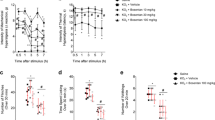
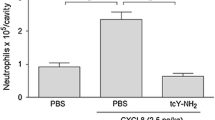
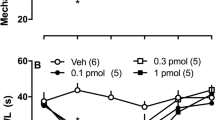
Abstract
Endothelin may contribute to the development of inflammatory events such as leukocyte recruitment and nociception. Herein, we investigated whether endothelin-mediated mechanical hypernociception (decreased nociceptive threshold, evaluated by electronic pressure-meter) and neutrophil migration (myeloperoxidase activity) are inter-dependent in antigen challenge-induced Th1-driven hind-paw inflammation. In antigen challenge-induced inflammation, endothelin (ET) ETA and ETB receptor antagonism inhibited both hypernociception and neutrophil migration. Interestingly, ET-1 peptide-induced hypernociception was not altered by inhibiting neutrophil migration or endothelin ETB receptor antagonism, but rather by endothelin ETA receptor antagonism. Furthermore, endothelin ETA, but not ETB, receptor antagonism inhibited antigen-induced PGE2 production, whereas either selective or combined blockade of endothelin ETA and/or ETB receptors reduced hypernociception and neutrophil recruitment caused by antigen challenge. Concluding, this study advances knowledge into the role for endothelin in inflammatory mechanisms and further supports the potential of endothelin receptor antagonists in controlling inflammation.





Similar content being viewed by others
References
Baamonde A, Lastra A, Villazon M, Bordallo J, Hidalgo A, Menendez L (2004) Involvement of endogenous endothelins in thermal and mechanical inflammatory hyperalgesia in mice. Naunyn Schmiedebergs Arch Pharmacol 369:245–251
Bisgaard H, Kristensen JK (1985) Leukotriene B4 produces hyperalgesia in humans. Prostaglandins 30:791–797
Bradley PP, Priebat DA, Christensen RD, Rothstein G (1982) Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol 78:206–209
Cambiaggi C, Mencarelli M, Muscettola M, Grasso G (2001) Gene expression of endothelin-1 (ET-1) and release of mature peptide by activated human neutrophils. Cytokine 14:230–233
Canetti C, Silva JS, Ferreira SH, Cunha FQ (2001) Tumour necrosis factor-alpha and leukotriene B(4) mediate the neutrophil migration in immune inflammation. Br J Pharmacol 134:1619–1628
Casagrande R, Georgetti SR, Verri WA Jr., Dorta DJ, dos Santos AC, Fonseca MJ (2006) Protective effect of topical formulations containing quercetin against UVB-induced oxidative stress in hairless mice. J Photochem Photobiol B 84:21–27
Chen D, Balyakina EV, Lawrence M, Christman BW, Meyrick B (2003) Cyclooxygenase is regulated by ET-1 and MAPKs in peripheral lung microvascular smooth muscle cells. Am J Physiol Lung Cell Mol Physiol 284:L614–621
Chichorro JG, Zampronio AR, Rae GA (2006) Endothelin ET(B) receptor antagonist reduces mechanical allodynia in rats with trigeminal neuropathic pain. Exp Biol Med (Maywood, NJ) 231:1136–1140
Conte FD, Barja-Fidalgo C, Verri WA Jr, Cunha FQ, Rae GA, Penido C, Henriques MD (2008) Endothelins modulate inflammatory reaction in zymosan-induced arthritis: participation of LTB4, TNF-α, and CXCL-1. J Leukoc Biol 84:652–660
Coroneos E, Kester M, Thomas P, Dunn MJ (1995) Endothelin regulates PGE2 formation in rat mesangial cells through induction of prostaglandin endoperoxide synthase-2. Adv Prostaglandin Thromboxane Leukot Res 23:117–119
Cunha TM, Verri WA Jr, Poole S, Parada CA, Cunha FQ, Ferreira SH (2007) Pain facilitation by proinflammatory cytokine actions at peripheral nerve terminals. In: Sorkin L, DeLeo J, Watkins LR (eds) Immune and glial regulation of pain. IASP, Seattle
