Abstract
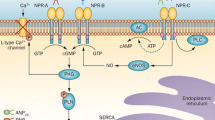
We found previously that stimulation of natriuretic peptide receptor (NPR)-B by C-type natriuretic peptide (CNP) in failing rat ventricle potentiates β1-adrenoceptor (β1-AR)-mediated inotropic response to noradrenaline through cGMP-mediated inhibition of phosphodiesterase (PDE) 3, thereby enhancing cAMP-mediated signalling. Increased cAMP-mediated signalling is deleterious in chronic heart failure (HF; basis for the use of β-blockers in HF) and we propose to consider NPR-B antagonists as new HF treatment in addition to conventional therapy. Since there is no NPR-B-selective antagonist available for clinical studies, we aimed at identifying a novel small molecule (non-peptide) NPR-B antagonist. An assay was developed and high throughput screening performed on a chemical library of about 20,000 small molecule compounds (<500 Da) to identify NPR-B antagonists based on inhibition of CNP-stimulated cGMP production in NPR-B-expressing HEK293 cells. The screen revealed several potential NPR-B antagonists, of which six were selected for further studies. Three showed selective NPR-B vs NPR-A inhibition and three were partially selective. The compounds mediated reversible, noncompetitive inhibition and most likely act as allosteric modulators binding outside the agonist binding site of NPR-B. In rat ventricular muscle strips, the potentiating effect of CNP upon β1-AR-evoked inotropic effects could be attenuated by at least one of these compounds. We identified several small molecule NPR-B antagonists by high throughput screening and show in a functional heart preparation that blocking NPR-B stimulation with a small molecule compound can reduce the potentiating effect of CNP on the β1-AR-mediated inotropic response to noradrenaline.






Similar content being viewed by others
References
Afzal F, Qvigstad E, Aronsen JM, Moltzau LR, Sjaastad I, Skomedal T, Osnes JB, Levy FO (2011) Agents increasing cyclic GMP amplify 5-HT4-elicited positive inotropic response in failing rat cardiac ventricle. Naunyn Schmiedeberg’s Arch Pharmacol 384:543–553
Amsallem E, Kasparian C, Haddour G, Boissel JP, Nony P (2005) Phosphodiesterase III inhibitors for heart failure. Cochrane Database Syst Rev CD002230
Anand-Srivastava MB, Sairam MR, Cantin M (1990) Ring-deleted analogs of atrial natriuretic factor inhibit adenylate cyclase/cAMP system. Possible coupling of clearance atrial natriuretic factor receptors to adenylate cyclase/cAMP signal transduction system. J Biol Chem 265:8566–8572
Bach T, Syversveen T, Kvingedal AM, Krobert KA, Brattelid T, Kaumann AJ, Levy FO (2001) 5-HT4(a) and 5-HT4(b) receptors have nearly identical pharmacology and are both expressed in human atrium and ventricle. Naunyn-Schmiedeberg’s Arch Pharmacol 363:146–160
Bockaert J, Hunzicker-Dunn M, Birnbaumer L (1976) Hormone-stimulated desensitization of hormone-dependent adenylyl cyclase. Dual action of luteinizing hormone on pig graafian follicle membranes. J Biol Chem 251:2653–2663
Christopoulos A (2002) Allosteric binding sites on cell-surface receptors: novel targets for drug discovery. Nat Rev Drug Discov 1:198–210
Conn PJ, Christopoulos A, Lindsley CW (2009) Allosteric modulators of GPCRs: a novel approach for the treatment of CNS disorders. Nat Rev Drug Discov 8:41–54
Das A, Xi L, Kukreja RC (2008) Protein kinase G-dependent cardioprotective mechanism of phosphodiesterase-5 inhibition involves phosphorylation of ERK and GSK3beta. J Biol Chem 283:29572–29585
Del Ry S, Maltinti M, Piacenti M, Passino C, Emdin M, Giannessi D (2006) Cardiac production of C-type natriuretic peptide in heart failure. J Cardiovasc Med (Hagerstown) 7:397–399
Deschenes J, Dupere C, McNicoll N, L’Heureux N, Auger F, Fournier A, De LA (2005) Development of a selective peptide antagonist for the human natriuretic peptide receptor-B. Peptides 26:517–524
Deschepper CF (2010) Cardioprotective actions of cyclic GMP: lessons from genetic animal models. Hypertension 55:453–458
Ebihara Y, Karmazyn M (1996) Inhibition of β- but not α1-mediated adrenergic responses in isolated hearts and cardiomyocytes by nitric oxide and 8-bromo cyclic GMP. Cardiovasc Res 32:622–629
Koh GY, Nussenzveig DR, Okolicany J, Price DA, Maack T (1992) Dynamics of atrial natriuretic factor-guanylate cyclase receptors and receptor-ligand complexes in cultured glomerular mesangial and renomedullary interstitial cells. J Biol Chem 267:11987–11994
Kukreja RC, Salloum FN, Das A (2012) Cyclic guanosine monophosphate signaling and phosphodiesterase-5 inhibitors in cardioprotection. J Am Coll Cardiol 59:1921–1927
