Abstract
Rationale
Greater incidence of anxiety and depressive disorders of women compared to men may be due in part to progesterone (P) and its neuroactive metabolite, 5α-pregnan-3α-ol-20-one (3α,5α-THP), acting in limbic regions, such as the amygdala.
Objective
If P's metabolism via 5α-reduction to 3α,5α-THP in the amygdala is critical for antianxiety and antidepressive behavior, then blocking 5α-reductase in the amygdala of female rats is likely to attenuate the antianxiety and antidepressive effects of high progestin levels from both endogenous and exogenous sources.
Methods
Naturally receptive female rats with high endogenous estrogen (E2) and P and ovariectomized (ovx) rats administered E2 (10 μg) and P (500 μg) subcutaneously were administered finasteride (10 μg/μl), a Type II 5α-reductase inhibitor, or vehicle to the amygdala. Anxiety behavior (open field, elevated plus maze, defensive freezing) and depressive behavior (Porsolt forced swim test) were assessed.
Results
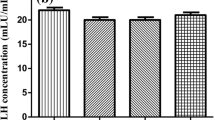
There were similar effects of finasteride administration to the amygdala to attenuate antianxiety behavior in naturally receptive and ovx, hormone-primed rats. Finasteride administration significantly decreased central entries in the open field, decreased open arm time in the elevated plus maze, increased defensive freezing in response to footshock, and increased time spent immobile compared to vehicle.
Conclusions
Thus, formation and subsequent actions of 3α,5α-THP in the amygdala may be important for antianxiety and antidepressive effects.





Similar content being viewed by others
References
Adamec R, Shallow T (2000) Effects of baseline anxiety on response to kindling of the right medial amygdala. Physiol Behav 70:67–80
Akirav I, Richter-Levin G (1999) Biphasic modulation of hippocampal plasticity by behavioral stress and basolateral amygdala stimulation in the rat. J Neurosci 19:10530–10535
Akwa Y, Purdy RH, Koob GF, Britton KT (1999) The amygdala mediates the anxiolytic-like effect of the neurosteroid allopregnanolone in rat. Behav Brain Res 106:119–125
Altomare G, Capella GL (2002) Depression circumstantially related to the administration of finasteride for androgenetic alopecia. J Dermatol 29:665–669
Bitran D, Hilvers RJ, Kellogg CK (1991) Anxiolytic effects of 3α-hydroxy-5α[β]-pregnan-20-one: endogenous metabolites of progesterone that are active at the GABAA receptor. Brain Res 561:157–161
Bitran D, Dugan M, Renda P, Ellis R, Foley M (1999) Anxiolytic effects of the neuroactive steroid pregnanolone (3α-OH-5α-pregnan-20-one) after microinjection in the dorsal hippocampus and lateral septum. Brain Res 850:217–224
Bitran D, Foley M, Audette D, Leslie N, Frye CA (2000) Activation of peripheral mitochondrial benzodiazepine receptors in the hippocampus stimulates allopregnanolone synthesis and produces anxiolytic-like effects in the rat. Psychopharmacology 151:64–71
Cheng YJ, Karavolas HJ (1973) Conversion of progesterone to 5α-pregnane-3,20-dione and 3α-hydroxy-5α-pregnan-20-one by rat medical basal hypothalami and the effects of estradiol and stage of estrous cycle on the conversion. Endocrinology 93:1157–1162
Davis M (1992a) The role of the amygdala in fear and anxiety. Annu Rev Neurosci 15:353–375
Davis M (1992b) The role of the amygdala in fear-potentiated startle: implications for animal models of anxiety. Trends Pharmacol Sci 13:35–41
Davis M, Whalen PJ (2001) The amygdala: vigilance and emotion. Mol Psychiatry 6:13–34
