Abstract
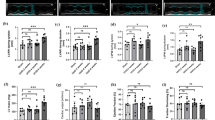
The incidence of chronic kidney disease is escalating; cardiorenal syndrome (CRS) type 4 is gaining a major health concern causing significant morbidity and mortality, putting major burdens on the healthcare system. This study was designed to compare the cardioprotective effects of carvedilol versus atenolol against CRS type 4 induced by subtotal 5/6 nephrectomy in rats and to explore the underlying mechanisms. Immediately after surgery, carvedilol (20 mg/kg/day) or atenolol (20 mg/kg/day) was added to drinking water for 10 weeks. Carvedilol was more effective than atenolol in improving kidney functions, decreasing elevated blood pressures, attenuating cardiac hypertrophy, reducing serum brain natriuretic peptide, and diminished cardiac fibrous tissue deposition. However, carvedilol was equivalent to atenolol in modulating β1-adrenergic receptors (β1ARs) and cardiac diacylglycerol (DAG) signaling, but carvedilol was superior in modulating β-arrestin2, phosphatidyl inositol 4,5 bisphosphates (PIP2), and caspase 3 levels. Carvedilol has superior cardioprotective effects than atenolol in a rat model of CRS type 4. These protective effects are mediated through modulating cardiac β1ARs/β-arrestin2/PIP2/DAG as well as abating cardiac apoptotic signaling pathways (caspase3/pS473 protein kinase B (Akt)).





Similar content being viewed by others
Data availability
The data of this study are available from the corresponding author upon request.
References
Abeyrathna P, Su Y (2015) The critical role of Akt in cardiovascular function. Vascul Pharmacol 74:38–48. https://doi.org/10.1016/j.vph.2015.05.008
Askari H, Seifi B, Kadkhodaee M (2016) Evaluation of renal-hepatic functional indices and blood pressure based on the progress of time in a rat model of chronic kidney disease. Nephrourol Mon 8:e37840. https://doi.org/10.5812/numonthly.37840
Bakris GL, Hart P, Ritz E (2006) Beta blockers in the management of chronic kidney disease. Kidney Int 70:1905–1913. https://doi.org/10.1038/sj.ki.5001835
Bathgate-Siryk A, Dabul S, Pandya K, Walklett K, Rengo G, Cannavo A, De Lucia C, Liccardo D, Gao E, Leosco D, Koch WJ, Lymperopoulos A (2014) Negative impact of β-arrestin-1 on post-myocardial infarction heart failure via cardiac and adrenal-dependent neurohormonal mechanisms. Hypertension 63:404–412. https://doi.org/10.1161/HYPERTENSIONAHA.113.02043
Carlstrom M, Montenegro MF (2019) Therapeutic value of stimulating the nitrate-nitrite-nitric oxide pathway to attenuate oxidative stress and restore nitric oxide bioavailability in cardiorenal disease. J Intern Med 285:2–18. https://doi.org/10.1111/joim.12818
Chatterjee S, Biondi-Zoccai G, Abbate A, D’Ascenzo F, Castagno D, Van Tassell B, Mukherjee D, Lichstein E (2013) Benefits of blockers in patients with heart failure and reduced ejection fraction: network meta-analysis. BMJ 346:f55–f55. https://doi.org/10.1136/bmj.f55
Cice G, Ferrara L, D’Andrea A, D’Isa S, Di Benedetto A, Cittadini A, Russo PE, Golino P, Calabrò R (2003) Carvedilol increases two-year survivalin dialysis patients with dilated cardiomyopathy. J Am Coll Cardiol 41:1438–1444. https://doi.org/10.1016/S0735-1097(03)00241-9
Cice G, Ferrara L, Di Benedetto A, Russo PE, Marinelli G, Pavese F, Iacono A (2001) Dilated cardiomyopathy in dialysis patients—beneficial effects of carvedilol: a double-blind, placebo-controlled trial. J Am Coll Cardiol 37:407–411. https://doi.org/10.1016/S0735-1097(00)01158-X