Langenickel TH, Buttgereit J, Pagel-Langenickel I, Lindner M, Monti J, Beuerlein K, Al-Saadi N, Plehm R, Popova E, Tank J, Dietz R, Willenbrock R, Bader M (2006) Cardiac hypertrophy in transgenic rats expressing a dominant-negative mutant of the natriuretic peptide receptor B. Proc Natl Acad Sci U S A 103:4735–4740
Levy FO (2013) Cardiac PDEs and crosstalk between cAMP and cGMP signalling pathways in the regulation of contractility. Naunyn Schmiedeberg’s Arch Pharmacol 386:665–670
Lisurek M, Rupp B, Wichard J, Neuenschwander M, von Kries JP, Frank R, Rademann J, Kuhne R (2010) Design of chemical libraries with potentially bioactive molecules applying a maximum common substructure concept. Mol Divers 14:401–408
Nishikimi T, Maeda N, Matsuoka H (2006) The role of natriuretic peptides in cardioprotection. Cardiovasc Res 69:318–328
O’Connor CM, Starling RC, Hernandez AF, Armstrong PW, Dickstein K, Hasselblad V, Heizer GM, Komajda M, Massie BM, McMurray JJ, Nieminen MS, Reist CJ, Rouleau JL, Swedberg K, Adams KF Jr, Anker SD, Atar D, Battler A, Botero R, Bohidar NR, Butler J, Clausell N, Corbalan R, Costanzo MR, Dahlstrom U, Deckelbaum LI, Diaz R, Dunlap ME, Ezekowitz JA, Feldman D, Felker GM, Fonarow GC, Gennevois D, Gottlieb SS, Hill JA, Hollander JE, Howlett JG, Hudson MP, Kociol RD, Krum H, Laucevicius A, Levy WC, Mendez GF, Metra M, Mittal S, Oh BH, Pereira NL, Ponikowski P, Tang WH, Tanomsup S, Teerlink JR, Triposkiadis F, Troughton RW, Voors AA, Whellan DJ, Zannad F, Califf RM (2011) Effect of nesiritide in patients with acute decompensated heart failure. N Engl J Med 365:32–43
Packer M, Carver JR, Rodeheffer RJ, Ivanhoe RJ, DiBianco R, Zeldis SM, Hendrix GH, Bommer WJ, Elkayam U, Kukin ML (1991) Effect of oral milrinone on mortality in severe chronic heart failure. The PROMISE Study Research Group. N Engl J Med 325:1468–1475
Potter LR, Abbey-Hosch S, Dickey DM (2006) Natriuretic peptides, their receptors, and cyclic guanosine monophosphate-dependent signaling functions. Endocr Rev 27:47–72
Potter LR, Yoder AR, Flora DR, Antos LK, Dickey DM (2009) Natriuretic peptides: their structures, receptors, physiologic functions and therapeutic applications. Handb Exp Pharmacol 341–366
Qvigstad E, Moltzau LR, Aronsen JM, Nguyen CH, Hougen K, Sjaastad I, Levy FO, Skomedal T, Osnes JB (2010) Natriuretic peptides increase beta1-adrenoceptor signalling in failing hearts through phosphodiesterase 3 inhibition. Cardiovasc Res 85:763–772
Rose RA, Giles WR (2008) Natriuretic peptide C receptor signalling in the heart and vasculature. J Physiol 586:353–366
Sackner-Bernstein JD, Kowalski M, Fox M, Aaronson K (2005) Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials. JAMA 293:1900–1905
Salomon Y, Londos C, Rodbell M (1974) A highly sensitive adenylate cyclase assay. Anal Biochem 58:541–548
Skomedal T, Borthne K, Aass H, Geiran O, Osnes JB (1997) Comparison between alpha-1 adrenoceptor-mediated and beta adrenoceptor-mediated inotropic components elicited by norepinephrine in failing human ventricular muscle. J Pharmacol Exp Ther 280:721–729
Takimoto E (2012) Cyclic GMP-dependent signaling in cardiac myocytes. Circ J 76:1819–1825
van den Akker F (2001) Structural insights into the ligand binding domains of membrane bound guanylyl cyclases and natriuretic peptide receptors. J Mol Biol 311:923–937
Weninger S, De Maeyer JH, Lefebvre RA (2012) Study of the regulation of the inotropic response to 5-HT4 receptor activation via phosphodiesterases and its cross-talk with C-type natriuretic peptide in porcine left atrium. Naunyn Schmiedeberg’s Arch Pharmacol 385:565–577
Weninger S, De Maeyer JH, Lefebvre RA (2013) Influence of phosphodiesterases and cGMP on cAMP generation and on phosphorylation of phospholamban and troponin I by 5-HT receptor activation in porcine left atrium. Naunyn Schmiedeberg’s Arch Pharmacol 386:671–684
Acknowledgments
We wish to thank Niko Sahlberg and Prof. Kjetil Taskén at the Biotechnology Centre of Oslo for excellent help and support with the screening of the compound libraries. This work was supported by The Norwegian Council on Cardiovascular Disease, The Research Council of Norway, The Kristian Gerhard Jebsen Foundation, Anders Jahre’s Foundation for the Promotion of Science, The Family Blix Foundation, The Simon Fougner Hartmann Family Foundation, South-Eastern Norway Regional Health Authority, University of Oslo and the COST Action BM1005 (European Network on Gasotransmitters).
Conflicts of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bach, T., Bergholtz, S., Riise, J. et al. Identification of small molecule NPR-B antagonists by high throughput screening — potential use in heart failure. Naunyn-Schmiedeberg's Arch Pharmacol 387, 5–14 (2014). https://doi.org/10.1007/s00210-013-0940-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-013-0940-6