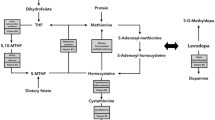
Abstract
Elevation of plasma total homocysteine concentrations were observed in levodopa/dopa decarboxylase inhibitor (DDI)-treated patients with Parkinson’s disease (PD). Degradation of levodopa to 3-O-methyldopa via the enzyme catechol-O-methyltransferase (COMT) is a methyl group demanding reaction. It generates homocysteine from the methyl group donor methionine. But there are inconsistent outcomes, as most investigators determined homocysteine after an overnight washout of levodopa. They did not consider the acute effects of levodopa/DDI intake in relation with COMT inhibition on homocysteine bioavailability. The purpose of this study is to measure levels of homocysteine, levodopa, and its metabolite 3-O-methyldopa in plasma after reiterated oral levodopa/DDI administration with and without the COMT-inhibitor entacapone (EN). Sixteen PD patients received 100 mg levodopa/carbidopa three times on day 1 and with EN on day 2 under standardized conditions. Homocysteine concentrations increased on day 1 and generally over the whole interval. No significant ascent of homocysteine appeared on day 2 only. Levodopa bioavailability was higher on day 2 due to the COMT inhibition. No change of 3-O-methyldopa appeared between both days. The correlation coefficients between homocysteine, levodopa, and 3-O-methyldopa were higher on day 1 than on day 2. Rise of homocysteine does not only depend on the oral levodopa dose, but also on the acute intake of levodopa/DDI with or without COMT inhibition. Measurements of homocysteine should consider acute repeated levodopa/DDI applications, as homocysteine and metabolically related 3-O-methyldopa accumulate due to their long plasma half-life in contrast to short-living levodopa.





Similar content being viewed by others
References
Ben Shlomo Y, Marmot MG (1995) Survival and cause of death in a cohort of patients with parkinsonism: possible clues to aetiology? J Neurol Neurosurg Psychiatry 58:293–299
Benetello P, Furlanut M, Fortunato M, Pea F, Baraldo M (1997) Levodopa and 3-O-methyldopa in cerebrospinal fluid after levodopa-carbidopa association. Pharmacol Res 35:313–315
Caccamo D, Gorgone G, Curro M, Parisi G, Di IW, Menichetti C, Belcastro V, Parnetti L, Rossi A, Pisani F, Ientile R, Calabresi P (2007) Effect of MTHFR polymorphisms on hyperhomocysteinemia in levodopa-treated Parkinsonian patients. Neuromolecular Med 9:249–254
Cacciapuoti F (2011) Hyper-homocysteinemia: a novel risk factor or a powerful marker for cardiovascular diseases? Pathogenetic and therapeutical uncertainties. J Thromb Thrombolysis. doi:10.1007/s11239-011-0550-4
Kuoppamaki M, Korpela K, Marttila R, Kaasinen V, Hartikainen P, Lyytinen J, Kaakkola S, Hanninen J, Loyttyniemi E, Kailajarvi M, Ruokoniemi P, Ellmen J (2009) Comparison of pharmacokinetic profile of levodopa throughout the day between levodopa/carbidopa/entacapone and levodopa/carbidopa when administered four or five times daily. Eur J Clin Pharmacol 65:443–455
Lamberti P, Zoccolella S, Iliceto G, Armenise E, Fraddosio A, de Mari M, Livrea P (2005) Effects of levodopa and COMT inhibitors on plasma homocysteine in Parkinson’s disease patients. Mov Disord 20:69–72
Miller JW, Shukitt-Hale B, Villalobos-Molina R, Nadeau MR, Selhub J, Joseph JA (1997) Effect of Levodopa and the catechol-O-methyltransferase inhibitor Ro 41–0960 on sulfur amino acid metabolites in rats. Clin Neuropharmacol 20:55–66