Cunha TM, Barsante MM, Guerrero AT, Verri WA Jr., Ferreira SH, Coelho FM, Bertini R, Di Giacinto C, Allegretti M, Cunha FQ, Teixeira MM (2008a) Treatment with DF 2162, a non-competitive allosteric inhibitor of CXCR1/2, diminishes neutrophil influx and inflammatory hypernociception in mice. Br J Pharmacol 154:460–470
Cunha TM, Verri WA Jr, Schivo IR, Napimoga MH, Parada CA, Poole S, Teixeira MM, Ferreira SH, Cunha FQ (2008b) Crucial role of neutrophils in the development of mechanical inflammatory hypernociception. J Leukoc Biol 83:824–832
Cunha TM, Verri WA Jr., Vivancos GG, Moreira IF, Reis S, Parada CA, Cunha FQ, Ferreira SH (2004) An electronic pressure-meter nociception paw test for mice. Braz J Med Biol Res 37:401–407
da Cunha JM, Rae GA, Ferreira SH, Cunha FQ (2004) Endothelins induce ETB receptor-mediated mechanical hypernociception in rat hindpaw: roles of cAMP and protein kinase C. Eur J Pharmacol 501:87–94
Davar G, Hans G, Fareed MU, Sinnott C, Strichartz G (1998) Behavioral signs of acute pain produced by application of endothelin-1 to rat sciatic nerve. Neuroreport 9:2279–2283
Dhaun N, Pollock DM, Goddard J, Webb DJ (2007) Selective and mixed endothelin receptor antagonism in cardiovascular disease. Trends Pharmacol Sci 28:573–579
Du X, Tabeta K, Mann N, Crozat K, Mudd S, Beutler B (2005) An essential role for Rxr alpha in the development of Th2 responses. Eur J Immunol 35:3414–3423
Elferink JG, de Koster BM (1994) Endothelin-induced activation of neutrophil migration. Biochem Pharmacol 48:865–871
Elferink JG, de Koster BM (1996) Modulation of human neutrophil chemotaxis by the endothelin-B receptor agonist sarafotoxin S6c. Chem Biol Interact 101:165–174
Ethuin F, Delarche C, Benslama S, Gougerot-Pocidalo MA, Jacob L, Chollet-Martin S (2001) Interleukin-12 increases interleukin 8 production and release by human polymorphonuclear neutrophils. J Leukoc Biol 70:439–446
Ethuin F, Gerard B, Benna JE, Boutten A, Gougereot-Pocidalo MA, Jacob L, Chollet-Martin S (2004) Human neutrophils produce interferon gamma upon stimulation by interleukin-12. Lab Invest 84:1363–1371
Ferreira SH, Romitelli M, de Nucci G (1989) Endothelin-1 participation in overt and inflammatory pain. J Cardiovasc Pharmacol 13(Suppl 5):S220–222
Filep JG, Telemaque S, Battistini B, Sirois P, D’Orleans-Juste P (1993) Increased plasma levels of endothelin during anaphylactic shock in the guinea-pig. Eur J Pharmacol 239:231–236
Griswold DE, Douglas SA, Martin LD, Davis TG, Davis L, Ao Z, Luttmann MA, Pullen M, Nambi P, Hay DW, Ohlstein EH (1999) Endothelin B receptor modulates inflammatory pain and cutaneous inflammation. Mol Pharmacol 56:807–812
Guerrero AT, Verri WA Jr., Cunha TM, Silva TA, Rocha FA, Ferreira SH, Cunha FQ, Parada CA (2006) Hypernociception elicited by tibio-tarsal joint flexion in mice: a novel experimental arthritis model for pharmacological screening. Pharmacol Biochem Behav 84:244–251
Guerrero AT, Verri WA Jr., Cunha TM, Silva TA, Schivo IR, Dal-Secco D, Canetti C, Rocha FA, Parada CA, Cunha FQ, Ferreira SH (2008) Involvement of LTB4 in zymosan-induced joint nociception in mice: participation of neutrophils and PGE2. J Leukoc Biol 83:122–130
Ishida K, Takeshige K, Minakami S (1990) Endothelin-1 enhances superoxide generation of human neutrophils stimulated by the chemotactic peptide N-formyl-methionyl-leucyl-phenylalanine. Biochem Biophys Res Commun 173:496–500
Khodorova A, Fareed MU, Gokin A, Strichartz GR, Davar G (2002) Local injection of a selective endothelin-B receptor agonist inhibits endothelin-1-induced pain-like behavior and excitation of nociceptors in a naloxone-sensitive manner. J Neurosci 22:7788–7796
