Abstract
Rationale
Nicotine infusions that are self-administered (contingent) or response-independent (noncontingent) increase lever pressing for a reinforcing nonpharmacological stimulus in rats, suggesting that in addition to primary reinforcement, nicotine self-administration may result from nicotine enhancing the reinforcement derived from nonnicotine stimuli.
Objectives
Based on our previous research, in this study, we tested the hypothesis that contingent and noncontingent nicotine would equally elevate responding for a moderately reinforcing visual stimulus, across a range of nicotine doses on both fixed ratio and progressive ratio reinforcement schedules.
Materials and methods
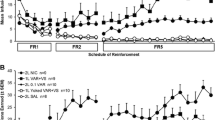
The rats lever pressed for a visual stimulus with contingent nicotine, noncontingent nicotine, or contingent saline. Separate groups responded for saline or nicotine without the visual stimulus. Three doses of nicotine (0.01, 0.03, and 0.09 mg/kg per infusion, free base) were tested in a between-groups design. After responding on an escalating fixed ratio reinforcement schedule, the rats were tested on a progressive ratio schedule.
Results
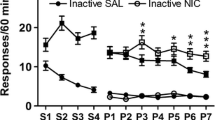
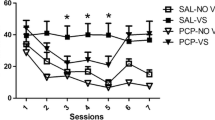
Compared to responding for the visual stimulus with saline, both contingent and noncontingent nicotine equally elevated lever pressing for the stimulus at each dose on fixed and progressive ratio schedules. In the absence of the stimulus, only the highest nicotine dose sustained self-administration.
Conclusions
The ability of noncontingent nicotine to elevate responding for a moderately reinforcing visual stimulus occurs across a range of doses, and both self-administered and noncontingent nicotine equally increase motivation to obtain the stimulus, as reflected by performance on a progressive ratio schedule. In the absence of a contingent stimulus, primary reinforcement from nicotine only supports self-administration at high nicotine doses in rats.




Similar content being viewed by others
References
Addy NA, Nakijama A, Levin ED (2003) Nicotinic mechanisms of memory: effects of acute local DHbetaE and MLA infusions in the basolateral amygdala. Brain Res Cogn Brain Res 16:51–57
Barr AM, Phillips AG (1999) Withdrawal following repeated exposure to d-amphetamine decreases responding for a sucrose solution as measured by a progressive ratio schedule of reinforcement. Psychopharmacology (Berl) 141:99–106
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, Hoffman A, Perkins KA, Sved AF (2002a) Environmental stimuli promote the acquisition of nicotine self-administration in rats. Psychopharmacology (Berl) 163:230–237
Caggiula AR, Donny EC, Chaudhri N, Perkins KA, Evans-Martin FF, Sved AF (2002b) Importance of nonpharmacological factors in nicotine self-administration. Physiol Behav 77:683–687
Chaudhri N (2005) Complex interactions between nicotine and nonpharmacological stimuli reveal a novel role for nicotine in reinforcement. Department of Neuroscience, University of Pittsburgh, Pittsburgh, PA
Chaudhri N, Caggiula AR, Donny EC, Booth S, Gharib MA, Craven LA, Allen SS, Sved AF, Perkins KA (2005) Sex differences in the contribution of nicotine and nonpharmacological stimuli to nicotine self-administration in rats. Psychopharmacology (Berl) 180:258–266
Chaudhri N, Caggiula AR, Donny EC, Palmatier MI, Liu X, Sved AF (2006) Complex interactions between nicotine and nonpharmacological stimuli reveal multiple roles for nicotine in reinforcement. Psychopharmacology (Berl) 184:353–366
Cohen C, Perrault G, Griebel G, Soubrie P (2005) Nicotine-associated cues maintain nicotine-seeking behavior in rats several weeks after nicotine withdrawal: reversal by the cannabinoid (CB1) receptor antagonist, rimonabant (SR141716). Neuropsychopharmacology 30:145–155
Corrigall WA, Coen KM (1989) Nicotine maintains robust self-administration in rats on a limited-access schedule. Psychopharmacology (Berl) 99:473–478