Muller T (2010a) Catechol-O-methyltransferase enzyme: cofactor S-adenosyl-L-methionine and related mechanisms. Int Rev Neurobiol 95:49–71
Muller T (2010b) The impact of COMT-inhibition on gastrointestinal levodopa absorption in patients with Parkinson’s disease. Clinical Medicine Insights: Therapeutics 2:155–168
Muller T, Kuhn W (2006) Tolcapone decreases plasma levels of S-adenosyl-L-homocysteine and homocysteine in treated Parkinson’s disease patients. Eur J Clin Pharmacol 62:447–450
Muller T, Kuhn W (2009) Homocysteine levels after acute levodopa intake in patients with Parkinson’s disease. Mov Disord 24:1339–1343
Muller T, Muhlack S (2009) Peripheral COMT inhibition prevents levodopa associated homocysteine increase. J Neural Transm 116:1253–1256
Muller T, Muhlack S (2010) Acute homocysteine rise after repeated levodopa application in patients with Parkinson’s disease. Parkinsonism Relat Disord 16:688–689
Muller T, Erdmann C, Muhlack S, Bremen D, Przuntek H, Goetze O, Woitalla D (2006) Pharmacokinetic behaviour of levodopa and 3-O-methyldopa after repeat administration of levodopa/carbidopa with and without entacapone in patients with Parkinson’s disease. J Neural Transm 113:1441–1448
Muller T, Jugel C, Ehret R, Ebersbach G, Bengel G, Muhlack S, Klostermann F (2011) Elevation of total homocysteine levels in patients with Parkinson’s disease treated with duodenal levodopa/carbidopa gel. J Neural Transm. doi:10.1007/s00702-011-0614-9
Nakaso K, Yasui K, Kowa H, Kusumi M, Ueda K, Yoshimoto Y, Takeshima T, Sasaki K, Nakashima K (2003) Hypertrophy of IMC of carotid artery in Parkinson’s disease is associated with levodopa, homocysteine, and MTHFR genotype. J Neurol Sci 207:19–23
Nevrly M, Kanovsky P, Vranova H, Langova K, Hlustik P (2010) Effect of entacapone on plasma homocysteine levels in Parkinson’s disease patients. Neurol Sci 31:565–569
Nissinen E, Nissinen H, Larjonmaa H, Vaananen A, Helkamaa T, Reenila I, Rauhala P (2005) The COMT inhibitor, entacapone, reduces levodopa-induced elevations in plasma homocysteine in healthy adult rats. J Neural Transm 112:1213–1221
Postuma RB, Espay AJ, Zadikoff C, Suchowersky O, Martin WR, Lafontaine AL, Ranawaya R, Camicioli R, Lang AE (2006) Vitamins and entacapone in levodopa-induced hyperhomocysteinemia: a randomized controlled study. Neurology 66:1941–1943
Valkovic P, Benetin J, Blazicek P, Valkovicova L, Gmitterova K, Kukumberg P (2005) Reduced plasma homocysteine levels in levodopa/entacapone treated Parkinson patients. Parkinsonism Relat Disord 11:253–256
Zesiewicz TA, Wecker L, Sullivan KL, Merlin LR, Hauser RA (2006) The controversy concerning plasma homocysteine in Parkinson disease patients treated with levodopa alone or with entacapone: effects of vitamin status. Clin Neuropharmacol 29:106–111
Acknowledgement
We thank Ulrike Beckmann, Marion Frickmann, Christa Kraushaar-Szesny, Christine Stamm, Tanja Steiner for technical assistance and the participating PD patients.
Conflicts of interests
The authors have nothing to disclose regarding this research project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Müller, T., Woitalla, D. & Muhlack, S. Inhibition of catechol-O-methyltransferase modifies acute homocysteine rise during repeated levodopa application in patients with Parkinson’s disease. Naunyn-Schmiedeberg's Arch Pharmacol 383, 627–633 (2011). https://doi.org/10.1007/s00210-011-0629-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-011-0629-7