Lavich TR, Siqueira Rde A, Farias-Filho FA, Cordeiro RS, Rodrigues e Silva PM, Martins MA (2006) Neutrophil infiltration is implicated in the sustained thermal hyperalgesic response evoked by allergen provocation in actively sensitized rats. Pain 125:180–187
Levine JD, Lau W, Kwiat G, Goetzl EJ (1984) Leukotriene B4 produces hyperalgesia that is dependent on polymorphonuclear leukocytes. Science 225:743–745
Lund AK, Peterson SL, Timmins GS, Walker MK (2005) Endothelin-1-mediated increase in reactive oxygen species and NADPH oxidase activity in hearts of aryl hydrocarbon receptor (AhR) null mice. Toxicol Sci 88:265–273
Menendez L, Lastra A, Hidalgo A, Baamonde A (2003) Nociceptive reaction and thermal hyperalgesia induced by local ET-1 in mice: a behavioral and Fos study. Naunyn Schmiedebergs Arch Pharmacol 367:28–34
Nagase T, Fukuchi Y, Jo C, Teramoto S, Uejima Y, Ishida K, Shimizu T, Orimo H (1990) Endothelin-1 stimulates arachidonate 15-lipoxygenase activity and oxygen radical formation in the rat distal lung. Biochem Biophys Res Commun 168:485–489
Petri B, Phillipson M, Kubes P (2008) The physiology of leukocyte recruitment: an in vivo perspective. J Immunol 180:6439–6446
Piovezan AP, D’Orleans-Juste P, Tonussi CR, Rae GA (1997) Endothelins potentiate formalin-induced nociception and paw edema in mice. Can J Physiol Pharmacol 75:596–600
Piovezan AP, D’Orleans-Juste P, Tonussi CR, Rae GA (1998) Effects of endothelin-1 on capsaicin-induced nociception in mice. Eur J Pharmacol 351:15–22
Piovezan AP, D’Orleans-Juste P, Souza GE, Rae GA (2000) Endothelin-1-induced ET(A) receptor-mediated nociception, hyperalgesia and oedema in the mouse hind-paw: modulation by simultaneous ET(B) receptor activation. Br J Pharmacol 129:961–968
Piovezan AP, D’Orleans-Juste P, Frighetto M, Souza GE, Henriques MG, Rae GA (2004) Endothelins contribute towards nociception induced by antigen in ovalbumin-sensitised mice. Br J Pharmacol 141:755–763
Pontiroli AE, Pizzocri P, Koprivec D, Vedani P, Marchi M, Arcelloni C, Paroni R, Esposito K, Giugliano D (2004) Body weight and glucose metabolism have a different effect on circulating levels of ICAM-1, E-selectin, and endothelin-1 in humans. Eur J Endocrinol 150:195–200
Raffa RB, Schupsky JJ, Jacoby HI (1996) Endothelin-induced nociception in mice: mediation by ETA and ETB receptors. J Pharmacol Exp Ther 276:647–651
Sampaio AL, Rae GA, Henriques MG (2000) Participation of endogenous endothelins in delayed eosinophil and neutrophil recruitment in mouse pleurisy. Inflamm Res 49:170–176
Sampaio AL, Rae GA, Henriques MG (2004) Effects of endothelin ETA receptor antagonism on granulocyte and lymphocyte accumulation in LPS-induced inflammation. J Leukoc Biol 76:210–216
Sessa WC, Kaw S, Hecker M, Vane JR (1991) The biosynthesis of endothelin-1 by human polymorphonuclear leukocytes. Biochem Biophys Res Commun 174:613–618
Siminiak T, Ozawa T (1993) Neutrophil mediated myocardial injury. Int J Biochem 25:147–156
Su SB, Silver PB, Grajewski RS, Agarwal RK, Tang J, Chan CC, Caspi RR (2005) Essential role of the MyD88 pathway, but nonessential roles of TLRs 2, 4, and 9, in the adjuvant effect promoting Th1-mediated autoimmunity. J Immunol 175:6303–6310
Ting E, Guerrero AT, Cunha TM, Verri WA Jr, Taylor SM, Woodruff TM, Cunha FQ, Ferreira SH (2007) Role of complement C5a in mechanical inflammatory hypernociception: potential use of C5a receptor antagonists to control inflammatory pain. Br J Pharmacol 153:1043–1053