Dani JA, De Biasi M (2001) Cellular mechanisms of nicotine addiction. Pharmacol Biochem Behav 70:439–446
Dani JA, Ji D, Zhou FM (2001) Synaptic plasticity and nicotine addiction. Neuron 31:349–352
Depoortere RY, Li DH, Lane JD, Emmett-Oglesby MW (1993) Parameters of self-administration of cocaine in rats under a progressive-ratio schedule. Pharmacol Biochem Behav 45:539–548
Donny EC, Caggiula AR, Mielke MM, Jacobs KS, Rose C, Sved AF (1998) Acquisition of nicotine self-administration in rats: the effects of dose, feeding schedule, and drug contingency. Psychopharmacology (Berl) 136:83–90
Donny EC, Caggiula AR, Mielke MM, Booth S, Gharib MA, Hoffman A, Maldovan V, Shupenko C, McCallum SE (1999) Nicotine self-administration in rats on a progressive ratio schedule of reinforcement. Psychopharmacology (Berl) 147:135–142
Donny EC, Caggiula AR, Rowell PP, Gharib MA, Maldovan V, Booth S, Mielke MM, Hoffman A, McCallum S (2000) Nicotine self-administration in rats: estrous cycle effects, sex differences and nicotinic receptor binding. Psychopharmacology (Berl) 151:392–405
Donny EC, Chaudhri N, Caggiula AR, Evans-Martin FF, Booth S, Gharib MA, Clements LA, Sved AF (2003) Operant responding for a visual reinforcer in rats is enhanced by noncontingent nicotine: implications for nicotine self-administration and reinforcement. Psychopharmacology (Berl) 169:68–76
Grillner P, Svensson TH (2000) Nicotine-induced excitation of midbrain dopamine neurons in vitro involves ionotropic glutamate receptor activation. Synapse 38:1–9
Grilly DM, Simon BB, Levin ED (2000) Nicotine enhances stimulus detection performance of middle-and old-aged rats: a longitudinal study. Pharmacol Biochem Behav 65:665–670
Jones IW, Wonnacott S (2004) Precise localization of alpha7 nicotinic acetylcholine receptors on glutamatergic axon terminals in the rat ventral tegmental area. J Neurosci 24:11244–11252
Lehman RS (1995) Statistics in the behavioral sciences. A conceptual approach. Brooks/Cole
Levin ED (2002) Nicotinic receptor subtypes and cognitive function. J Neurobiol 53:633–640
Liu X, Caggiula AR, Palmatier MI, Donny EC, Chaudhri N, Sved AF (2005) Reinforcement-enhancing effect of nicotine depends on the reinforcement valence of nondrug stimulus. Society for Neuroscience abstract 1027.14
Mansvelder HD, McGehee DS (2000) Long-term potentiation of excitatory inputs to brain reward areas by nicotine. Neuron 27:349–357
Mansvelder HD, McGehee DS (2002) Cellular and synaptic mechanisms of nicotine addiction. J Neurobiol 53:606–617
Mansvelder HD, Keath JR, McGehee DS (2002) Synaptic mechanisms underlie nicotine-induced excitability of brain reward areas. Neuron 33:905–919
Mansvelder HD, De Rover M, McGehee DS, Brussaard AB (2003) Cholinergic modulation of dopaminergic reward areas: upstream and downstream targets of nicotine addiction. Eur J Pharmacol 480:117–123
Markou A, Weiss F, Gold LH, Caine SB, Schulteis G, Koob GF (1993) Animal models of drug craving. Psychopharmacology (Berl) 112:163–182
Matteson GL, Palmatier MI, Liu X, Chaudhri N, Caggiula AR, Donny EC, Sved AF (2006) The reinforcement enhancing effect of nicotine depends on the incentive value of a nonpharmacological reinforcer. The 12th annual meeting of the Society for Research on Nicotine and Tobacco, Orlando, FL, February 15–18
Nicola SM, Deadwyler SA (2000) Firing rate of nucleus accumbens neurons is dopamine-dependent and reflects the timing of cocaine-seeking behavior in rats on a progressive ratio schedule of reinforcement. J Neurosci 20:5526–5537
Olausson P, Jentsch JD, Taylor JR (2004) Nicotine enhances responding with conditioned reinforcement. Psychopharmacology (Berl) 171:173–178
Palmatier MI, Evans-Martin FF, Hoffman A, Caggiula AR, Chaudhri N, Donny E, Liu X, Booth S, Gharib MA, Craven LA, Sved AF (2006) Dissociating the primary reinforcing and reinforcement-enhancing effects of nicotine using a rat self-administration paradigm with concurrently available drug and environmental reinforcers. Psychopharmacology (Berl) 184:391–400