Emmert MH, Herman JP (1999) Differential forebrain c-fos mRNA induction by ether inhalation and novelty; evidence for distinctive stress pathways. Brain Res 845:60–67
Fernandez-Guasti A, Picazo O (1992) Changes in burying behavior during the estrous cycle; effect of estrogen and progesterone. Psychoneuroendocrinology 17:681–689
Frye CA, Bayon LE (1999) Mating stimuli influence endogenous variations in the neurosteroids 3α,5α-THP and 3α-Diol. J Neuroendocrinol 11:839–847
Frye CA, Duncan JE (1996) Estradiol benzoate potentiates neuroactive steroids' effects on pain sensitivity. Pharmacol Biochem Behav 53:27–32
Frye CA, Edinger KL (2004) Testosterone's metabolism in the hippocampus may mediate its anti-anxiety effects in male rats. Pharmacol Biochem Behav 78:473–481
Frye CA, Vongher JM (1999) 3α,5α-THP in the midbrain ventral tegmental area of rats and hamsters is increased in exogenous hormonal states associated with estrous cyclicity and sexual receptivity. J Endocrinol Invest 22:455–464
Frye CA, Walf AA (2002) Changes in progesterone metabolites in the hippocampus can modulate open field and forced swim test behavior of proestrous rats. Horm Behav 41:306–315
Frye CA, Walf AA (2004a) Estrogen and/or progesterone administered systemically or to the amygdala can have anxiety-, fear-, and pain-reducing effects in ovariectomized rats. Behav Neurosci 118:306–313
Frye CA, Walf AA (2004b) Hippocampal 3α,5α-THP may alter depressive behavior of pregnant and lactating rats. Pharmacol Biochem Behav 78:531–540
Frye CA, Petralia SM, Rhodes ME (2000) Estrous cycle and sex differences in performance on anxiety tasks coincide with increases in hippocampal progesterone and 3α,5α-THP. Pharmacol Biochem Behav 67:587–596
Frye CA, Walf AA, Rhodes ME, Harney JP (2004) Progesterone enhances motor, anxiolytic, analgesic, and antidepressive behavior of wild-type mice, but not those deficient in type 1 5α-reductase. Brain Res 1004:116–124
Furukawa A, Miyatake A, Ohnishi T, Ichikawa Y (1998) Steroidogenic acute regulatory protein (StAR) transcripts constitutively expressed in the adult rat central nervous system: colocalization of StAR, cytochrome P-450SCC (CYP XIA1), and 3β-hydroxysteroid dehydrogenase in the rat brain. J Neurochem 71:2231–2238
Greco B, Blasberg ME, Kosinski EC, Blaustein JD (2003) Response of ERα-IR and ERβ-IR cells in the forebrain of female rats to mating stimuli. Horm Behav 43:444–453
Gulinello M, Smith SS (2003) Anxiogenic effects of neurosteroid exposure: sex differences and altered GABAA receptor pharmacology in adult rats. J Pharmacol Exp Ther 305:541–548
Gulinello M, Gong QH, Li X, Smith SS (2001) Short-term exposure to a neuroactive steroid increases α4 GABA(A) receptor subunit levels in association with increased anxiety in the female rat. Brain Res 910:55–66
Gulinello M, Orman R, Smith SS (2003) Sex differences in anxiety, sensorimotor gating and expression of the α4 subunit of the GABAA receptor in the amygdala after progesterone withdrawal. Eur J Neurosci 17:641–648
Herman JP, Mueller NK, Figueiredo H (2004) Role of GABA and glutamate circuitry in hypothalamo–pituitary–adrenocortical stress integration. Ann N Y Acad Sci 1018:35–45
Hojo Y, Hattori TA, Enami T, Furukawa A, Suzuki K, Ishii HT, Mukai H, Morrison JH, Janssen WG, Kominami S, Harada N, Kimoto T, Kawato S (2004) Adult male rat hippocampus synthesizes estradiol from pregnenolone by cytochromes P45017α and P450 aromatase localized in neurons. Proc Natl Acad Sci U S A 101:865–870
Kellogg CK, Barrett KA (1999) Reduced progesterone metabolites are not critical for plus-maze performance of lactating female rats. Pharmacol Biochem Behav 63:441–448