Clementi A, Virzì GM, Brocca A, de Cal M, Vescovo G, Granata A, Ronco C (2013a) Cardiorenal syndrome type 4: management. Blood Purif 36:200–209. https://doi.org/10.1159/000356369
Clementi A, Virzì GM, Goh CY, Cruz DN, Granata A, Vescovo G, Ronco C (2013b) Cardiorenal syndrome type 4: a review. Cardiorenal Med 3:63–70. https://doi.org/10.1159/000350397
Cowie M (2003) Clinical applications of B-type natriuretic peptide (BNP) testing. Eur Heart J 24:1710–1718. https://doi.org/10.1016/S0195-668X(03)00476-7
Doughty RN, White HD (2007) Carvedilol: use in chronic heart failure. Expert Rev Cardiovasc Ther 5:21–31. https://doi.org/10.1586/14779072.5.1.21
Esmaeeli A, Keshavarz Z, Dehdar F, Assadi M, Seyedabadi M (2020) The effects of carvedilol, metoprolol and propranolol on cisplatin-induced kidney injury. Drug Chem Toxicol 1–7https://doi.org/10.1080/01480545.2020.1846551
Fares H, Lavie CJ, Ventura HO (2012) Vasodilating versus first-generation β-blockers for cardiovascular protection. Postgrad Med 124:7–15. https://doi.org/10.3810/pgm.2012.03.2532
Feng X, Liu X, Zhang W, Xiao W (2011) p53 directly suppresses BNIP3 expression to protect against hypoxia-induced cell death. EMBO J 30:3397–3415. https://doi.org/10.1038/emboj.2011.248
Fernandes-Charpiot IMM,Caldas HC, Mendes GEF,Gomes de Sá Neto L, Oliveira HL, Baptista MSF,Abbud-Filho M (2016) (2016) Validation of an Experimental Model to Study Less Severe Chronic Renal Failure. J Investig Surg 29(5):309–315. https://doi.org/10.3109/08941939.2015.1114689
Fruman DA, Chiu H, Hopkins BD, Bagrodia S, Cantley LC, Abraham RT (2017) The PI3K pathway in human disease. Cell 170:605–635. https://doi.org/10.1016/j.cell.2017.07.029
Go AS, Chertow GM, Fan D, McCulloch CE, Hsu C (2004) Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351:1296–1305. https://doi.org/10.1056/NEJMoa041031
Gori M, Senni M, Gupta DK, Charytan DM, Kraigher-Krainer E, Pieske B, Claggett B, Shah AM, Santos ABS, Zile MR, Voors AA, McMurray JJV, Packer M, Bransford T, Lefkowitz M, Solomon SD (2014) Association between renal function and cardiovascular structure and function in heart failure with preserved ejection fraction. Eur Heart J 35:3442–3451. https://doi.org/10.1093/eurheartj/ehu254
Gounden V, Bhatt H, Jialal I (2021) Renal function tests. In: StatPearls [Internet]. Treasure Island: StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK507821/. Accessed 13 June 2021
Hamzaoui M, Djerada Z, Brunel V, Mulder P, Richard V, Bellien J,Guerrot D (2020) 5/6 nephrectomy induces different renal cardiac and vascular consequences in 129/Sv and C57BL/6JRj mice. Scientific Reports 10(1). https://doi.org/10.1038/s41598-020-58393-w
Hawwa N, Schreiber MJ, Tang WHW (2013) Pharmacologic management of chronic reno-cardiac syndrome. Curr Heart Fail Rep 10:54–62. https://doi.org/10.1007/s11897-012-0122-8
Hong F, Zhang Y, Cheng W, Sun X, Wang J (2019) β-arrestin-2 up-regulates toll-like receptor 2 signaling and inhibits apoptosis in human endometrial cancer heterotransplants in nude mice. BMC Cancer 19:1035. https://doi.org/10.1186/s12885-019-6254-4
Ibrahim NE, Gaggin HK, Konstam MA, Januzzi JL (2016) Established and emerging roles of biomarkers in heart failure clinical trials. Circ Hear Fail 9:e002528. https://doi.org/10.1161/CIRCHEARTFAILURE.115.002528