Phillips AG, Fibiger HC (1990) Role of reward and enhancement of conditioned reward in persistence of responding for cocaine. Behav Pharmacol 1:269–282
Pidoplichko VI, Noguchi J, Areola OO, Liang Y, Peterson J, Zhang T, Dani JA (2004) Nicotinic cholinergic synaptic mechanisms in the ventral tegmental area contribute to nicotine addiction. Learn Mem 11:60–69
Rasmussen T, Swedberg MD (1998) Reinforcing effects of nicotinic compounds: intravenous self-administration in drug-naive mice. Pharmacol Biochem Behav 60:567–573
Rezvani AH, Bushnell PJ, Levin ED (2002) Effects of nicotine and mecamylamine on choice accuracy in an operant visual signal detection task in female rats. Psychopharmacology (Berl) 164:369–375
Rice ME, Cragg SJ (2004) Nicotine amplifies reward-related dopamine signals in striatum. Nat Neurosci 7:583–584
Risner ME, Goldberg SR (1983) A comparison of nicotine and cocaine self-administration in the dog: fixed-ratio and progressive-ratio schedules of intravenous drug infusion. J Pharmacol Exp Ther 224:319–326
Robbins TW, Cador M, Taylor JR, Everitt BJ (1989) Limbic–striatal interactions in reward-related processes. Neurosci Biobehav Rev 13:155–162
Robbins TW (1976) Relationship between reward-enhancing and stereotypical effects of psychomotor stimulant drugs. Nature 264:57–59
Rose JE, Levin ED (1991) Inter-relationships between conditioned and primary reinforcement in the maintenance of cigarette smoking. Br J Addict 86:605–609
Schilstrom B, Nomikos GG, Nisell M, Hertel P, Svensson TH (1998) N-Methyl-d-aspartate receptor antagonism in the ventral tegmental area diminishes the systemic nicotine-induced dopamine release in the nucleus accumbens. Neuroscience 82:781–789
Shoaib M, Schindler CW, Goldberg SR (1997) Nicotine self-administration in rats: strain and nicotine pre-exposure effects on acquisition. Psychopharmacology (Berl) 129:35–43
Silva AJ (2003) Molecular and cellular cognitive studies of the role of synaptic plasticity in memory. J Neurobiol 54:224–237
Stafford D, LeSage MG, Glowa JR (1998) Progressive-ratio schedules of drug delivery in the analysis of drug self-administration: a review. Psychopharmacology (Berl) 139:169–184
Stewart J (1960) Reinforcing effects of light as a function of intensity and reinforcement schedule. J Comp Physiol Psychol 53:187–193
Stolerman IP, Mirza NR, Hahn B, Shoaib M (2000) Nicotine in an animal model of attention. Eur J Pharmacol 393:147–154
Taylor JR, Robbins TW (1984) Enhanced behavioural control by conditioned reinforcers following microinjections of d-amphetamine into the nucleus accumbens. Psychopharmacology (Berl) 84:405–412
Terry AV, Jr., Risbrough VB, Buccafusco JJ, Menzaghi F (2002) Effects of (+/−)-4-[[2-(1-methyl-2-pyrrolidinyl)ethyl]thio]phenol hydrochloride (SIB-1553A), a selective ligand for nicotinic acetylcholine receptors, in tests of visual attention and distractibility in rats and monkeys. J Pharmacol Exp Ther 301:284–292
USDHHS (1988) Nicotine addiction: a report of the surgeon general. US Department of Health and Human Services, Office of the Assistant Secretary for Health, Office on Smoking and Health, Rockville, MD
Wise RA (1987) The role of reward pathways in the development of drug dependence. Pharmacol Ther 35:227–263
Acknowledgements
“Principles of laboratory animal care” (NIH No. 85-23, revised 1985) were followed throughout all experiments. The University of Pittsburgh Institutional Animal Care and Use Committee assurance number A3187-01 approved this research. The preparation of this manuscript was supported by National Institute on Drug Abuse research grants DA-010464 and DA-012655 and by a Howard Hughes pre-doctoral research fellowship awarded to N. Chaudhri.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaudhri, N., Caggiula, A.R., Donny, E.C. et al. Self-administered and noncontingent nicotine enhance reinforced operant responding in rats: impact of nicotine dose and reinforcement schedule. Psychopharmacology 190, 353–362 (2007). https://doi.org/10.1007/s00213-006-0454-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0454-8