Khanna M, Qin KN, Cheng KC (1995) Distribution of 3α-hydroxysteroid dehydrogenase in rat brain and molecular cloning of multiple cDNAs encoding structurally related proteins in humans. J Steroid Biochem Mol Biol 53:41–46
Laconi MR, Casteller G, Gargiulo PA, Bregonzio C, Cabrera RJ (2001) The anxiolytic effect of allopregnanolone is associated with gonadal hormonal status in female rats. Eur J Pharmacol 417:111–116
LeDoux JE (1992) Brain mechanisms of emotion and emotional learning. Curr Opin Neurobiol 2:191–197
LeDoux JE (1995) Emotion: clues from the brain. Annu Rev Psychol 46:209–235
LeDoux JE (2000) Emotion circuits in the brain. Annu Rev Neurosci 23:155–184
Lee Y, Davis M (1997) Role of the hippocampus, the bed nucleus of the stria terminalis, and the amygdala in the excitatory effect of corticotropin-releasing hormone on the acoustic startle reflex. J Neurosci 17:6434–6446
Leigland LA, Schulz LE, Janowsky JS (2004) Age related changes in emotional memory. Neurobiol Aging 25:1117–1124
Li X, Berties PJ, Karavolas HJ (1997) Regional distribution of cytostolic and particulate 5α-dihydroprogesterone 3α-hydroxysteroid oxidoreductase in female rat brain. J Steroid Biochem Mol Biol 60:311–318
Majewska MD, Harrison NL, Schwartz RD, Barker JL, Paul SM (1986) Steroid hormone metabolites are barbiturate-like modulators of the GABA receptor. Science 232:1004–1007
Martinez-Mota L, Contreras CM, Saavedra M (1999) Progesterone reduces immobility in rats forced to swim. Arch Med Res 30:286–289
Martinez-Mota L, Estrada-Camarena E, Lopez-Rubalcava C, Contreras CM, Fernandez-Guasti A (2000) Interaction of desipramine with steroid hormones on experimental anxiety. Psychoneuroendocrinology 25:109–120
McCarthy MM, Felzenberg E, Robbins A, Pfaff DW, Schwartz-Giblin S (1995) Infusions of diazepam and allopregnanolone into the midbrain central gray facilitate open-field behavior and sexual receptivity in female rats. Horm Behav 29:279–295
Mellon SH, Deschepper CF (1993) Neurosteroid biosynthesis: genes for adrenal steroidogenic enzymes are expressed in the brain. Brain Res 629:283–292
Mellon SH, Griffin LD (2002) Neurosteroids: biochemistry and clinical significance. Trends Endocrinol Metab 13:35–43
Molina-Hernandez M, Perez JG, Olivera Lopez JI (2002) Female Wistar rats tested during late proestrus or during pregnancy and ovariectomized rats tested after receiving progesterone or allopregnanolone displayed reduced conflict behavior. Prog Neuropsychopharmacol Biol Psychiatry 26:839–844
Molina-Hernandez M, Tellez-Alcantara NP, Perez Garcia J, Olivera Lopez JI, Teresa Jaramillo M (2003) Anti-conflict-like actions of intralateral septal infusions of allopregnanolone in Wistar rats. Pharmacol Biochem Behav 75:397–404
Mora S, Dussaubat N, Diaz-Veliz G (1996) Effects of the estrous cycle and ovarian hormones on behavioral indices of anxiety in female rats. Psychoneuroendocrinology 21:609–620
Nomikos GG, Spyraki C (1988) Influence of oestrogen on spontaneous and diazepam-induced exploration of rats in an elevated plus maze. Neuropharmacology 27:691–696
Patchev VK, Hassan AH, Holsboer DF, Almeida OF (1996) The neurosteroid tetrahydroprogesterone attenuates the endocrine response to stress and exerts glucocorticoid-like effects on vasopressin gene transcription in the rat hypothalamus. Neuropsychopharmacology 15:533–540
Paxinos G, Watson C (1986) The rat brain in stereotaxic coordinates. Academic, New York, NY
Pellow S, File SE (1986) Anxiolytic and anxiogenic drug effects on exploratory activity in an elevated plus-maze as measure of anxiety in the rat. J Neurosci Methods 14:149–167