Ibrahim NE, Januzzi JL (2018) Established and emerging roles of biomarkers in heart failure. Circ Res 123:614–629. https://doi.org/10.1161/CIRCRESAHA.118.312706
Ibrahim WS, Ibrahim IAAE-H, Mahmoud MF, Mahmoud AAA (2020) Carvedilol diminishes cardiac remodeling induced by high-fructose/high-fat diet in mice via enhancing cardiac β-arrestin2 signaling. J Cardiovasc Pharmacol Ther 25:354–363. https://doi.org/10.1177/1074248420905683
Jornayvaz FR, Shulman GI (2012) Diacylglycerol activation of protein kinase Cε and hepatic insulin resistance. Cell Metab 15:574–584. https://doi.org/10.1016/j.cmet.2012.03.005
Kim I-M, Tilley DG, Chen J, Salazar NC, Whalen EJ, Violin JD, Rockman HA (2008) β-Blockers alprenolol and carvedilol stimulate -arrestin-mediated EGFR transactivation. Proc Natl Acad Sci 105:14555–14560. https://doi.org/10.1073/pnas.0804745105
Kim J, Grotegut CA, Wisler JW, Mao L, Rosenberg PB, Rockman HA, Lefkowitz RJ (2020) The β-arrestin-biased β-adrenergic receptor blocker carvedilol enhances skeletal muscle contractility. Proc Natl Acad Sci 117:12435–12443. https://doi.org/10.1073/pnas.1920310117
Kovesdy CP, Quarles LD (2013) Fibroblast growth factor-23: what we know, what we don’t know, and what we need to know. Nephrol Dial Transplant 28:2228–2236. https://doi.org/10.1093/ndt/gft065
Kovesdy CP, Quarles LD (2016) FGF23 from bench to bedside. Am J Physiol Physiol 310:F1168–F1174. https://doi.org/10.1152/ajprenal.00606.2015
Li Y, Kang L, Rong K, Zhang Y, Suo Y, Yuan M, Bao Q, Shao S, Tse G, Li R, Liu T, Li G (2021) Renal protective effects and mechanisms of the angiotensin receptor-neprilysin inhibitor LCZ696 in mice with cardiorenal syndrome. Life Sci 280:119692. https://doi.org/10.1016/j.lfs.2021.119692
Lin L-L, Wang D, Wang W, Cheng Y-Q, Su D-F, Liu A-J (2015) Long-term treatment of clonidine, atenolol, amlodipine and dihydrochlorothiazide, but not enalapril, impairs the sexual function in male spontaneously hypertensive rats. PLoS ONE 10:e0116155. https://doi.org/10.1371/journal.pone.0116155
Manning G, Whyte DB, Martinez R, Hunter T, Sudarsanam S (2002) The protein kinase complement of the human genome. Science 298:1912–1934. https://doi.org/10.1126/science.1075762 ((80- ))
Matthees ESF, Haider RS, Hoffmann C, Drube J (2021) Differential regulation of GPCRs—are GRK expression levels the key? Front Cell Dev Biol 9:1303. https://doi.org/10.3389/fcell.2021.687489
McCrink KA, Maning J, Vu A, Jafferjee M, Marrero C, Brill A, Bathgate-Siryk A, Dabul S, Koch WJ, Lymperopoulos A (2017) β-Arrestin2 improves post–myocardial infarction heart failure via sarco(endo)plasmic reticulum Ca 2+-ATPase–dependent positive inotropy in cardiomyocytes. Hypertension 70:972–981. https://doi.org/10.1161/HYPERTENSIONAHA.117.09817
Mohamed RMSM, Morimoto S, Ibrahim IAAE-H, Zhan D-Y, Du C-K, Arioka M, Yoshihara T, Takahashi-Yanaga F, Sasaguri T (2016) GSK-3β heterozygous knockout is cardioprotective in a knockin mouse model of familial dilated cardiomyopathy. Am J Physiol Circ Physiol 310:H1808–H1815. https://doi.org/10.1152/ajpheart.00771.2015
Nelson CD, Perry SJ, Regier DS, Prescott SM, Topham MK, Lefkowitz RJ (2007) Targeting of diacylglycerol degradation to M1 muscarinic receptors by β-arrestins. Science 315:663–666. https://doi.org/10.1126/science.1134562 ((80- ))