Tolone G, Bonasera L, Tolone C (1977) PGE2 production by human polymorphonuclear leukocytes. Boll Ist Sieroter Milan 56:399–400
Valerio DA, Cunha TM, Arakawa NS, Lemos HP, Da Costa FB, Parada CA, Ferreira SH, Cunha FQ, Verri WA Jr (2007) Anti-inflammatory and analgesic effects of the sesquiterpene lactone budlein A in mice: inhibition of cytokine production-dependent mechanism. Eur J Pharmacol 562:155–163
Verri WA Jr, Schivo IR, Cunha TM, Liew FY, Ferreira SH, Cunha FQ (2004) Interleukin-18 induces mechanical hypernociception in rats via endothelin acting on ETB receptors in a morphine-sensitive manner. J Pharmacol Exp Ther 310:710–717
Verri WA Jr, Molina RO, Schivo IR, Cunha TM, Parada CA, Poole S, Ferreira SH, Cunha FQ (2005) Nociceptive effect of subcutaneously injected interleukin-12 is mediated by endothelin (ET) acting on ETB receptors in rats. J Pharmacol Exp Ther 315:609–615
Verri WA Jr, Cunha TM, Parada CA, Poole S, Cunha FQ, Ferreira SH (2006a) Hypernociceptive role of cytokines and chemokines: targets for analgesic drug development? Pharmacol Ther 112:116–138
Verri WA Jr, Cunha TM, Parada CA, Wei XQ, Ferreira SH, Liew FY, Cunha FQ (2006b) IL-15 mediates immune inflammatory hypernociception by triggering a sequential release of IFN-γ, endothelin, and prostaglandin. Proc Natl Acad Sci U S A 103:9721–9725
Verri WA Jr, Cunha TM, Parada CA, Poole S, Liew FY, Ferreira SH, Cunha FQ (2007a) Antigen-induced inflammatory mechanical hypernociception in mice is mediated by IL-18. Brain Behav Immun 21:535–543
Verri WA Jr, Cunha TM, Poole S, Ferreira SH, Cunha FQ (2007b) Cytokine inhibitors and pain control. Rev Bras Reumatol 47(5):341–353
Verri WA Jr, Cunha TM, Ferreira SH, Wei X, Leung BP, Fraser A, McInnes IB, Liew FY, Cunha FQ (2007c) IL-15 mediates antigen-induced neutrophil migration by triggering IL-18 production. Eur J Immunol 37:3373–3380
Verri WA Jr, Cunha TM, Magro DA, Domingues AC, Vieira SM, Souza GR, Liew FY, Ferreira SH, Cunha FQ (2008a) Role of IL-18 in overt pain-like behaviour in mice. Eur J Pharmacol 588:207–212
Verri WA Jr., Guerrero AT, Fukada SY, Valerio DA, Cunha TM, Xu D, Ferreira SH, Liew FY, Cunha FQ (2008b) IL-33 mediates antigen-induced cutaneous and articular hypernociception in mice. Proc Natl Acad Sci U S A 105:2723–2728
Yanagisawa M, Kurihara H, Kimura S, Tomobe Y, Kobayashi M, Mitsui Y, Yazaki Y, Goto K, Masaki T (1988) A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332:411–415
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16:109–110
Zoja C, Benigni A, Renzi D, Piccinelli A, Perico N, Remuzzi G (1990) Endothelin and eicosanoid synthesis in cultured mesangial cells. Kidney Int 37:927–933
Acknowledgments
We thank Ieda R.S. Schivo, Giuliana B. Francisco, and Sérgio R. Rosa for technical assistance. We also thank Actelion Pharmaceuticals Ltd (Allschwil, Switzerland) for the generous gift of bosentan. This work was supported by grants from Fundação de Amparo à Pesquisa do Estado de São Paulo, Conselho Nacional de Pesquisa, and Coordenadoria de Aperfeiçoamento de Pessoal de Nível Superior, Brazil. WAVJ and TMC contributed equally to this work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Verri Jr, W.A., Cunha, T.M., Magro, D.A. et al. Targeting endothelin ETA and ETB receptors inhibits antigen-induced neutrophil migration and mechanical hypernociception in mice. Naunyn-Schmied Arch Pharmacol 379, 271–279 (2009). https://doi.org/10.1007/s00210-008-0360-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-008-0360-1