Picazo O, Fernandez-Guasti A (1995) Anti-anxiety effects of progesterone and some of its reduced metabolites: an evaluation using the burying behavior test. Brain Res 680:135–141
Rhodes ME, Frye CA (2001) Inhibiting progesterone metabolism in the hippocampus of proestrous rats decreases anxiolytic, and enhances, exploration and analgesia. Cogn Affect Behav Neurosci 1:287–296
Sierra A (2004) Neurosteroids: the StAR protein in the brain. J Neuroendocrinol 16:787–793
Silveira MC, Sandner G, Graeff FG (1993) Induction of Fos immunoreactivity in the brain by exposure to elevated plus maze. Behav Brain Res 56:115–118
Sinchak K, Mills RH, Tao L, LaPolt P, Lu JK, Micevych P (2003) Estrogen induces de novo progesterone synthesis in astrocytes. Dev Neurosci 25:343–348
Toufexis DJ, Davis C, Hammond A, Davis M (2004) Progesterone attenuates corticotropin-releasing factor-enhanced but not fear-potentiated startle via the activity of its neuroactive metabolite, allopregnanolone. J Neurosci 24:10280–10287
Treit D, Pinel JP, Fibiger HC (1981) Conditioned defensive burying: a new paradigm for the study of anxiolytic agents. Pharmacol Biochem Behav 15:619–626
Vongher JM, Frye CA (1999) Progesterone in conjunction with estradiol has neuroprotective effects in an animal model of neurodegeneration. Pharmacol Biochem Behav 64:777–785
Walf AA, Frye CA (2004) Anti-nociception following exposure to trimethylthiazoline, peripheral or intra-amygdala estrogen and/or progesterone. Behav Brain Res 144:77–85
Walf AA, Frye CA (2005a) Antianxiety and antidepressive behavior produced by physiological estradiol regimen may be modulated by hypothalamic-pituitary-adrenal axis activity. Neuropsychopharmacology 30:1288–1301
Walf AA, Frye CA (2005b) ERβ-selective estrogen receptor modulators produce anti-anxiety behavior when administered systemically to ovariectomized rats. Neuropsychopharmacology (in press)
Walf AA, Rhodes ME, Frye CA (2004) Anti-depressant effects of ERβ selective estrogen receptor modulators in the forced swim test. Pharmacol Biochem Behav 78:523–529
Walker DL, Davis M (1997) Double dissociation between the involvement of the bed nucleus of the stria terminalis and the central nucleus of the amygdala in startle increases produced by conditioned versus unconditioned fear. J Neurosci 17:9375–9383
Walker DL, Toufexis DJ, Davis M (2003) Role of the bed nucleus of the stria terminalis versus the amygdala in fear, stress, and anxiety. Eur J Pharmacol 463:199–216
Wilensky AE, Schafe GE, LeDoux JE (2000) The amygdala modulates memory consolidation of fear-motivated inhibitory avoidance learning but not classical fear conditioning. J Neurosci 20:7059–7066
Acknowledgements
This research was supported by grants from the National Science Foundation (IBN98-96263; IBN03-16083), National Institute of Mental Health (MH0676980), Whitehall Foundation (096-010), and Ronald McNair Research Program to support minority undergraduates. Technical assistance provided by Lesley Ann Cole, Kate Fagan-Solis, and Dr. Madeline Rhodes is greatly appreciated. All experiments comply with the current laws of the United States of America, where they were performed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Walf, A.A., Sumida, K. & Frye, C.A. Inhibiting 5α-reductase in the amygdala attenuates antianxiety and antidepressive behavior of naturally receptive and hormone-primed ovariectomized rats. Psychopharmacology 186, 302–311 (2006). https://doi.org/10.1007/s00213-005-0100-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-005-0100-x