Abstract
Rationale
The pedunculopontine tegmental nucleus (PPTg) is involved in the execution and regulation of a variety of behaviors. Most investigations used brain lesions that have certain disadvantages, such as functional compensation over time.
Objectives
In the present study, we investigated by temporary, reversible inhibition of neurons the role of the PPTg in sensorimotor gating, measured as prepulse inhibition (PPI) of the acoustic startle response (ASR) using variable interstimulus intervals (ISI). In a second set of experiments we examined by the same technique the role of the PPTg in a progressive-ratio instrumental response task.
Methods
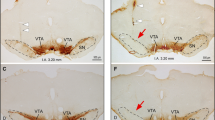
Local infusions of the GABAA-receptor agonist muscimol (0.05 μg and 0.5 μg/0.3 μl, or vehicle) were applied through indwelling microinfusion cannulae into the PPTg of freely moving rats. ASR and PPI were measured using acoustic stimuli of 100 dB (pulse) and 80 dB (prepulse) using ISIs of 25, 120, 520 and 1,020 ms. Instrumental behavior (lever pressing for casein pellets) was assessed in a Skinner box. Motor activity was measured in an open field.
Results
Intra-PPTg infusions of muscimol dose-dependently attenuated PPI at ISIs of 120 ms and 520 ms, but not at longer or shorter ISIs. ASR magnitude in pulse-alone trials was not significantly affected. Intra-PPTg infusion of 0.5 μg muscimol reduced the break point of instrumental responding (testing sequence where the rats fail to respond according to an increased ratio of reinforcement). No effects on food-preference and open-field activity were found.
Conclusions
These findings suggest that GABAergic neurotransmission in the PPTg plays an important role for sensorimotor gating at intermediate ISIs and for response selection under demanding schedules of reinforcement.




Similar content being viewed by others
References
Alderson HL, Brown VJ, Latimer MP, Brasted PJ, Robertson AH et al. (2002) The effect of excitotoxic lesions of the pedunculopontine tegmental nucleus on performance of a progressive ratio schedule of reinforcement. Neuroscience 112:417–425
Fendt M, Li L, Yeomans JS (2001) Brain stem circuits mediating prepulse inhibition of the startle reflex. Psychopharmacology 156:216–224
Hodos W (1961) Progressive ratio as a measure of reward strength. Science 134:943–944
Hoffman HS, Ison JR (1980) Reflex modification in the domain of startle. I. Some empirical findings and their implications for how the nervous system processes sensory input. Psychol Rev 87:175–189
Homs-Ormo S, Coll-Andreu M, Satorra-Marin N, Arevalo-Garcia R, Morgado-Bernal I (2003) Effects of pedunculopontine tegmental nucleus lesions on emotional reactivity and locomotion in rats. Brain Res Bull 59:495–503
Inglis WL, Winn P (1995) The pedunculopontine tegmental nucleus: where the striatum meets the reticular formation. Prog Neurobiol 47:1–29
Inglis WL, Dunbar JS, Winn P (1994) Outflow from the nucleus accumbens to the pedunculopontine tegmental nucleus: a dissociation between locomotor activity and the acquisition of responding for conditioned reinforcement stimulated by d-amphetamine. Neuroscience 62:51–64
Inglis WL, Olmstead MC, Robbins TW (2000) Pedunculopontine tegmental nucleus lesions impair stimulus-reward learning in autoshaping and conditioned reinforcement paradigms. Behav Neurosci 114:285–294
Jones CK, Shannon HE (2004) Lesions of the laterodorsal tegmental nucleus disrupt prepulse inhibition of the acoustic startle reflex. Pharmacol Biochem Behav 78:229–237
Kang Y, Kitai ST (1990) Electrophysiological properties of pedunculopontine neurons and their postsynaptic responses following stimulation of substantia nigra reticulata. Brain Res 535:79–95
Karnovsky MJ, Roots L (1964) A “direct-coloring” thiocholine method for cholinesterases. J Histochem Cytochem 12:219–221
Koch M, Fendt M (2003) Startle response modulation as a behavioral tool in neuropharmacology. Curr Neuropharmacol 1:175–185
Koch M, Kungel M, Herbert H (1993) Cholinergic neurons in the pedunculopontine tegmental nucleus are involved in the mediation of prepulse inhibition of the acoustic startle response in the rat. Exp Brain Res 97:71–82
Koch M, Fendt M, Kretschmer BD (2000) Role of the substantia nigra pars reticulata in sensorimotor gating, measured by prepulse inhibition of startle in rats. Behav Brain Res 117:153–162
Lepore M, Franklin KB (1996) N-Methyl-d-aspartate lesions of the pedunculopontine nucleus block acquisition and impair maintenance of responding reinforced with brain stimulation. Neuroscience 71:147–155
Martin JH, Ghez C (1999) Pharmacological inactivation in the analysis of the central control of movement. J Neurosci Meth 86:145–159
Mobini S, Chiang TJ, Ho MY, Bradshaw CM, Szabadi E (2000) Comparison of the effects of clozapine, haloperidol, chlorpromazine and d-amphetamine on performance on a time-constrained progressive ratio schedule and on locomotor behaviour in the rat. Psychopharmacology 152:47–54
Olmstead MC, Munn EM, Franklin KB, Wise RA (1998) Effects of pedunculopontine tegmental nucleus lesions on responding for intravenous heroin under different schedules of reinforcement. J Neurosci 18:5035–5044
Olmstead MC, Inglis WL, Bordeaux CP, Clarke EJ, Wallum EP et al. (1999) Lesions of the pedunculopontine tegmental nucleus increase sucrose consumption but do not affect discrimination or contrast effects. Behav Neurosci 113:732–743
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates. Academic, San Diego
Reijmers LG, Peeters BW (1994) Effects of acoustic prepulses on the startle reflex in rats: a parametric analysis. Brain Res 661:174–180
Reilly S (1999) Reinforcement value of gustatory stimuli determined by progressive ratio performance. Pharmacol Biochem Behav 63:301–311
Swerdlow NR, Geyer MA (1993) Prepulse inhibition of acoustic startle in rats after lesions of the pedunculopontine tegmental nucleus. Behav Neurosci 107:104–117
Acknowledgements
This work was supported by the DFG SFB 517 (TP A11). We thank Ms. M. Brand for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Diederich, K., Koch, M. Role of the pedunculopontine tegmental nucleus in sensorimotor gating and reward-related behavior in rats. Psychopharmacology 179, 402–408 (2005). https://doi.org/10.1007/s00213-004-2052-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-004-2052-y