Noor N, Patel CB, Rockman HA (2011) β-Arrestin: a signaling molecule and potential therapeutic target for heart failure. J Mol Cell Cardiol 51:534–541. https://doi.org/10.1016/j.yjmcc.2010.11.005
Paolillo S, Dell’Aversana S, Esposito I, Poccia A, Perrone Filardi P (2021) The use of β-blockers in patients with heart failure and comorbidities: Doubts, certainties and unsolved issues. Eur J Intern Med 88:9–14. https://doi.org/10.1016/j.ejim.2021.03.035
Park M, Hsu C, Li Y, Mishra RK, Keane M, Rosas SE, Dries D, Xie D, Chen J, He J, Anderson A, Go AS, Shlipak MG (2012) Associations between kidney function and subclinical cardiac abnormalities in CKD. J Am Soc Nephrol 23:1725–1734. https://doi.org/10.1681/ASN.2012020145
Park M, Steinberg SF (2018) Carvedilol prevents redox inactivation of cardiomyocyte β1-adrenergic receptors. JACC Basic to Transl Sci 3:521–532. https://doi.org/10.1016/j.jacbts.2018.06.002
Pateinakis P, Papagianni A (2011) Cardiorenal syndrome type 4—cardiovascular disease in patients with chronic kidney disease: epidemiology, pathogenesis, and management. Int J Nephrol 2011:1–8. https://doi.org/10.4061/2011/938651
Rangaswami J, Bhalla V, Blair JEA, Chang TI, Costa S, Lentine KL, Lerma EV, Mezue K, Molitch M, Mullens W, Ronco C, Tang WHW, McCullough PA (2019) Cardiorenal syndrome: Classification, pathophysiology, diagnosis, and treatment Strategies: a scientific statement from the American Heart Association. Circulation 139:e840–e878. https://doi.org/10.1161/CIR.0000000000000664
Rehman B, Sanchez DP, Shah S (2021) Atenolol. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK539844/. Accessed 9 June 2021
Rodriguez-Perez JC, Losada A, Anabitarte A, Cabrera J, Llobet J, Palop L, Plaza C (1997) Effects of the novel multiple-action agent carvedilol on severe nephrosclerosis in renal ablated rats. J Pharmacol Exp Ther 283:336–344
Ronco C, Bellasi A, Di Lullo L (2018) Cardiorenal syndrome: an overview. Adv Chronic Kidney Dis 25:382–390. https://doi.org/10.1053/j.ackd.2018.08.004
Ronco C, Haapio M, House AA, Anavekar N, Bellomo R (2008) Cardiorenal syndrome. J Am Coll Cardiol 52:1527–1539. https://doi.org/10.1016/j.jacc.2008.07.051
Ronco C, McCullough P, Anker SD, Anand I, Aspromonte N, Bagshaw SM, Bellomo R, Berl T, Bobek I, Cruz DN, Daliento L, Davenport A, Haapio M, Hillege H, House AA, Katz N, Maisel A, Mankad S, Zanco P, Mebazaa A, Palazzuoli A, Ronco F, Shaw A, Sheinfeld G, Soni S, Vescovo G, Zamperetti N, Ponikowski P (2010) Cardio-renal syndromes: report from the consensus conference of the Acute Dialysis Quality Initiative. Eur Heart J 31:703–711. https://doi.org/10.1093/eurheartj/ehp507
Seyedabadi M, Gharghabi M, Gurevich EV, Gurevich VV (2021) Receptor-Arrestin Interactions: the GPCR Perspective. Biomolecules 11:218. https://doi.org/10.3390/biom11020218
Sgobbo P, Pacelli C, Grattagliano I, Villani G, Cocco T (2007) Carvedilol inhibits mitochondrial complex I and induces resistance to H2O2-mediated oxidative insult in H9C2 myocardial cells. Biochim Biophys Acta - Bioenerg 1767:222–232. https://doi.org/10.1016/j.bbabio.2007.01.023
Shin J-I, Jeon Y-J, Lee S, Lee YG, Kim JB, Lee K (2019) G-protein-coupled receptor 120 mediates DHA-induced apoptosis by regulating IP3R, ROS and ER stress levels in cisplatin-resistant cancer cells. Mol Cells 42:252–261. https://doi.org/10.14348/molcells.2019.2440
