Ther 302:731–41
Montgomery SA (1997) Clinical measures of rapid onset of action. Eur Psychiatry 12:295s-300s
Nickell JR, Grinevich VP, Siripurapu KB, Smith AM, Dwoskin LP (2013) Potential therapeutic uses of mecamylamine and its stereoisomers. Pharmacol Biochem Behav 108:28–43
Nirogi R, Mohammed AR, Shinde AK et al (2020) Discovery and development of 3-(6-Chloropyridine-3-yloxymethyl)-2-azabicyclo [3.1. 0] hexane hydrochloride (SUVN-911): A novel, potent, selective, and orally active neuronal nicotinic acetylcholine α4β2 receptor antagonist for the treatment of depression. J Med Chem 63:2833–2853
Nirogi R, Abraham R, Benade V et al (2019) SUVN-502, a novel, potent, pure, and orally active 5-HT6 receptor antagonist: pharmacological, behavioral, and neurochemical characterization. Behav Pharmacol 30:16–35
O’Donnell JM, Marek GJ, Seiden LS (2005) Antidepressant effects assessed using behavior maintained under a differential-reinforcement-of-low-rate (DRL) operant schedule. Neurosci Biobehav Rev 29:785–798
Perini G, Ramusino MC, Sinforiani E, Bernini S, Petrachi R, Costa A (2019) Cognitive impairment in depression: recent advances and novel treatments. Neuropsychiatr Dis Treat 15:1249
Philip NS, Carpenter LL, Tyrka AR, Price LH (2010) Nicotinic acetylcholine receptors and depression: a review of the preclinical and clinical literature. Psychopharmacology 212:1–2
Philip NS, Carpenter LL, Tyrka AR (2012) The nicotinic acetylcholine receptor as a target for antidepressant drug development. The Scientific World Journal 104105–12
Pitsikas N, Markou A (2014) The metabotropic glutamate 2/3 receptor agonist LY379268 counteracted ketamine-and apomorphine-induced performance deficits in the object recognition task, but not object location task, in rats. Neuropharmacology 85:27–35
Popik P, Kozela E, Krawczyk M (2003) Nicotine and nicotinic receptor antagonists potentiate the antidepressant - like effects of imipramine and citalopram. Br J Pharmacol 139:1196–1202
Riemann D, Hohagen F, Krieger S et al (1994) Cholinergic REM induction test: muscarinic supersensitivity underlies polysomnographic findings in both depression and schizophrenia. J Psychiatr Res 28:195–210
Salin-Pascual RJ (2002) Relationship between Mood Improvement and Sleep Changes with Acute Nicotine Administration in Non-Smoking Major Depressed Patients 54:36–40
Saricicek A, Esterlis I, Maloney KH, Mineur YS et al (2012) Persistent β2*-nicotinic acetylcholinergic receptor dysfunction in major depressive disorder. Am J Psychiatry 169:851–859
Sarter M, Parikh V, Howe WM (2009) nAChR agonist-induced cognition enhancement: integration of cognitive and neuronal mechanisms. Biochem Pharmacol 78:658–667
Sayyah M, Eslami K, AlaiShehni S, Kouti L (2016) Cognitive function before and during treatment with selective serotonin reuptake inhibitors in patients with depression or obsessive-compulsive disorder. Psychiatry J 2016:5480391
Schmitt JA, Wingen M, Ramaekers JG, Evers EA, Riedel WJ (2006) Serotonin and human cognitive performance. Curr Pharm Des 12:2473–2486
Schmidtner AK, Slattery DA, Gläsner J, Hiergeist A, Gryksa K et al (2019) Minocycline alters behavior, microglia and the gut microbiome in a trait-anxiety-dependent manner. Transl Psychiatry 9:1–2
Serchov T, Schwarz I, Theiss A et al (2020) Enhanced adenosine A1 receptor and Homer1a expression in hippocampus modulates the resilience to stress-induced depression-like behavior. Neuropharmacology 162:107834
