Abstract
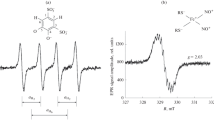
A variety of Cu reconstituted metallothioneins (MTs) containing different amounts of copper ions together with Cd7-MT free of copper were prepared and used in spin trapping experiments designed to show cadmium is not a Fenton active metal. A significant increase of the DMPO/·OH adduct was observed, with increased concentrations of the copper containing MTs, H2O2 enhanced DMPO/·OH adduct formation, catalase and the Cu (I) specific chelating agent bathocuproine, reduced DMPO/·OH adduct formation. These results suggest that Cu (I) and H2O2 both have important roles in the production of active species in these systems and cause DMPO/·OH formation. However, Cd7-MT showed no ability to cause generation of DMPO adducts with H2O2 seeming to indicate cadmium is not a Fenton metal. To test this hypothesis further trapping studies were run with added sulphite and lipid peroxide using both commercial MT and Cd7-MT since cadmium causes peroxidation in vivo. Commercial MT generates radicals with added sulphite and peroxide, Cd7-MT does not, demonstrating that cadmium is not a Fenton metal. These results help to explain the oxidative damage to DNA observed in the presence of MT and cadmium in vitro.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 3 February 1998 / Accepted: 24 July 1998
Rights and permissions
About this article
Cite this article
O'Brien, P., Salacinski, H. Evidence that the reactions of cadmium in the presence of metallothionein can produce hydroxyl radicals. Arch Toxicol 72, 690–700 (1998). https://doi.org/10.1007/s002040050562
Issue Date:
DOI: https://doi.org/10.1007/s002040050562