Abstract
Rationale
The α4β2 nicotinic acetylcholine receptor partial agonist varenicline has been reported to reduce drinking among both heavy-drinking smokers and primary alcoholics, and this effect may be related to varenicline-mediated reduction of alcohol craving. Among smokers, varenicline has been reported to modulate cigarette cue-elicited brain activation in several reward-related areas.
Objectives
This pilot study tested varenicline’s effects on drinking, alcohol craving, and alcohol cue-elicited activation of reward-related brain areas among non-treatment-seeking alcohol-dependent individuals.
Methods
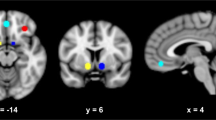
Thirty-five such individuals (mean age = 30, 57 % male, 76 % heavy drinking days in the past month, 15 smokers) were randomized to either varenicline (titrated to 2 mg) or placebo for 14 days, and were administered an alcohol cue reactivity fMRI task on day 14. A priori regions of interest (ROIs) were bilateral and medial orbitofrontal cortex (OFC), right ventral striatum (VS), and medial prefrontal cortex (mPFC).
Results
Despite good medication adherence, varenicline did not reduce heavy drinking days or other drinking parameters. It did, however, increase self-reported control over alcohol-related thoughts and reduced cue-elicited activation bilaterally in the OFC, but not in other brain areas.
Conclusions
These data indicate that varenicline reduces alcohol craving and some of the neural substrates of alcohol cue reactivity. However, varenicline effects on drinking mediated by cue-elicited brain activation and craving might be best observed among treatment-seekers motivated to reduce their alcohol consumption.



Similar content being viewed by others
References
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders (4th edn., text rev.). Author, Washington
Anton RF, Moak DH, Latham PK (1996) The obsessive compulsive drinking scale: a new method of assessing outcome in alcoholism treatment studies. Arch Gen Psychiatry 53:225–231
Childs E, Roche DJ, King AC, de Wit H (2012) Varenicline potentiates alcohol-induced negative subjective responses and offsets impaired eye movements. Alcohol Clin Exp Res 36:906–914
Claus ED, Ewing SW, Filbey FM, Sabbineni A, Hutchison KE (2011) Identifying neurobiological phenotypes associated with alcohol use disorder severity. Neuropsychopharmacology 36:2086–2096
Di Chiara G (2000) Role of dopamine in the behavioural actions of nicotine related to addiction. Eur J Pharmacol 393:295–314
Ericson M, Blomqvist O, Engel JA, Soderpalm B (1998) Voluntary ethanol intake in the rat and the associated accumbal dopamine overflow are blocked by ventral tegmental mecamylamine. Eur J Pharmacol 358:189–196
Filbey FM, Claus E, Audette AR, Niculescu M, Banich MT, Tanabe J et al (2008) Exposure to the taste of alcohol elicits activation of the mesocorticolimbic neurocircuitry. Neuropsychopharmacology 33:1391–1401
First MB, Spitzer RL, Gibbon M, Williams JBW (2002) Structured clinical interview for DSM-IV-TR axis I disorders, research version, non-patient edition. Biometrics Research, New York State Psychiatric Institute, New York
Franklin T, Wang Z, Suh JJ, Hazan R, Cruz J, Li Y et al (2011) Effects of varenicline on smoking cue-triggered neural and craving responses. Arch Gen Psychiatry 68:516–526
Fucito LM, Toll BA, Wu R, Romano DM, Tek E, O’Malley SS (2011) A preliminary investigation of varenicline for heavy drinking smokers. Psychopharmacology (Berl) 215:655–663
Hermann D, Smolka MN, Wrase J, Klein S, Nikitopoulos J, Georgi A, Braus DF, Flor H, Mann K, Heinz A (2006) Blockade of cue-induced brain activation of abstinent alcoholics by a single administration of amisulpride as measured with fMRI. Alcohol Clin Exp Res 30:1349–1354
Kamens HM, Andersen J, Picciotto MR (2010a) The nicotinic acetylcholine receptor partial agonist varenicline increases the ataxic and sedative-hypnotic effects of acute ethanol administration in C57BL/6J mice. Alcohol Clin Exp Res 34:2053–2060