Song H-P, Chu Z-G, Zhang D-X, Dang Y-M, Zhang Q (2018) PI3K–AKT pathway protects cardiomyocytes against hypoxia-induced apoptosis by MitoKATP-mediated mitochondrial translocation of pAKT. Cell Physiol Biochem 49:717–727. https://doi.org/10.1159/000493037
Švíglerová J, Kuncová J, Nalos L, Holas J, Tonar Z, Rajdl D, Štengl M (2012) Cardiac remodeling in rats with renal failure shows interventricular differences. Exp Biol Med 237:1056–1067. https://doi.org/10.1258/ebm.2012.012045
Švíglerová J, Kuncová J, Nalos L, Tonar Z, Rajdl D, Štengl M (2010) Cardiovascular parameters in rat model of chronic renal failure induced by subtotal nephrectomy. Physiol Res 59:S81–S88. https://doi.org/10.33549/physiolres.932003
Ter Maaten JM, Damman K, Verhaar MC, Paulus WJ, Duncker DJ, Cheng C, van Heerebeek L, Hillege HL, Lam CSP, Navis G, Voors AA (2016) Connecting heart failure with preserved ejection fraction and renal dysfunction: the role of endothelial dysfunction and inflammation. Eur J Heart Fail 18:588–598. https://doi.org/10.1002/ejhf.497
Ungerer M, Parruti G, Böhm M, Puzicha M, DeBlasi A, Erdmann E, Lohse MJ (1994) Expression of beta-arrestins and beta-adrenergic receptor kinases in the failing human heart. Circ Res 74:206–213. https://doi.org/10.1161/01.RES.74.2.206
van Gastel J, Hendrickx JO, Leysen H, Santos-Otte P, Luttrell LM, Martin B, Maudsley S (2018) β-arrestin based receptor signaling paradigms: potential therapeutic targets for complex age-related disorders. Front Pharmacol 9:1369. https://doi.org/10.3389/fphar.2018.01369
Weir MR (2009) β-blockers in the treatment of hypertension: are there clinically relevant differences? Postgrad Med 121:90–98. https://doi.org/10.3810/pgm.2009.05.2007
Wisler JW, DeWire SM, Whalen EJ, Violin JD, Drake MT, Ahn S, Shenoy SK, Lefkowitz RJ (2007) A unique mechanism of beta-blocker action: carvedilol stimulates beta-arrestin signaling. Proc Natl Acad Sci 104:16657–16662. https://doi.org/10.1073/pnas.0707936104
Zhang Y, Kompa AR (2014) A practical guide to subtotal nephrectomy in the rat with subsequent methodology for assessing renal and cardiac function. Nephrol.19(9):552–561. https://doi.org/10.1111/nep.12279
Author information
Authors and Affiliations
Contributions
“The authors declare that all data were generated in-house and that no paper mill was used.” RM, SE, and DA: conception and design the experiment. RM and ON: writing the manuscript and data management and analysis. All authors contributed to drafting and revising the article, approved the final version, and agreed to be accountable for all aspects of the study.
Corresponding author
Ethics declarations
Ethics approval
All animals received humane care in compliance with the Animal Care Guidelines of the National Institutes of Health (NIH), and the Research Ethics Committee of the Faculty of Pharmacy, Zagazig University, Zagazig, Egypt, approved the design of the experiment (approval number P4-8–2017).
Consent to participate
Not applicable.
Consent for publication
All authors approved the manuscript to be published in its final version.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohamed, R.M.S.M., Elshazly, S.M., Nafea, O.E. et al. Comparative cardioprotective effects of carvedilol versus atenolol in a rat model of cardiorenal syndrome type 4. Naunyn-Schmiedeberg's Arch Pharmacol 394, 2117–2128 (2021). https://doi.org/10.1007/s00210-021-02130-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-021-02130-1