Serretti A, Calati R, Goracci A, Di Simplicio M, Castrogiovanni P, De Ronchi D (2010) Antidepressants in healthy subjects: what are the psychotropic/psychological effects? Eur Neuropsychopharmacol 20:433–453
Shytle RD, Silver AA, Sheehan KH, Sheehan DV, Sanberg PR (2002) Neuronal nicotinic receptor inhibition for treating mood disorders: preliminary controlled evidence with mecamylamine. Depress Anxiety 16:89–92
Slattery DA, Cryan JF (2012) Using the rat forced swim test to assess antidepressant-like activity in rodents. Nat Protoc 7:1009–1014
Spring B, Gelenberg AJ, Garvin R, Thompson S (1992) Amitriptyline, clovoxamine and cognitive function: a placebo-controlled comparison in depressed outpatients. Psychopharmacology 108:327–332
Sullivan JP, Bannon AW (1996) Epibatidine: pharmacological properties of a novel nicotinic acetylcholine receptor agonist and analgesic agent. CNS Drug Rev 2:21–39
Terry AV Jr, Buccafusco JJ, Prendergast MA (1999) Dose-specific improvements in memory-related task performance by rats and aged monkeys administered the nicotinic-cholinergic antagonist mecamylamine. Drug Dev Res 47:127–136
Tucci SA, Genn RF, File SE (2003) Methyllycaconitine (MLA) blocks the nicotine evoked anxiogenic effect and 5-HT release in the dorsal hippocampus: possible role of a7 receptors. Neuropharmacology 44:367–373
Tummala R, Desai D, Szamosi J et al (2015) Safety and tolerability of dexmecamylamine (TC-5214) adjunct to ongoing antidepressant therapy in patients with major depressive disorder and an inadequate response to antidepressant therapy: results of a long-term study. J Clin Psychopharmacol 35:77–81
Vieta E, Thase ME, Naber D, D’Souza B, Rancans E et al (2014) Efficacy and tolerability of flexibly-dosed adjunct TC-5214 (dexmecamylamine) in patients with major depressive disorder and inadequate response to prior antidepressant. Eur Neuropsychopharmacol 24:564–574
Wang YL, Han QQ, Gong WQ et al (2018) Microglial activation mediates chronic mild stress-induced depressive-and anxiety-like behavior in adult rats. J Neuroinflammation 15:21
Walsh RN, Cummins RA (1976) The open-field test: a critical review. Psychol Bull 83:482
Willner P (2005) Chronic mild stress (CMS) revisited: consistency and behavioural-neurobiological concordance in the effects of CMS. Neuropsychobiology 52:90–110
Willner P (1997) Validity, reliability and utility of the chronic mild stress model of depression: a 10-year review and evaluation. Psychopharmacology 134:319–329
Wilkerson JL, Deba F, Crowley ML, Hamouda AK, McMahon LR (2020) Advances in the In vitro and In vivo pharmacology of Alpha4beta2 nicotinic receptor positive allosteric modulators. Neuropharmacology 168:108008
Yirmiya R, Rimmerman N, Reshef R (2015) Depression as a microglial disease. Trends Neurosci 38:637–658
Yu LF, Zhang HK, Caldarone BJ et al (2014) Recent developments in novel antidepressants targeting α4β2-nicotinic acetylcholine receptors: Miniperspective. J Med Chem 57(20):8204–8223
Acknowledgements
All experiments described in this research were funded by Suven Life Sciences Ltd.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors are employees of Suven Life Sciences Ltd.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nirogi, R., Abraham, R., Jayarajan, P. et al. Ropanicant (SUVN-911), an α4β2 nicotinic acetylcholine receptor antagonist intended for the treatment of depressive disorders: pharmacological, behavioral, and neurochemical characterization. Psychopharmacology 239, 2215–2232 (2022). https://doi.org/10.1007/s00213-022-06108-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-022-06108-6