Kamens HM, Andersen J, Picciotto MR (2010b) Modulation of ethanol consumption by genetic and pharmacological manipulation of nicotinic acetylcholine receptors in mice. Psychopharmacology (Berl) 208:613–626
Koob GF, Volkow ND (2010) Neurocircuitry of addiction. Neuropsychopharmacology 35:217–238
Kringelbach ML, Rolls ET (2004) The functional neuroanatomy of the human orbitofrontal cortex: evidence from neuroimaging and neuropsychology. Prog Neurobiol 72:341–372
Levine J, Schooler NR (1986) SAFTEE: a technique for the systematic assessment of side effects in clinical trials. Psychopharmacol Bull 22:343–381
Litten RZ, Ryan ML, Fertig JB, Falk DE, Johnson B, Dunn KE et al (2013) A double-blind, placebo-controlled trial assessing the efficacy of varenicline tartrate for alcohol dependence. J Addict Med 7(4):277–286
Lotfipour S, Mandelkern M, Alvarez-Estrada M, Brody AL (2012) A single administration of low-dose varenicline saturates alpha4beta2* nicotinic acetylcholine receptors in the human brain. Neuropsychopharmacology 37:1738–1748
Loughead J, Ray R, Wileyto EP, Ruparel K, Sanborn P, Siegel S et al (2010) Effects of the alpha4beta2 partial agonist varenicline on brain activity and working memory in abstinent smokers. Biol Psychiatry 67:715–721
Lukas SE, Lowen SB, Lindsey KP, Conn N, Tartarini W, Rodolico J et al (2013) Extended-release naltrexone (XR-NTX) attenuates brain responses to alcohol cues in alcohol-dependent volunteers: a bold FMRI study. NeuroImage 78:176–185
McKee SA, Harrison EL, O’Malley SS, Krishnan-Sarin S, Shi J, Tetrault JM et al (2009) Varenicline reduces alcohol self-administration in heavy-drinking smokers. Biol Psychiatry 66:185–190
Mitchell JM, Teague CH, Kayser AS, Bartlett SE, Fields HL (2012) Varenicline decreases alcohol consumption in heavy-drinking smokers. Psychopharmacology (Berl) 223:299–306
Mocking RJ, Patrick Pflanz C, Pringle A, Parsons E, McTavish SF, Cowen PJ et al (2013) Effects of a short-term varenicline administration on emotional and cognitive processing in healthy, non-smoking adults: a randomized, double-blind, study. Neuropsychopharmacology 38:476–484
Myrick H, Anton RF, Li X, Henderson S, Drobes D, Voronin K et al (2004) Differential brain activity in alcoholics and social drinkers to alcohol cues: relationship to craving. Neuropsychopharmacology 29:393–402
Myrick H, Anton RF, Li X, Henderson S, Randall PK, Voronin K (2008) Effect of naltrexone and ondansetron on alcohol cue-induced activation of the ventral striatum in alcohol-dependent people. Arch Gen Psychiatry 65:466–475
Myrick H, Li X, Randall PK, Henderson S, Voronin K, Anton RF (2010) The effect of aripiprazole on cue-induced brain activation and drinking parameters in alcoholics. J Clin Psychopharmacol 30:365–372
Plebani JG, Lynch KG, Rennert L, Pettinati HM, O’Brien CP, Kampman KM (2013) Results from a pilot clinical trial of varenicline for the treatment of alcohol dependence. Drug Alcohol Depend 133(2):754–758
Ray LA, Lunny K, Bujarski S, Moallem N, Krull JL, Miotto K (2013) The effects of varenicline on stress-induced and cue-induced craving for cigarettes. Drug Alcohol Depend 131:136–142
Roberts JS, Anton RF, Latham PK, Moak DH (1999) Factor structure and predictive validity of the Obsessive Compulsive Drinking Scale. Alcohol Clin Exp Res 23:1484–1491
Rollema H, Chambers LK, Coe JW, Glowa J, Hurst RS, Lebel LA et al (2007) Pharmacological profile of the alpha4beta2 nicotinic acetylcholine receptor partial agonist varenicline, an effective smoking cessation aid. Neuropharmacology 52:985–994
Sayette MA, Shiffman S, Tiffany ST, Niaura RS, Martin CS, Shadel WG (2000) The measurement of drug craving. Addiction 95(Suppl 2):S189–S210
Schacht JP, Anton RF, Randall PK, Li X, Henderson S, Myrick H (2011) Stability of fMRI striatal response to alcohol cues: a hierarchical linear modeling approach. NeuroImage 56:61–68
Schacht JP, Anton RF, Myrick H (2013a) Functional neuroimaging studies of alcohol cue reactivity: a quantitative meta-analysis and systematic review. Addict Biol 18:121–133
Schacht JP, Anton RF, Randall PK, Li X, Henderson S, Myrick H (2013b) Effects of a GABA-ergic medication combination and initial alcohol withdrawal severity on cue-elicited brain activation among treatment-seeking alcoholics. Psychopharmacology (Berl) 227(4):627–637
Schacht JP, Anton RF, Voronin KE, Randall PK, Li X, Henderson S et al (2013c) Interacting effects of naltrexone and OPRM1 and DAT1 variation on the neural response to alcohol cues. Neuropsychopharmacology 38:414–422
Skinner HA, Allen BA (1982) Alcohol dependence syndrome: measurement and validation. J Abnorm Psychol 91:199–209
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TE, Johansen-Berg H et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage 23(Suppl 1):S208–S219
Sobell LC, Sobell MB (1992) Timeline follow-back: a technique for assessing self-reported alcohol consumption. In: Allen JP, Litten RZ (eds) Measuring alcohol consumption: psychosocial and biochemical methods. Humana Press, Totowa, pp 41–72
Steensland P, Simms JA, Holgate J, Richards JK, Bartlett SE (2007) Varenicline, an alpha4beta2 nicotinic acetylcholine receptor partial agonist, selectively decreases ethanol consumption and seeking. Proc Natl Acad Sci U S A 104:12518–12523
Stritzke WG, Breiner MJ, Curtin JJ, Lang AR (2004) Assessment of substance cue reactivity: advances in reliability, specificity, and validity. Psychol Addict Behav 18:148–159
Sullivan JT, Sykora K, Schneiderman J, Naranjo CA, Sellers EM (1989) Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict 84:1353–1357
Tizabi Y, Copeland RL Jr, Louis VA, Taylor RE (2002) Effects of combined systemic alcohol and central nicotine administration into ventral tegmental area on dopamine release in the nucleus accumbens. Alcohol Clin Exp Res 26:394–399
Volkow ND, Wang G-J, Telang F, Fowler JS, Logan J, Jayne M et al (2007) Profound decreases in dopamine release in striatum in detoxified alcoholics: possible orbitofrontal involvement. J Neurosci 27:12700–12706
Volkow ND, Wang GJ, Fowler JS, Tomasi D, Telang F (2011) Addiction: beyond dopamine reward circuitry. Proc Natl Acad Sci U S A 108:15037–15042
Wiers RW, Eberl C, Rinck M, Becker ES, Lindenmeyer J (2011) Retraining automatic action tendencies changes alcoholic patients’ approach bias for alcohol and improves treatment outcome. Psychol Sci 22:490–497
Wouda JA, Riga D, De Vries W, Stegeman M, van Mourik Y, Schetters D et al (2011) Varenicline attenuates cue-induced relapse to alcohol, but not nicotine seeking, while reducing inhibitory response control. Psychopharmacology (Berl) 216:267–277
Acknowledgements
Portions of this work were presented at the 34th Annual Meeting of the Research Society on Alcoholism (June 2011, Atlanta) and the 52nd Annual Meeting of the American College on Neuropsychopharmacology (December 2013, Hollywood, FL). This research was supported by grants from the National Institute on Alcohol Abuse and Alcoholism (NIAAA), including the Charleston Alcohol Research Center (P50 AA010761). NIAAA had no involvement in the study design, in the collection, analysis, and interpretation of data, in the writing of the paper, or in the decision to submit for publication. Drs. Schacht and Anton are supported by K99 AA021419 and K05 AA017435, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schacht, J.P., Anton, R.F., Randall, P.K. et al. Varenicline effects on drinking, craving and neural reward processing among non-treatment-seeking alcohol-dependent individuals. Psychopharmacology 231, 3799–3807 (2014). https://doi.org/10.1007/s00213-014-3518-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3518-1